Abstract
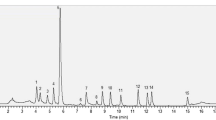
Leafy vegetables are the important components of our diet and are the source of several beneficial phytochemicals. A sensitive, simple analytical method is therefore required to precisely measure the phytochemical composition. A validated reversed phase high performance liquid chromatography with diode array detection (HPLC-DAD) method was developed to determine simultaneously carotenoids, chlorophylls and α-tocopherol composition of six leafy vegetables (B. compestris, B. rapa, B. juncea, M. neglecta, and two spinach varieties). Carotenoids were extracted and efficiently separated using a tertiary mobile gradient system of methanol–water, water and MTBE in 40 min on a reversed phase C18 column. The method was simple, precise, accurate and highly reproducible. Twelve carotenoids namely lutein and its three isomers, β-carotene-5,6-epoxide, neoxanthin, violaxanthin, two cis-isomers of zeaxanthin, 8-apo-carotenal, all-trans-β-carotene and its 13-cis-isomer; one fatty acid ester (β-cryptoxanthin ester); α-tocopherol and chlorophyll a & b were quantified in vegetable leaves. α-Tocopherol, neoxanthin, violaxanthin, lutein, 8-apo-carotenal, chlorophyll a and all-trans-β-carotene were present in higher amounts. Significant variations in the major compounds were observed in the selected vegetables. It was concluded that the developed method was highly sensitive, accurate and can be used to analyze carotenoids, chlorophylls and α-tocopherol simultaneously in leafy vegetables as well as in other plant leaves.



Similar content being viewed by others
Abbreviations
- HPLC:
-
High performance liquid chromatography
- DAD:
-
Diode array detector
- RP:
-
Reversed phase
- ACN:
-
Acetonitrite
- DCM:
-
Dichloromethane
- MeOH:
-
Methanol
- E.A:
-
Ethylacetate
- THF:
-
Tetrahydrofuran
- MTBE:
-
Methyl tertiary butyl ether
- BHT:
-
Butylated hydroxytoluene
- LOQ:
-
Limit of quantification
- LOD:
-
Limit of detection
- RSD:
-
Relative standard deviation
References
K. Jomova, M. Valko, Health protective effects of carotenoids and their interactions with other biological antioxidants. Eur. J. Med. Chem. 70, 102–110 (2013)
S. Takagi, Determination of green leaf carotenoids by HPLC. Agric. Biol. Chem. 49, 1211–1213 (1985)
S.R.A. Adewusi, J.H. Bradbury, Carotenoids in cassava: Comparison of open-column and HPLC methods of analysis. J. Sci. Food Agric. 62, 375–383 (1993)
H. Müller, Determination of the carotenoid content in selected vegetables and fruit by hplc and photodiode array detection. Zeitschrift für Lebensmitteluntersuchung und -Forschung A. 204, 88–94 (1997).
C.W. Huck, M. Popp, H. Scherz, G.K. Bonn, Development and evaluation of a new method for the determination of the carotenoid content in selected vegetables by hplc and HPLC-MS-MS. J. Chromatogr. Sci. 38, 441–449 (2000)
M. Dachtler, T. Glaser, K. Kohler, K. Albert, Combined HPLC-MS and HPLC-NMR on-line coupling for the separation and determination of lutein and zeaxanthin stereoisomers in spinach and in retina. Anal. Chem. 73, 667–674 (2001)
M. Edelenbos, L.P. Christensen, K. Grevsen, HPLC determination of chlorophyll and carotenoid pigments in processed green pea cultivars (pisum sativum l.). J. Agric. Food Chem. 49, 4768–4774 (2001)
M.C. De Sá, D.B. Rodriguez-Amaya, Optimization of HPLC quantification of carotenoids in cooked green vegetables—comparison of analytical and calculated data. J. Food Comp. Anal. 17, 37–51 (2004)
E. Larsen, L.P. Christensen, Simple saponification method for the quantitative determination of carotenoids in green vegetables. J. Agric. Food Chem. 53, 6598–6602 (2005)
K.L. Taylor, A.E. Brackenridge, M.A. Vivier, A. Oberholster, High-performance liquid chromatography profiling of the major carotenoids in arabidopsis thaliana leaf tissue. J. Chromatogr. A 1121, 83–91 (2006)
H. Kupper, S. Seibert, A. Parameswaran, Fast, sensitive, and inexpensive alternative to analytical pigment HPLC: Quantification of chlorophylls and carotenoids in crude extracts by fitting with gauss peak spectra. Anal. Chem. 79, 7611–7627 (2007)
J.G. Lashbrooke, P.R. Young, A.E. Strever, C. Stander, M.A. Vivier, The development of a method for the extraction of carotenoids and chlorophylls from grapevine leaves and berries for HPLC profiling. Aust. J. Grape Wine Res. 16, 349–360 (2010)
Z. Kotíková, M. Šulc, J. Lachman, V. Pivec, M. Orsák, K. Hamouz, Carotenoid profile and retention in yellow-, purple- and red-fleshed potatoes after thermal processing. Food Chem. 197, Part A, 992–1001 (2016).
K.T. Amorim-Carrilho, A. Cepeda, C. Fente, P. Regal, Review of methods for analysis of carotenoids. TrAC. Trends Anal. Chem. 56, 49–73 (2014)
M. Kimura, D.B. Rodriguez-Amaya, A scheme for obtaining standards and HPLC quantification of leafy vegetable carotenoids. Food. Chem. 78, 389–398 (2002)
B. Chauveau-Duriot, M. Doreau, P. Noziere, B. Graulet, Simultaneous quantification of carotenoids, retinol, and tocopherols in forages, bovine plasma, and milk: Validation of a novel UPLC method. Anal. Bioanal. Chem. 397, 777–790 (2010)
A.I.O. Barba, M.C. Hurtado, M.C.S. Mata, V.F. Ruiz, M.L.S.D. Tejada, Application of a UV-Vis detection-HPLC method for a rapid determination of lycopene and β-carotene in vegetables. Food Chem. 95, 328–336 (2006)
K.J. Mcgraw, E. Adkins-Regan, R.S. Parker, Anhydrolutein in the zebra finch: A new, metabolically derived carotenoid in birds. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 132, 811–818 (2002)
Zeb, Oxidation and formation of oxidation products of β-carotene at boiling temperature, Chem. Phys. Lipids 165, 277–281 (2012)
C.H. Azevedo-Meleiro, D.B. Rodriguez-Amaya, Qualitative and quantitative differences in carotenoid composition among cucurbita moschata, cucurbita maxima, and cucurbita pepo. J. Agric. Food Chem. 55, 4027–4033 (2007)
A.A. Updike, S.J. Schwartz, Thermal processing of vegetables increases cis isomers of lutein and zeaxanthin. J. Agric. Food Chem. 51, 6184–6190 (2003)
R. Aman, J. Biehl, R. Carle, J. Conrad, U. Beifuss, A. Schieber, Application of HPLC coupled with DAD, APCI-MS and NMR to the analysis of lutein and zeaxanthin stereoisomers in thermally processed vegetables. Food Chem. 92, 753–763 (2005)
J.M. Humphries, F. Khachik, Distribution of lutein, zeaxanthin, and related geometrical isomers in fruit, vegetables, wheat, and pasta products. J. Agric. Food Chem. 51, 1322–1327 (2003)
I. Krumbein, M. Schonhof, Schreiner, Composition and contents of phytochemicals (glucosinolates, carotenoids and chlorophylls) and ascorbic acid in selected brassica species (B. Juncea, B. Rapa subsp. Nipposinica var. Chinoleifera, B. Rapa subsp. Chinensis and B. Rapa subsp. Rapa). J. Appl. Bot. Food Qual. 79, 168–174 (2005)
Acknowledgements
The author is highly grateful for the financial assistant by the Higher Education Commission (HEC) Pakistan under National Research Program for Universities (NRPU), Project No. 2344.
Authors Contributions
The author is the sole contributor of the article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declare no conflict of interest to any body or institution.
Rights and permissions
About this article
Cite this article
Zeb, A. A simple, sensitive HPLC-DAD method for simultaneous determination of carotenoids, chlorophylls and α-tocopherol in leafy vegetables. Food Measure 11, 979–986 (2017). https://doi.org/10.1007/s11694-017-9472-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-017-9472-y