Summary
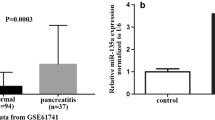
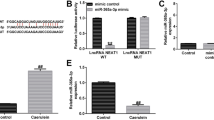
The expression of microRNA-19b (miR-19b) in acute necrotizing pancreatitis (ANP) and its functional role in acinar cell necrosis of SD rats were investigated. Twelve SD rats were divided into two groups randomly, including control group and ANP group. The rat ANP models were established by intraperitoneal injection of L-arginine (2400 mg/kg body weight), and equal volume of 0.9% NaCl was injected in the control group. MiRNA chip assay was performed to examine the expression of miRNAs in the pancreas in two different groups. Besides, to further explore the role of miR-19b in ANP in vitro, taurolithocholic acid 3-sulfate disodium salt (TLC-S) (200 μmol/L) was administrated to treat the rat pancreatic acinar cell line, AR42J, for establishing the ANP cells model. The quantitative real-time PCR (qRT-PCR) was adopted to measure the miR-19b expression. Moreover, the mimic miRNA, miRNA antisense oligonucleotide (AMO) and control vector were used to transfect AR42J cells, the expression of miR-19b was confirmed by qRT-PCR and the necrotizing rate of AR42J cells was detected with AO/EB method. The expression of miR-19b was significantly higher in ANP group than in control group as displayed by the miRNA chip assay. Furthermore, after inducing necrosis of AR42J cells in vitro, the expression of miR-19b was significantly increased by 2.51±0.14 times in comparison with the control group. As revealed by qRT-PCR assay, the expression of miR-19b was 5.94±0.95 times higher in the mimic miRNA group than in the control vector group, companied with an obviously increased acinar cell necrotizing rate (50.3%±1.5% vs. 39.6%±2.3%, P<0.05). Moreover, the expression of miR-19b in the miRNA AMO group was 0.38±0.15 times lower than in the control vector group, and the cell necrosis rate was much lower accordingly (23.1%±3.3% vs. 39.6%±2.3%, P<0.05). Besides, there was no significant difference between the control vector cells and the cells without treatment (P>0.05). The expression of miR-19b was significantly induced in ANP. In addition, up-regulation of miR-19b could promote the necrosis of pancreatic acinar cells and miR-19b deficiency could decrease the rate of pancreatic acinar cell necrosis.
Similar content being viewed by others
References
Booth DM, Mukherjee R, Sutton R, et al. Calcium and reactive oxygen species in acute pancreatitis: Friend or Foe? Antioxid Redox Signal, 2011,15(10):2683–2698
Banks PA, Banks TL, Dervenis C, et al. Classification of acute pancreatitis—2012: revision of the Atlanta classification and definitions by international consensus. Gut, 2013,62(1):102–111
Xu M, Wang KN, Wu K, et al. Pyrrolidine dithiocarbamate inhibits nuclear factor ?B and Toll-like receptor 4 expression in rats with acute necrotizing pancreatitis. Gut Liver, 2015,9(3):411–416
Lakner AM, Steuerwald NM, Walling TL, et al. Inhibitory effects of miRNA-19b in hepatic stellate cell-mediated fibrogenesis. Hepatology, 2012,56(1):300–310
Popov A, Szabo A, Mandys V. Small nucleolar RNA U91 is a new internal control for accurate microRNAs quantification in pancreatic cancer. BMC Cancer, 2015,15(1): 774
Paik WH, Song BJ, Kim HW, et al. MicroRNA-200c as a prognostic biomarker for pancreatic cancer. Korean J Gastroenterol, 2015,66(4):215–220
Masamune A, Nakano E, Hamada S, et al. Alteration of the microRNA expression profile during the activation of pancreatic stellate cells. Scand J Gastroenterol, 2014, 49(3):323–331
Qin T, Fu Q, Pan YF, et al. Expressions of miR-22 and miR-135a in acute pancreatitis. J Huazhong Univ Sci Technolog Med Sci, 2014,34(2):225–233
Kaiser AM, Saluja AK, Sengupta A, et al. Relationship between severity, necrosis,and apoptosis in five mod els of experimental acute pancreatitis. Am J Physiol, 1995,269(5 Pt 1):C1295–304
Jacob TG, Raghav R, Kumar A. Duration of injury correlates with necrosis in caerulein-induced experimental acute pancreatitis: implications for pathophysiology. Int J Exp Path, 2014,95:199–208
Hu GY, Shen JQ, Cheng L. Reg4 protects against acinar cell necrosis in experimental pancreatitis. Gut, 2011,60(6): 820–828
Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell, 2004,116(2): 281–297
Carthew RW. Gene regulation by microRNAs. Curr Opin Genet Dev, 2006,16(2):203–208
Tian R, Wang RL, Xie H, et al. Overexpressed miRNA-155 dysregulates intestinal epithelial apical junctional complex in severe acute pancreatitis. World J Gastroenterol, 2013,19(45):8282–8291
Goodwin D, Rosenzweig B, Zhang J, et al. Evaluation of miR-216a and miR-217 as potential biomarkers of acute pancreatic injury in rats and mice. Biomarkers, 2014,19(6):517–529
Fan Y, Yin SY, Hao Y, et al. miR-19b promotes tumor growth and metastasis via targeting TP53. RNA, 2014, 20(6):765–772
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hu, Mx., Zhang, Hw., Fu, Q. et al. Functional role of MicroRNA-19b in acinar cell necrosis in acute necrotizing pancreatitis. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 36, 221–225 (2016). https://doi.org/10.1007/s11596-016-1570-2
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-016-1570-2