Abstract
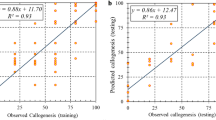
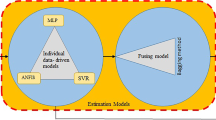
Trachyspermum ammi (L.) Sprague (Ajowan) is an endangered medicinal plant with useful pharmaceutical properties. Ex situ conservation of this medicinal plant needs the development of an in vitro regeneration protocol using somatic embryogenesis. In the present study, a high-precision image-processing approach was successfully applied to measure physical properties of embryogenic callus. Explant age and the concentrations of 2,4-dichlorophenoxyacetic acid (2,4-D), kinetin (Kin), and sucrose were used as inputs, and an artificial intelligence technique was applied to predict physical properties of embryogenic callus, and the number of somatic embryos produced. Artificial neural network (ANN) models were tested to find the best combinations of input variables that affected output variables. The lower values of root mean square error, and mean absolute error, and the highest values of determination coefficient, were achieved when all four input variables were applied to predict the number of somatic embryos, the area of the callus, the perimeter of the callus, the Feret diameter of the callus, the roundness of the callus, and the true density of the callus in ANN models. The highest measured and predicted number of somatic embryos were achieved from the interaction of 15-d-old explants × 1.5 mg L−1 2,4-D × 0.5 mg L−1 Kin × 2.5% (w/v) sucrose. Based on sensitivity analysis, the 2,4-D concentration was the most important component in the culture medium that affected the number of somatic embryos and physical properties of the embryogenic callus tissue.




Similar content being viewed by others
References
Ahmadi SH, Sepaskhah AR, Andersen MN, Plauborg F, Jensen JR, Hansen S (2014) Modeling root length density of field grown potatoes under different irrigation strategies and soil textures using artificial neural networks. Field Crops Res 162:99–107. https://doi.org/10.1016/j.fcr.2013.12.008
Arab MM, Yadollahi A, Shojaeiyan A, Ahmadi H (2016) Artificial neural network genetic algorithm as powerful tool to predict and optimize in vitro proliferation mineral medium for G× N15 rootstock. Front Plant Sci 7:1526. https://doi.org/10.3389/fpls.2016.01526
Ashraf M, Orooj A (2006) Salt stress effects on growth, ion accumulation and seed oil concentration in an arid zone traditional medicinal plant ajwain (Trachyspermum ammi [L.] Sprague). J Arid Environ 64(2):209–220. https://doi.org/10.1016/j.jaridenv.2005.04.015
Boskabady MH, Alitaneh S, Alavinezhad A (2014) Carum copticum L.: a herbal medicine with various pharmacological effects. Biomed Res Int 11p, https://doi.org/10.1155/2014/569087. Cited 02 February 2017
Chegini GR, Khazaei J, Ghobadian B, Goudarzi AM (2008) Prediction of process and product parameters in an orange juice spray dryer using artificial neural networks. J Food Eng 84(4):534–543. https://doi.org/10.1016/j.jfoodeng.2007.06.007
Dalkani M, Hassani A, Darvishzadeh R (2012) Determination of the genetic variation in Ajowan (Carum Copticum L.) populations using multivariate statistical techniques. Rev Ciênc Agron 43(4):698–705. https://doi.org/10.1590/S1806-66902012000400011
Emamgholizadeh S, Parsaeian M, Baradaran M (2015) Seed yield prediction of sesame using artificial neural network. Eur J Agron 68:89–96. https://doi.org/10.1016/j.eja.2015.04.010
Erzin Y, Rao H, Singh D (2008) Artificial neural network models for predicting soil thermal resistivity. Int J Theor Sci Algor 47(10):1347–1358. https://doi.org/10.1016/j.ijthermalsci.2007.11.001
Govindaraju RS (2000a) Artificial neural networks in hydrology. I: preliminary concepts. J Hydrol Eng 5:115–123
Govindaraju RS (2000b) Artificial neural networks in hydrology. II: hydrologic applications. J Hydro Eng 5:124–137
Gupta SD, Pattanayak AK (2017) Intelligent image analysis (IIA) using artificial neural network (ANN) for non-invasive estimation of chlorophyll content in micropropagated plants of potato. In: In Vitro Cell Dev Biol-Plant, pp 1–7. https://doi.org/10.1007/s11627-017-9825-6
Jamshidi S, Yadollahi A, Ahmadi H, Arab MM, Eftekhari M (2016) Predicting in vitro culture medium macro-nutrients composition for pear rootstocks using regression analysis and neural network models. Front Plant Sci 7:274. https://doi.org/10.3389/fpls.2016.00274
Kumar M, Raghuwanshi N.S., Singh R (2011) Artificial neural networks approach in evapotranspiration modeling: a review. Irrig Sci 29:11–25, 1, https://doi.org/10.1007/s00271-010-0230-8
Mansouri A, Fadavi A, Mortazavian SMM (2015) Effects of length and position of hypocotyl explants on Cuminum cyminum L. callogensis by image processing analysis. Plant Cell Tissue Organ Cult 121(3):657–666. https://doi.org/10.1007/s11240-015-0736-0
Mansouri A, Fadavi A, Mortazavian SMM (2016) An artificial intelligence approach for modeling volume and fresh weight of callus—a case study of cumin (Cuminum cyminum L.) J Theor Biol 397:199–205. https://doi.org/10.1016/j.jtbi.2016.03.009
Mehrotra S, Prakash O, Mishra BN, Dwevedi B (2008) Efficiency of neural networks for prediction of in vitro culture conditions and inoculum properties for optimum productivity. Plant Cell Tissue Organ Cult 95(1):29–35. https://doi.org/10.1007/s11240-008-9410-0
Mokarram M, Bijanzadeh E (2016) Prediction of biological and grain yield of barley using multiple regression and artificial neural network models. Aust J Crop Sci 10(6):895–903. https://doi.org/10.21475/ajcs.2016.10.06.p7634
Moon HK, Lee H, Paek KY, Park SY (2015) Osmotic stress and strong 2, 4-D shock stimulate somatic-to-embryogenic transition in Kalopanax septemlobus (Thunb.) Koidz. Acta Physiol Plant 37:1–9
Movagharnejad K, Nikzad M (2007) Modelling of tomato drying using artificial neural network. Comput Electron Agric 59(1-2):78–85. https://doi.org/10.1016/j.compag.2007.05.003
Naroui Rad MR, Koohkan S, Fanaei HR, Pahlavan Rad MR (2015) Application of artificial neural networks to predict the final fruit weight and random forest to select important variables in native population of melon (Cucumis melo L.) Sci Hortic 181:108–112. https://doi.org/10.1016/j.scienta.2014.10.025
Nezami Alanagh E, Garoosi G, Haddad R, Maleki S, Landı’n M, Gallego P.P (2014) Design of tissue culture media for efficient Prunus rootstock micropropagation using artificial intelligence models. Plant Cell Tissue Organ Cult 117:349–359, 3, https://doi.org/10.1007/s11240-014-0444-1
Niazian M, Sadat Noori SA, Galuszka P, Tohidfar M, Mortazavian SMM (2017a) Genetic stability of regenerated plants via indirect somatic embryogenesis and indirect shoot regeneration of Carum copticum L. Ind Crop Prod 97:330–3307. https://doi.org/10.1016/j.indcrop.2016.12.044
Niazian M, Sadat Noori S, Tohidfar M, Mortazavian S.M.M (2017b) Essential oil yield and agro-morphological traits in some Iranian ecotypes of Ajowan (Carum copticum L.). J Essent Oil Bear Plant 20:1151–1156, 4, https://doi.org/10.1080/0972060X.2017.1326849
Nourani V, Sayyah Fard M (2012) Sensitivity analysis of the artificial neural network outputs in simulation of the evaporation process at different climatologic regimes. Adv Eng Softw 47(1):127–146. https://doi.org/10.1016/j.advengsoft.2011.12.014
Olofsdotter M (1993) Image processing: a non-destructive method for measuring growth in cell and tissue culture. Plant Cell Rep 12(4):216–219. https://doi.org/10.1007/BF00237057
Panda SS, Ames DP, Panigrahi S (2010) Application of vegetation indices for agricultural crop yield prediction using neural network techniques. Remote Sens 2(3):673–696. https://doi.org/10.3390/rs2030673
Prasad VSS, Gupta SD (2008) Photometric clustering of regenerated plants of gladiolus by neural networks and its biological validation. Comput Electron Agric 60(1):8–17. https://doi.org/10.1016/j.compag.2007.05.006
Prasad A, Prakash O, Mehrotra S, Khan F, Mathur AK, Mathur A (2017) Artificial neural network-based model for the prediction of optimal growth and culture conditions for maximum biomass accumulation in multiple shoot cultures of Centella asiatica. Protoplasma 254(1):335–341. https://doi.org/10.1007/s00709-016-0953-3
Ramakrishna D, Shasthree T (2016) High efficient somatic embryogenesis development from leaf cultures of Citrullus colocynthis. Physiol Mol Biol Plants 22(2):279–285. https://doi.org/10.1007/s12298-016-0357-z
Sadat Noori SA, Norouzi M, Karimzadeh G, Shirkool K, Niazian M (2017) Effect of colchicine-induced polyploidy on morphological characteristics and essential oil composition of ajowan (Trachyspermum ammi L.) Plant Cell Tissue Organ Cult 130(3):543–551. https://doi.org/10.1007/s11240-017-1245-0
Safa M, Samarasinghe S, Nejat M (2015) Prediction of wheat production using artificial neural networks and investigating indirect factors affecting it: case study in Canterbury province, New Zealand. J Agric Sci Tech 17:791–803
Satish L, Rency AS, Rathinapriya P, Ceasar SA, Pandian S, Rameshkumar R, Rao TB, Balachandran SM, Ramesh M (2016) Influence of plant growth regulators and spermidine on somatic embryogenesis and plant regeneration in four Indian genotypes of finger millet (Eleusine coracana (L.) Gaertn). Plant Cell Tissue Organ Cult 124(1):15–31. https://doi.org/10.1007/s11240-015-0870-8
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH image to ImageJ: 25 years of image analysis. Nat Methods 9(7):671–675. https://doi.org/10.1038/nmeth.2089
Sharma K, Dubey S (2011) Biotechnology and conservation of medicinal plants. J Expl Sci 2:60–61
Sharma S, Rathi N, Kamal B, Pundir D, Kaur B, Arya S (2010) Conservation of biodiversity of highly important medicinal plants of India through tissue culture technology—a review. Agric Biol J North Am 1(5):827–833. https://doi.org/10.5251/abjna.2010.1.5.827.833
Somarante S, Seneviratne G, Coomaraswamy U (2005) Prediction of soil organic carbon across different land-use patterns: a neural network approach. Soil Sci Soc Am J 69:1580–1589
Tufail M, Ormsbee LE, Teegavarapu R (2008) Artificial intelligence-based inductive models for prediction and classification of fecal coliform in surface waters. J Environ Eng 134(9):789–799. https://doi.org/10.1061/(ASCE)0733-9372(2008)134:9(789)
Uozumi N, Yoshinoa T, Shiotanib S, Sueharaa KI, Araib F, Fukudab T, Kobayashi T (1993) Application of image analysis with neural network for plant somatic embryo culture. J Ferment Bioeng 76(6):505–509. https://doi.org/10.1016/0922-338X(93)90249-8
Wu J, Zhang X, Nie Y, Jin S, Liang S (2004) Factors affecting somatic embryogenesis and plant regeneration from a range of recalcitrant genotypes of Chinese cottons (Gossypium hirsutum L.) In: In Vitro Cell Dev Biol—Plant, vol 40, pp 371–375
Zhang C, Timmis R, WS H (1999) A neural network based pattern recognition system for somatic embryos of Douglas fir. Plant Cell Tissue Organ Cult 56(1):25–35. https://doi.org/10.1023/A:1006287917534
Zhang H, Hu H, Zhang X, Zhu L, Zheng K, Jin Q, Zeng F (2011) Estimation of rice neck blasts severity using spectral reflectance based on BP-neural network. Acta Physiol Plant 33(6):2461–2466. https://doi.org/10.1007/s11738-011-0790-0
Acknowledgements
The authors are thankful to the Research Institute of Forests and Rangelands of Iran for procuring the seeds, and also grateful to Dr. M.H Asare, the secretary of science and technological development staff of medicinal plants and traditional medicine of Islamic Republic of Iran, for his kind support of ajowan project. The first author is thankful to Piama Svoboda for her kind assistance in the English language editing of the manuscript.
Funding
This work was financially supported by the Biotechnology Development Council of the Islamic Republic of Iran [grant number 950620].
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Randall P. Niedz
Electronic supplementary material
ESM 1
(DOCX 24 kb)
Rights and permissions
About this article
Cite this article
Niazian, M., Sadat-Noori, S.A., Abdipour, M. et al. Image Processing and Artificial Neural Network-Based Models to Measure and Predict Physical Properties of Embryogenic Callus and Number of Somatic Embryos in Ajowan (Trachyspermum ammi (L.) Sprague). In Vitro Cell.Dev.Biol.-Plant 54, 54–68 (2018). https://doi.org/10.1007/s11627-017-9877-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-017-9877-7