Abstract
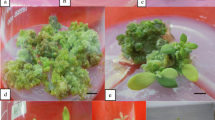
A method was developed to initiate multiple shoots from mature nodal explants of Sida cordifolia Linn. High frequency of regeneration was achieved on Murashige and Skoog (MS) medium supplemented with 2.0 mg l−1 6-benzylaminopurine, 0.5 mg l−1 α-naphthalene acidic acid, 1.0 mg l−1 adenine sulfate, and 10% (v/v) coconut milk. Multiple shoots were initiated within 21 d and the above media was capable of inducing the formation of more than 20 shoots from each explant. Regenerated shoots were successfully rooted on half-strength MS medium supplemented with 2.0 mg l−1 indole-3-butyric acid and 3% (w/v) sucrose. Rooted plantlets were established in soil. The regenerated plantlets showed no morphological differences from the parent material. This protocol could be useful for germplasm conservation, cultivation, and genetic improvement of S. cordifolia.


Similar content being viewed by others
References
Anonymous. The Wealth of India. A Dictionary of Indian raw materials and industrial products, Newdelhi, Publication and Information Directorate CSIR. 323–324; 1988.
Asha B; Bannerjee NR. Polarographic studies on active constituents of Sida cordifolia. Cur. Sci. 54: 690–692; 1985.
Balachandran SM; Bhat SR; Chandal KPS. In vitro clonal multiplication of turmeric (Curcuma Spp.) and ginger (Zingiber officinale Rose). Plant Cell Rep. 8: 521–524; 1990.
Bhat SR; Chandal KPS; Malik SK. Plant regeneration from various explants of cultivated Piper species. Plant Cell Rep. 14: 398–402; 1995.
Chen CC; Chen SJ; Sagare AP; Tsay HS. Adventitious shoot regeneration from stem internode explants of Adenophora triphylla (Thunb.) A. DC. (Campanulaceae)—An important medicinal herb. Bot. Bull. Acad. Sin. 42: 1–7; 2001.
Chopra RN; Handa KL; Kapur LD. Chopra’s indigenous drugs of India 2nd Edn., Academic Publishers. 409; 1958.
Das G; Rout GR. Direct plant regeneration from leaf explants of Plumbago species. Plant Cell Tiss. Organ Cult. 68: 311–314; 2002.
George EF; Sherrington PD. In vitro: Plant propagation by tissue culture, England, Eversley, Exegetics Ltd. 39–71; 1984.
Ghosh S; Dutt A. Chemical examination of Sida cordifolia Linn. J. Ind. Chem. Soc. 7: 825–820; 1930.
Grigoriadou K; Vasilakakis M; Eleftheriou EP. In vitro propagation of the Greek olive cultivar ‘Chondrolia Chalkidikis’. Plant Cell Tiss. Organ Cult. 71: 47–54; 2002.
Gunatilaka AAL; Sotheeswaran S; Balasubramaniam S; Chandrasekara AI; Badrasriyani HT. Studies on medicinal plants of Sri Lanka. Planta Med. 39: 66–72; 1980.
Handique PJ; Bora P. In vitro regeneration of a medicinal plant—Houttuynia cordata Thunb. from nodal explants. Cur. Sci. 76: 1245–1247; 1999.
Hasson E; Poljakoff-Mayber A. Callus culture from hypocotyls of Kosteletzkya virginica (L.) seedlings. Plant Cell Tiss. Organ Cult. 43: 279–285; 1995.
Herath SP; Suzuki T; Hattori K. Multiple shoot regeneration from young shoots of kenaf (Hibiscus cannabinus). Plant Cell Tiss. Organ Cult. 77: 49–53; 2004.
Hu CY; Wang PJ. Meristem, shoot tip and bud cultures. In Evans DA, Sharp WR, Ammirato PV and Yamada Y (eds), Handbook of plant cell culture: Techniques for propagation and breeding vol. I. New York, Macmillan Publishing Co. 177–277; 1983.
Kanth VR; Diwan PV. Analgesic, antiinflammatory and hypoglycaemic activities of Sida cordifolia. Phytother Res. 13: 75–77; 1999.
Kirtikar KR; Basu BD. Indian Medicinal Plants. 2nd Edn., Bishen Singh Mahendra Pal Singh, Dehradun. 312; 1980.
Lundergan CA; Janick J. Regulation of apple shoot proliferation and growth in vitro. Hort. Res. 20: 19–24; 1980.
Mechanda SM; Baum BR; Johnson DA; Arnason JT. Direct shoot regeneration from leaf segments of mature plants of Echinacea purpurea (L.) Moench. In Vitro Cell. Dev. Biol.-Plant. 39: 505–509; 2003.
Medeiros IA; Santos MR; Nascimento NM; Duarte JC. Cardiovascular effects of Sida cordifolia leaves extracts in rats. Fitoterapia. 77: 19–27; 2006.
Murashige T; Skoog F. A revised for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant. 15: 473–497; 1962.
Patnaik J; Debata BK. Micropropagation of Hemidesmus indicus (L.) R. Br. through axillary bud culture. Plant Cell Rep. 15: 427–430; 1996.
Purohit SD; Dave A; Kukda G. Micropropagation of safed musli (Chlorophytum borivilianum), a rare Indian medicinal herb. Plant Cell Tiss. Organ Cult. 39: 93–96; 1994.
Raghav Swamy BV; Himabindu K; Lakshmi sita G. In vitro micropropagatipon of elite rosewood (Dalbergia latifolia Roxb.) Plant Cell Rep. 11: 126–131; 1992.
Rahaman MA; Blake J. Factors affecting in vitro proliferation and rooting of shoots of jackfruit (Artocarpus heterophyllum Lam). Plant Cell Tiss. Organ Cult. 13: 179–187; 1988.
Rout GR; Saxena C; Samantaray S; Das P. Rapid plant regeneration from callus cultures of Plumbago zeylanica. Plant Cell Tiss. Organ Cult. 56: 47–51; 1999.
Rout GR; Samantaray S; Das P. In vitro manipulation and propagation of medicinal plants. Biotechnol. Advances. 18: 91–120; 2000.
Sutradhar RK; Rahman AKMM; Ahmad M; Bachar SC; Saha A; Guha SK. Analgesic and Anti-inflammatory Principle from Sida cordifolia Linn. J. Biological Sci. 6: 160–163; 2006.
Wei X; Gou X; Yuan T; Russell SD. A highly efficient in vitro plant regeneration system and Agrobacterium-mediated transformation in Plumbago zeylanica. Plant Cell Rep. 25: 513–521; 2006.
Yusuf M; Kabir M. Medicinal plants of Bangladesh. Dhaka, Bangladesh, Council of Scientific and Industrial Research, CSIR. 226; 1999.
Zapata C; Srivatanakul M; Park SH; Lee MO; Salas GS; Smith RH. Improvements in shoot apex regeneration of two fiber crops: Cotton and kenaf. Plant Cell Tiss. Organ Cult. 56: 185–191; 1999.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Dudits Denes
Rights and permissions
About this article
Cite this article
Sivanesan, I., Jeong, B.R. Direct shoot regeneration from nodal explants of Sida cordifolia Linn. In Vitro Cell.Dev.Biol.-Plant 43, 436–441 (2007). https://doi.org/10.1007/s11627-007-9090-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-007-9090-1