Abstract
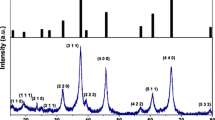
LiAl5O8 doped with Fe was synthesized by a propellant combustion route at furnace temperature of 773 K. The phosphor was characterized using powder x-ray diffraction, optical absorption, electron paramagnetic resonance (EPR) and photoluminescence spectroscopic techniques. The optical absorption spectrum exhibits a broad band at 242 nm characteristic of charge transfer between Fe3+–O2−. On excitation with 293 nm, emission band for the Fe3+ ion was observed at 687 nm. The CIE (International Commission on Illumination or Commission Internationale de l’Elcairage) coordinates for the system were evaluated adopting standard procedure which suggested that the system can be effective as a deep red emitting phosphor. The EPR spectrum of this phosphor exhibits a number of resonance signals characteristic of Fe3+ ions. The resonance signals at g = 3.16, 2.27 are attributed to Fe3+ present at tetrahedral site with an axial symmetry. The resonance signals at g = 1.98 and 1.43 are attributed to Fe3+ ions in octahedral site with an axial symmetry. Various EPR parameters such as the number of spins, Gibbs free energy, magnetic susceptibility, Curie constant and effective magnetic moment values are calculated and compared at room temperature and 110 K.
Similar content being viewed by others
References
T. Abritta, N.T. Melamed, J. MariaNeto, and F. Souza De Barros, J. Lumin. 18–19, 179 (1979).
X. Li, G. Jiang, S. Zhou, X. Wei, Y. Chen, C. KuiDuan, and M. Yin, Sensor Actuat. B Chem. 202, 1065 (2014).
V. Singh, R.P.S. Chakradhar, J.L. Rao, and D.-K. Kim, Mater. Chem. Phys. 110, 43 (2008).
X. Yang, G. Ning, X. Li, and Y. Lin, Mater. Lett. 61, 4694 (2007).
N. Suriyamurthy, B.S. Panigrahi, and A. Natarajan, Mat. Sci. Eng. A Struct.: A 403, 182 (2005).
S. Hashimoto, K. Hattori, K. Inoue, A. Nakahashi, S. Honda, and Y. Iwamoto, Mater. Res. Bull. 44, 70 (2009).
V. Singh and T.K. Gundu Rao, J. Solid State Chem. 181, 1387 (2008).
J. Kai, Y. Xing-Hai, Y. Ming-Xin, H. Wei-Shi, and H. Jing-Gen, Chin. J. Lumin. 24, 517 (2003).
S.S. Pitale, V. Kumar, I.M. Nagpure, O.M. Ntwaeaborwa, E. Coetsee, and H.C. Swart, J. Appl. Phys. 109, 013105 (2011).
Z. Mu, Y. Hu, L. Chen, X. Wang, and G. Ju, Radiat. Meas. 47, 426 (2012).
V. Singh, V.K. Rai, I. Ledoux-Rak, and H.Y. Kwak, Appl. Phys. B 97, 103 (2009).
C. Justin Raj, M.B. Lincoln, and S. Jerome Das, Cryst. Res. Technol. 43, 823 (2008).
S.C. Bhargava, J. Phys. C: Solid State Phys. 19, 7045 (1986).
M.M.C. Chou, C.C. Hsu, C.-Y. Lee, and C. Chen, Cryst. Growth Des. 10, 191 (2010).
S.S. Pitale, V. Kumar, I. Nagpure, O.M. Ntwaeaborwa, and H.C. Swart, Curr. Appl. Phys. 11, 341 (2011).
G.N. Nikhare, S.C. Gedam, and S.J. Dhoble, J. Lumin. 137, 290 (2013).
V. Singh, R.P.S. Chakradhar, J.L. Rao, and H.-Y. Kwak, Solid State Sci. 11, 870 (2009).
D. Pan, D. Yuan, H. Sun, X. Duan, C. Luan, S. Guo, Z. Li, and L. Wang, Mater. Chem. Phys. 96, 317 (2006).
D. Deng, H. Ma, S. Xu, Q. Wang, L. Huang, S. Zhao, H. Wang, and C. Li, J. Non-Cryst. Solids. 357, 1426 (2011).
X. Duan and D. Yuan, J. Non-Cryst. Solids. 351, 2348 (2005).
M. Aoyama, Y. Amano, K. Inoue, S. Honda, S. Hashimoto, and Y. Iwamoto, J. Lumin. 135, 211 (2013).
K.E. Fox, T. Furukawa, and W.B. White, J. Am. Ceram. Soc. 64, C42 (1981).
D.T. Palumbo, J. Lumin. 4, 89 (1971).
S.A. Brawer and W.B. White, J. Mater. Sci. 13, 907 (1978).
W. Lu, X. Gong, M. Nan, Y. Liu, S. Shuang, and C. Dong, Anal. Chim. Acta 898, 116 (2015).
E.L. Que, D.W. Domaille, and C.J. Chang, Chem. Rev. 108, 1517 (2008).
M. Parker and H. Bothwick, Plant Physiol. 24, 345 (1949).
V. Singh, R.P.S. Chakradhar, J.L. Rao, and J.-J. Zhu, Mater. Chem. Phys. 111, 143 (2008).
V. Singh, T.K. Gundu Rao, and J.-J. Zhu, J. Lumin. 128, 583 (2008).
V. Singh, T.K. Gundu Rao, and D.-K. Kim, Radiat. Meas. 43, 1198 (2008).
T.R.N. Kutty and M. Nayak, J. Alloys Compds. 269, 75 (1998).
W. Shu, R.F. Qiang, S. Xiao, X. Yang, and J.W. Ding, J. Alloys Compds. 509, 3886 (2011).
N.T. Melamed, J. Lumin. 1–2, 348 (1970).
V. Singh, G. Sivaramaiah, J.L. Rao, N. Singh, A.K. Srivastava, P.K. Singh, S.U. Pawar, H. Gao, and P. Mardina, J. Mater. Sci.: Mater. Electron. 27, 4494 (2016).
D. Jia, D. Hunter, and J. Wilhelm, ECS Trans. 19, 6 (2008).
A.P. Jadhav, A. Pawar, U. Pal, B.K. Kim, and Y.S. Kang, Sci. Adv. Mater. 4, 597 (2012).
B. Bleany and K.W.H. Stevens, Rep. Prog. Phys. 16, 108 (1953).
R.M. Golding, T. Singhasuwich, and W.C. Tennant, Mol. Phys. 34, 1343 (1977).
Y. Pan, M. Mao, and J. Lin, Canad. Mineral. 47, 933 (2009).
I. Ardelean, P. Pascuta, and L.V. Giurgiu, Int J. Mod. Phys B. 17, 3049 (2003).
E. Burzo, I. Ardelean, and I. Ursu, J. Mater. Sci. 15, 581 (1980).
G. Sivaramaiah, J. Lin, and Y. Pan, Phys. Chem. Miner. 38, 159 (2011).
G. Sivaramaiah and Y. Pan, Phys. Chem. Miner. 39, 515 (2012).
E. Boseman and D. Schoemaker, C. R. Acad. Sci. Paris 252, 1931 (1961).
R. Kedzie, D. Lyons, and M. Kestigian, Phys. Rev. 138, A918 (1965).
J.A. Weil and J.R. Bolton, Electron Paramagnetic Resonance: Elementary Theory and Practical Applications, 2nd ed. (New York: Wiley, 2007), p. 545.
A.J. Dekker, Solid State Physics (Groningen: Department of Electrical Engineering, University of Groningen, 2006).
C. Kittel, Introduction to Solid State Physics, 7th ed. (New York: Wiley, 1996).
M. Borgheresi, F. Di Benedetto, A. Caneschi, G. Pratesi, M. Romanelli, and L. Sorace, Phys. Chem. Miner. 34, 609 (2007).
A.M. Hashem, H.M. Abuzeid, D. Mikhailova, H. Ehrenberg, A. Mauger, and C.M. Julien, J. Mater. Sci. 47, 2479 (2012).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Singh, V., Sivaramaiah, G., Rao, J.L. et al. Optical and EPR Spectroscopic Studies of Deep Red Light Emitting Fe-Doped LiAl5O8 Phosphor Prepared Via Propellant Combustion Route. J. Electron. Mater. 46, 1525–1531 (2017). https://doi.org/10.1007/s11664-016-5192-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-016-5192-z