Abstract
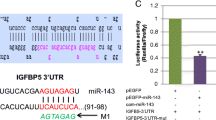
MicroRNAs (miRNAs) have been found to play essential roles in muscle cell proliferation and differentiation. MicroRNA-1 (miR-1) and microRNA-206 (miR-206), which are similar and have the same seed sequence, have specific roles in modulating skeletal muscle proliferation and differentiation in vitro and in vivo. However, there is no information about their function during bovine skeletal muscle satellite cell development. In this study, the profiles of miR-1 and miR-206 and their biological functions in bovine skeletal muscle cell development was investigated. The target genes were predicted, and we used a dual-luciferase reporter assay to demonstrate that miR-1 and miR-206 directly targeted the 3′ untranslated region (3′UTR) of paired-box transcription factor Pax7 and histone deacetylase 4 (HDAC4). We showed that miR-1 and miR-206 facilitate bovine skeletal muscle satellite cell myogenic differentiation by restricting the expression of their target gene and that inhibition of miR-1 and miR-206 increased the Pax7 and HDAC4 protein levels and substantially enhanced satellite cell proliferation. Therefore, our results revealed the mechanism in which miR-1 and miR-206 positively regulate bovine skeletal muscle satellite cell myogenic differentiation via Pax7 and HDAC4 downregulation.




Similar content being viewed by others
References
Bartel DP (2004) MicrsoRNAs: genomics, biogenesis, mechanism, and function. Cell 116:281–297
Buckingham M (2007) Skeletal muscle progenitor cells and the role of Pax genes. C R Biol 330:530–533
Callis TE, Wang DZ (2008) Taking microRNAs to heart. Trends Mol Med 14:254–260
Chen JF, Mandel EM, Thomson JM, Wu Q, Callis TE, Hammond SM, Conlon FL, Wang DZ (2006) The role of microRNA-1 and microRNA-133 in skeletal muscle proliferation and differentiation. Nat Genet 38:228–233
Chen JF, Tao YZ, Li J, Deng ZL, Yan Z, Xiao X, Wang DZ (2010) microRNA-1 and microRNA-206 regulate skeletal muscle satellite cell proliferation and differentiation by repressing Pax7. J Cell Biol 190:867–879
Clop A, Marcq F, Takeda H, Pirottin D, Tordoir X, Bibé B, Bouix J, Caiment F, Elsen JM, Eychenne F, Larzul C, Laville E, Meish F, Milenkovic D, Tobin J, Charlier C, Georges M (2006) A mutation creating a potential illegitimate microRNA target site in the myostatin gene affects muscularity in sheep. Nat Genet 38:813–818
Dey BK, Gagan J, Dutta A (2011) MiR-206 and -486 induce myoblast differentiation by downregulating Pax7. Mol Cell Biol 31:203–214
Griffiths-Jones S (2004) The microRNA registry. Nucleic Acids Res 32:D109–111
Kim HK, Lee YS, Sivaprasad U, Malhotra A, Dutta A (2006) Muscle-specific microRNA miR-206 promotes muscle differentiation. J Cell Biol 174:677–687
Koutsoulidou A, Mastroyiannopoulos NP, Furling D, Uney JB, Phylactou LA (2011) Expression of miR-1, miR-133a, miR-133b and miR-206 increases during development of human skeletal muscle. BMC Dev Biol 11:34
Kuang S, Chargé SB, Seale P, Huh M, Rudnicki MA (2006) Distinct roles for Pax7 and Pax3 in adult regenerative myogenesis. J Cell Biol 172:103–113
Kuang S, Gillespie MA, Rudnicki MA (2008) Niche regulation of muscle satellite cell self-renewal and differentiation. Cell Stem Cell 2:22–31
Lagos-Quintana M, Rauhut R, Yalcin A, Meyer J, Lendeckel W, Tuschl T (2002) Identification of tissue-specific microRNAs from mouse. Curr Biol 12:735–739
Lee RC, Ambros V (2001) An extensive class of small RNAs in Caenorhabditis elegans. Science 294:862–864
Li T, Wu R, Zhang Y, Zhu D (2011) A systematic analysis of the skeletal muscle miRNA transcriptome of chicken varieties with divergent skeletal muscle growth identifies novel miRNAs and differentially expressed miRNAs. BMC Genomics 12:186
Liu N, Williams AH, Kim Y, McAnally J, Bezprozvannaya S, Sutherland LB, Richardson JA, Bassel-Duby R, Olson EN (2007) An intragenic MEF2-dependent enhancer directs muscle-specific expression of microRNAs 1 and 133. Proc Natl Acad Sci U S A 104:20844–20849
Lu J, McKinsey TA, Zhang CL, Olson EN (2000) Regulation of skeletal myogenesis by association of the MEF2 transcription factor with class II histone deacetylases. Mol Cell 6:233–244
McDaneld TG, Smith TP, Doumit ME, Miles JR, Coutinho LL, Sonstegard TS, Matukumalli LK, Nonneman DJ, Wiedmann RT (2009) MicroRNA transcriptome profiles during swine skeletal muscle development. BMC Genomics 10:77
McKinsey TA, Zhang CL, Lu J, Olson EN (2000) Signal-dependent nuclear export of a histone deacetylase regulates muscle differentiation. Nature 408:106–111
Meister G, Tuschl T (2004) Mechanisms of gene silencing by double-stranded RNA. Nature 431:343–349
Olguin HC, Olwin BB (2004) Pax-7 up-regulation inhibits myogenesis and cell cycle progression in satellite cells: a potential mechanism for self-renewal. Dev Biol 275:375–388
Oustanina S, Hause G, Braun T (2004) Pax7 directs postnatal renewal and propagation of myogenic satellite cells but not their specification. EMBO J 23:3430–3439
Rao PK, Kumar RM, Farkhondeh M, Baskerville S, Lodish HF (2006) tors that regulate expression of muscle-specific microRNAs. Proc Natl Acad Sci U S A 103:8721–8726
Relaix F, Montarras D, Zaffran S, Gayraud-Morel B, Rocancourt D, Tajbakhsh S, Mansouri A, Cumano A, Buckingham M (2006) Pax3 and Pax7 have distinct and overlapping functions in adult muscle progenitor cells. J Cell Biol 172:91–102
Relaix F, Rocancourt D, Mansouri A, Buckingham M (2005) A Pax3/Pax7-dependent population of skeletal muscle progenitor cells. Nature 435:948–953
Rosenberg MI, Georges SA, Asawachaicharn A, Analau E, Tapscott SJ (2006) MyoD inhibits Fstl1 and Utrn expression by inducing transcription of miR-206. J Cell Biol 175:77–85
Sabourin LA, Rudnicki MA (2000) The molecular regulation of myogenesis. Clin Genet 57:16–25
Sempere LF, Freemantle S, Pitha-Rowe I, Moss E, Dmitrovsky E, Ambros V (2004) Expression profiling of mammalian microRNAs uncovers a subset of brain-expressed microRNAs with possible roles in murine and human neuronal differentiation. Genome Biol 5:R13
Sun J, Li M, Li Z, Xue J, Lan X, Zhang C, Lei C, Chen H (2013) Identification and profiling of conserved and novel microRNAs from Chinese Qinchuan bovine longissimus thoracis. BMC Genomics 14:42
van Rooij E, Liu N, Olson EN (2008) MicroRNAs flex their muscles. Trends Genet 24:159–166
Wang YM, Dai Y, Liu XF, Liu ZW, Li JX, Guo H, Ding XB (2014) Culture, identification and induced differentiation of bovine skeletal muscle satellite cells. China Anim Husb Vet Med 41:142–147 (in Chinese)
Wang YM, Ding XB, Dai Y, Liu XF, Guo H, Zhang Y (2015) Identification and bioinformatics analysis of miRNAs involved in bovine skeletal muscle satellite cell myogenic differentiation. Mol Cell Biochem 404:113–122
Zammit PS, Relaix F, Nagata Y, Ruiz AP, Collins CA, Partridge TA, Beauchamp JR (2006) Pax7 and myogenic progression in skeletal muscle satellite cells. J Cell Sci 119:1824–1832
Zhao Y, Samal E, Srivastava D (2005) Serum response factor regulates a muscle-specific microRNA that targets Hand2 during cardiogenesis. Nature 436:214–220
Acknowledgments
This work was supported by the National Natural Science Foundation of China (31201021), Natural Science Foundation of Tianjin (13JCQNJC14600), the Excellent Young Teachers Program of Tianjin and the Veterinary Biotechnology Scientific Research Innovation Team of Tianjin (TD12-5019).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Editor: Tetsuji Okamoto
Yang Dai and Yi Min Wang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Dai, Y., Wang, Y.M., Zhang, W.R. et al. The role of microRNA-1 and microRNA-206 in the proliferation and differentiation of bovine skeletal muscle satellite cells. In Vitro Cell.Dev.Biol.-Animal 52, 27–34 (2016). https://doi.org/10.1007/s11626-015-9953-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-015-9953-4