Abstract
Objective
To observe the effect of norcantharidin (NCTD) on collagen-induced arthritis (CIA) rats.
Methods
Sixty Sprague-Dawley(SD) rats were randomly divided into 6 groups (n=10): normal group, CIA model group(model group), NCTD low-dose group [1.35 mg/(kg•d)], NCTD middle-dose group [2.7 mg/(kg•d)], NCTD high-dose group [5.4 mg/(kg•d)] and methotrexate (MTX) group [1.8 mg/(kg/w)]. Anesthetized rats were sacrificed by luxation of cervical vertebra after 4 weeks of administration. The arthritis scores were evaluated twice a week. The pathological changes in the ankle joints of rats were observed by hematoxylin-eosin (H&E) staining. The serum levels of interleukin (IL) 1β, IL-6, tumor necrosis factor (TNF)-α, vascular endothelial growth factor (VEGF), IL-17 and transform growth factor (TGF) β were detected by enzyme linked immunosorbent assay (ELISA). The mRNA expression of retinoid-related orphan nuclear receptorγt (RORγt) and forkhead box P3 (Foxp3) in peripheral blood lymphocytes were confirmed by real-time polymerase chain reaction.
Results
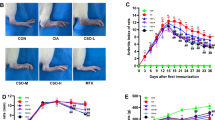
MTX and high-dose NCTD not only decreased the arthritis scores but also alleviated the pathological changes in CIA rats’ ankle joints compared with the model group (P<0.05 or P<0.01). All doses of NCTD significantly inhibited the serum levels of IL-6, IL-17 and TNF-α in CIA rats (P<0.05). Only middle- and high-dose of NCTD prominently decreased serum IL-1β and TGF-β levels of CIA rats (P<0.05). However, NCTD has no effect on vascular endothelial growth factor (VEGF) level in CIA rats. The Foxp3 mRNA expression in all NCTD groups were increased significantly than in the model group (P<0.05). The mRNA expression of RORγt in NCTD high-dose group was decreased apparently in comparison with the model group (P<0.05).
Conclusions
NCTD showed therapeutic effect on CIA rats by inhibition of cytokines and regulation of Th17/Treg cells.
Similar content being viewed by others
References
Smolen JS, Aletaha D, McInnes IB. Rheumatoid arthritis. Lancet 2016; 388:2023–2038.
Firestein GS. Evolving concepts of rheumatoid arthritis. Nature 2003;423:356–361.
Fan LJ, Cui ZY, Sun SZ. Clinical observation on vesiculation moxibustion with cantharides and Indigo Naturalis Paste plus Chinese medication for rheumatoid arthritis. Shanghai J Acu-Mox (Chin) 2012; 31:161–163.
Xiong YK, Yu B. Experimental studies on anti-asthmatic effect of composite Tremella Mesenterica Decoction. J Zhejiang College Tradit Chin Med (Chin) 2000;24:71–72.
Aschner B. Successful therapy of non-infectious rheumatoid arthritis; constitutional therapy, plus the use of cantharides and vesicants. Hippokrates 1950; 21:452–458.
Puerto Galvis CE, Vargas Méndez LY, Kouznetsov VV. Cantharidin-based small molecules as potential therapeutic agents. Chem Biol Drug Des 2013;82:477–499.
Deng L, Tang S. Norcantharidin analogues: a patent review (2006–2010). Expert Opin Ther Pat 2011;21:1743–1753.
McInnes IB, Schett G. Cytokines in the pathogenesis of rheumatoid arthritis. Nat Rev Immunol 2007;7:429–442.
Alunno A, Manetti M, Caterbi S, Ibba-Manneschi L, Bistoni O, Bartoloni E, et al. Altered immunoregulation in rheumatoid arthritis: the role of regulatory T cells and proinflammatory Th17 cells and therapeutic implications. Mediators Inflamm 2015; 2015:751793.
Nistala K, Wedderburn LR. Th17 and regulatory T cells: rebalancing pro- and anti-inflammatory forces in autoimmune arthritis. Rheumatology (Oxford) 2009;48:602–606.
Kimura A, Kishimoto T. IL-6: regulator of Treg/Th17 balance. Eur J Immunol 2010;40:1830–1835.
Ito H, Yamada H, Shibata TN, Mitomi H, Nomoto S, Ozaki S. Dual role of interleukin-17 in pannus growth and osteoclastogenesis in rheumatoid arthritis. Arthritis Res Ther 2011;13:R14.
Isozaki T, Kasama T, Takahashi R, Odai T, Wakabayashi K, Kanemitsu H, et al. Synergistic induction of CX3CL1 by TNF alpha and IFN gamma in osteoblasts from rheumatoid arthritis: involvement of NF-kappa B and STAT-1 signaling pathways. J Inflam Res 2008;1:19–28.
Kim SJ, Chen Z, Chamberlain ND, Volin MV, Swedler W, Volkov S, et al. Angiogenesis in rheumatoid arthritis is fostered directly by toll-like receptor 5 ligation and indirectly through interleukin-17 induction. Arthritis Rheum 2013; 65:2024–2036.
Komatsu N, Takayanagi H. Regulatory T cells in arthritis. Prog Mol Biol Transl Sci 2015;136:207–215.
Hsu YH, Hsieh PP, Chang MS. Interleukin-19 blockade attenuates collagen-induced arthritis in rats. Rheumatology (Oxford) 2012;51:434–442.
Zhu X, Zhang J, Huo R, Lin J, Zhou Z, Sun Y, et al. Evaluation of the efficacy and safety of different Tripterygium preparations on collagen-induced arthritis in rats. J Ethnopharmacol 2014;158 Pt A:283–290.
Fan D, He X, Bian Y, Guo Q, Zheng K, Zhao Y, et al. Triptolide modulates TREM-1 signal pathway to inhibit the inflammatory response in rheumatoid arthritis. Int J Mol Sci 2016;17:498.
Xiao J, Li G, Hu J, Qu L, Ma D, Chen Y. Anti-inflammatory effects of recombinant human PDCD5 (rhPDCD5) in a rat collagen-induced model of arthritis. Inflammation 2015;38:70–78.
Wu L, Luo LH, Zhang YX, Li Q, Xu B, Zhou GX, et al. Alteration of Th17 and Treg cells in patients with unexplained recurrent spontaneous abortion before and after lymphocyte immunization therapy. Reprod Biol Endocrinol 2014;12:74.
Li C, Yuan J, Zhu YF, Yang XJ, Wang Q, Xu J, et al. Imbalance of Th17/Treg in different subtypes of autoimmune thyroid diseases. Cell Physiol Biochem 2016;40:245–252.
Maini RN, Taylor PC. Anti-cytokine therapy for rheumatoid arthritis. Annu Rev Med 2000; 51:207–229.
McInnes IB, Liew FY. Cytokine networks-towards new therapies for rheumatoid arthritis. Nat Clin Pract Rheumatol 2005;1:31–39.
Maruotti N, Cantatore FP, Ribatti D. Putative effects of potentially anti-angiogenic drugs in rheumatic diseases. Eur J Clin Pharmacol 2014;70:135–140.
Zhang L, Ji Q, Liu X, Chen X, Chen Z, Qiu Y, et al. Norcantharidin inhibits tumor angiogenesis via blocking VEGFR2/MEK/ERK signaling pathways. Cancer Sci 2013;104:604–610.
Bettelli E, Carrier Y, Gao W, Korn T, Strom TB, Oukka M, et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 2006;441:235–238.
Lubberts E, Koenders MI, van den Berg WB. The role of T-cell interleukin-17 in conducting destructive arthritis: lessons from animal models. Arthritis Res Ther 2005;7:29–37.
Weaver CT, Hatton RD, Mangan PR, Harrington LE. IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu Rev Immunol 2007; 25:821–852.
Steinman L. A brief history of T(H)17, the first major revision in the T(H)1/T(H)2 hypothesis of T cell-mediated tissue damage. Nat Med 2007;13:139–145.
Metawi SA, Abbas D, Kamal MM, Ibrahim MK. Serum and synovial fluid levels of interleukin-17 in correlation with disease activity in patients with RA. Clin Rheumatol 2011;30:1201–1207.
Chen DY, Chen YM, Chen HH, Hsieh CW, Lin CC, Lan JL. Increasing levels of circulating Th17 cells and interleukin-17 in rheumatoid arthritis patients with an inadequate response to anti-TNF-alpha therapy. Arthritis Res Ther 2011;13:R126.
Wing K, Sakaguchi S. Regulatory T cells exert checks and balances on self tolerance and autoimmunity. Nat Immunol 2010;11:7–13.
Sempere-Ortells JM, Pérez-García V, Marín-Alberca G, Peris-Pertusa A, Benito JM, Marco FM, et al. Quantification and phenotype of regulatory T cells in rheumatoid arthritis according to disease activity score-28. Autoimmunity 2009;42:636–645.
Kawashiri SY, Kawakami A, Okada A, Koga T, Tamai M, Yamasaki S, et al. CD4+CD25(high)CD127(low/-) Treg cell frequency from peripheral blood correlates with disease activity in patients with rheumatoid arthritis. J Rheumatol 2011;38:2517–2521.
Chen W, Jin W, Hardegen N, Lei KJ, Li L, Marinos N, et al. Conversion of peripheral CD4+CD25- naive T cells to CD4+CD25+ regulatory T cells by TGF-beta induction of transcription factor Foxp3. J Exp Med 2003;198:1875–1886.
Shevach EM, Thornton AM. tTregs, pTregs, and iTregs: similarities and differences. Immunol Rev 2014;259:88–102.
Yuan X, Cheng G, Malek TR. The importance of regulatory T-cell heterogeneity in maintaining self-tolerance. Immunol Rev 2014;259:103–114.
Ivanov II, McKenzie BS, Zhou L, Tadokoro CE, Lepelley A, Lafaille JJ, et al. The orphan nuclear receptor RORgammat directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 2006;126:1121–1133.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shen, Hb., Huo, Zj., Bai, Yj. et al. Protective Effect of Norcantharidin on Collagen-Induced Arthritis Rats. Chin. J. Integr. Med. 24, 278–283 (2018). https://doi.org/10.1007/s11655-017-2792-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-017-2792-2