Abstract
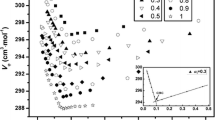
pH changes as a function of concentration for sodium dodecyl sulfate (SDS) and cetyltrimethylammonium bromide (CTAB) were observed by addition of 1 N HCl and 1 N KOH. pH values increased up to the critical micellar concentration (CMC) for the SDS/HCl system and decreased for the CTAB/KOH system. In the SDS/HCl and CTAB/KOH systems, the micellar phase had a fixed composition and was homogeneous and monodisperse above the CMC. However, in the SDS/KOH and CTAB/HCl systems, pH values increased continuously and gradually below and above the CMC, and the properties of the micellar phase changed as a function of concentration, giving rise to inhomogeneity and polydispersity.
Similar content being viewed by others
References
Stainsby, G., and A.E. Alexander, Factors Influencing Aggregation in Soap Solutions, Trans. Faraday Soc. 46:587–597 (1950).
Goddard, E.D., and G.C. Benson, Conductivity of Aqueous Solutions of Some Paraffin Chain Satls, Can. J. Chem. 35:986–991 (1957).
Shinoda, K., T. Nakagawa, B. Tamamushi, and T. Isemura, Colloidal Surfactants: Some Physicochemical Properties, Academic Press, New York, 1963, pp. 25–28.
Tanford, C., The Hydrophobic Effect: Formation of Micelles and Biological Membranes, 2nd edn. Wiley-Interscience, New York, 1980, pp. 63–70.
Elworthy, P.H., and K.J. Mysels, The Surface Tension of Sodium Dodecylsulfate Solutions and the Phase Separation Model of Micelle Formation, J. Colloid Interface Sci. 21:331–347 (1966).
Mukerjee, P., Ionic Partial Molal Volumes and Electrostrictions in Aqueous Solution, Adv. Colloid Interface Sci. 1:241–275 (1967).
Williams, R.J., J.N. Philips, and K.J. Mysels, The Critical Micelle Concentration of Sodium Lauryl Sulphate at 25°C, Trans. Faraday Soc. 51:728–737 (1955).
Goddard, E.D., C.A.J. Hoeve, and G.C.J. Benson, Heats of Micelle Formation of Paraffin Chain Salts in Water, J. Phys. Chem. 61:593–598 (1957).
Mysels, K.J., and L.H. Princen, Light Scattering by Some Lauryl Sulfate Solutions, J. Phys. Chem. 63:1696–1700 (1959).
Mysels, K.J., P. Mukerjee, and M. Abu-Hamdiyyah, The Activity of Association Colloids Above the Critical Micelle Concentration, J. Phys. Chem. 67:1943–1944 (1963).
Mukerjee, P., K.J. Mysels, and C.I. Dulin, Dilute Solutions of Amphipathic Ions I. Conductivity of Strong Salts and Dimerization, J. Phys. Chem. 62:1390–1396 (1958).
White, P., and G.C. Benson, The Temperature Variation of the Heat of Micellization of Potassium Octanoate in Aqueous Solutions, Trans. Faraday Soc. 55:1025–1029 (1959).
Cistola, D.P., and D.M. Small, On Micelle Formation and Phase Separation, J. Am. Chem. Soc. 112:3214–3215 (1990).
Cistola, D.P., J.A. Hamilton, and D.M. Small, Ionization and Phase Behavior of Fatty Acids in Water: Application of the Gibbs Phase Rule, Biochemistry 27:1881–1888 (1988).
Cistola, D.P., D. Atkinson, J.A. Hamilton, and D.M. Small, Phase Behavior and Bilayer Properties of Fatty Acids: Hydrated 1∶1 Acid Soaps, Biochemistry 25:2804–2812 (1986).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Topallar, H., Karadag, B. Mechanism of micelle formation in sodium dodecyl sulfate and cetyltrimethylammonium bromide. J Surfact Deterg 1, 49–51 (1998). https://doi.org/10.1007/s11743-998-0007-5
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11743-998-0007-5