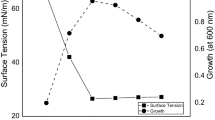
Abstract
Background
Benzo[ghi]perylene (BghiP), a polycyclic aromatic hydrocarbon (PAH) containing six fused benzene rings is considered as priority pollutant because of its carcinogenicity, mutagenicity and acute toxicity.
Methods
The synthesis of ZnO nanoparticles was done following the standard method. Biosurfactant production by yeast consortium YC04 in MSM was confirmed by various tests viz. drop collapse test, methylene blue agar plate method and emulsification test (E24) using the standard procedures. Efficiency of YC04 was tested to remediate BghiP in presence of ZnO nanoparticles and produced biosurfactant in the growth medium.
Results
Response surface methodology (RSM), 3-level five variables Box-Behnken design (BBD) was employed to optimize the factors viz. pH 7.0, temperature 30°C, shaking speed 130 rpm, inoculum dosage 3% and ZnO nanoparticles concentration 2 g/L after a period of 6 days of incubation for the enhanced degradation of BghiP (63.83±0.01%). It was well in close agreement with the predicated value obtained by RSM model yield (63.83±0.08%). Analysis of variance (ANOVA) showed F-value of 51.70, R2 of 0.9764, probability of<0.0001 and coefficient of variation of 1.25% confirmed the validity of the model. Degradation of BghiP was assessed using GC-MS and FTIR analysis. Kinetic study demonstrated that BghiP degradation fitted first order kinetic model.
Conclusions
To the best of our knowledge, this is the first report on process optimization toward nanobioremediation of BghiP using yeast consortium in presence of ZnO nanoparticles and produced biosurfactant in medium.
Similar content being viewed by others
References
Agarry S E, Aremu M O, Aworanti O A (2013). Kinetic modelling and half-life study on enhanced soil bioremediation of bonny light crude oil amended with crop and animal-derived organic wastes. J Pet Environ Biotechnol, 4(02): 137
Arulazhagan P, Vasudevan N, Yeom I T (2010). Biodegradation of polycyclic aromatic hydrocarbon by a halotolerant bacterial consortium isolated from marine environment. Int J Environ Sci Technol, 7(4): 639–652
Bahia FM, de Almeida G C, de Andrade L P, Campos C G, Queiroz L R, da Silva R L V, Abdelnur P V, Corrêa J R, Bettiga M, Parachin N S (2018). Rhamnolipids production from sucrose by engineered Saccharomyces cerevisiae. Sci Rep, 8(1): 2905
Bodour A A, Guerrero-Barajas C, Jiorle B V, Malcomson M E, Paull A K, Somogyi A, Trinh L N, Bates R B, Maier R M (2004). Structure and characterization of flavolipids, a novel class of biosurfactants produced by Flavobacterium sp. strain MTN11. Appl Environ Microbiol, 70(1): 114–120
Bodour A A, Miller-Maier R M (1998). Application of a modified dropcollapse technique for surfactant quantitation and screening of biosurfactant-producing microorganisms. J Microbiol Methods, 32 (3): 273–280
Capellos C, Bielski B H (1972). Kinetic systems: mathematical description of chemical kinetics in solution. New York, USA: Wiley-Inter science.
Cerniglia C E (1992). Biodegradation of polycyclic aromatic hydrocarbons. Biodegradation, 3(2-3): 351–368
Cuny P, Faucet J, Acquaviva M, Bertrand J C, Gilewicz M (1999). Enhanced biodegradation of phenanthrene by a marine bacterium in presence of a synthetic surfactant. Lett Appl Microbiol, 29(4): 242–245
EFSA (2008). Polycyclic aromatic hydrocarbons in food scientific opinion of the panel on contaminants in the food chain. EFSA J, 724: 1–114
El-Sheshtawy H S, Ahmed W (2017). Bioremediation of crude oil by Bacillus licheniformis in the presence of different concentration nanoparticles and produced biosurfactant. Int J Environ Sci Technol, 14(8): 1603–1614
García-Delgado C, Alfaro-Barta I, Eymar E (2015). Combination of biochar amendment and mycoremediation for polycyclic aromatic hydrocarbons immobilization and biodegradation in creosote-contaminated soil. J Hazard Mater, 285: 259–266
Habs M, Jahn S A A, Schmähl D (1984). Carcinogenic activity of condensate from coloquint seeds (Citrullus colocynthis) after chronic epicutaneous administration to mice. J Cancer Res Clin Oncol, 108 (1): 154–156
Haghighat S, Akhavan A, Assadi M M, Pasdar S H (2008). Ability of indigenous Bacillus licheniformis and Bacillus subtilis in microbial EOR. Int J Environ Sci Technol, 5: 385–390
Hesham A E L, Wang Z, Zhang Y, Zhang J, Lv W, Yang M (2006). Isolation and identification of a yeast strain capable of degrading four and five ring aromatic hydrocarbons. Ann Microbiol, 56(2): 109–112
Ibrahim H M M (2018). Characterization of biosurfactants produced by novel strains of Ochrobactrum anthropi HM-1 and Citrobacter freundii HM-2 from used engine oil-contaminated soil. Egypt J Petrol, 27(1): 21–29
Jianlong W, Xiangchun Q, Liping H, Yi Q, Hegemann W (2002). Microbial degradation of quinoline by immobilized cells of Burkholderia pickettii. Water Res, 36(9): 2288–2296
Jin X, Tian W, Liu Q, Qiao K, Zhao J, Gong X (2017). Biodegradation of the benzo[a]pyrene-contaminated sediment of the Jiaozhou Bay wetland using Pseudomonas sp. immobilization. Mar Pollut Bull, 117(1-2): 283–290
Kumar H, Rani R (2013). Structural and optical characterization of ZnO nanoparticles synthesized by microemulsion route. Int Lett Chem Phys Astron, 19: 26–36
Litt G, Almquist G (2009). An investigation of CuO/Fe2O3 catalysts for the gas-phase oxidation of ethanol. Appl Catal B, 90(1-2): 10–17
Mandal S K, Das N (2018). Biodegradation of perylene and benzo[ghi] perylene (5–6 rings) using yeast consortium: kinetic study, enzyme analysis and degradation pathway. J Environ Biol, 39(1): 5–15
Mishra S, Singh S N, Pande V (2014). Bacteria induced degradation of fluoranthene in minimal salt medium mediated by catabolic enzymes in vitro condition. Bioresour Technol, 164: 299–308
Mueller-Spitz S R, Crawford K D (2014). Silver nanoparticle inhibition of polycyclic aromatic hydrocarbons degradation by Mycobacterium species RJGII-135. Lett Appl Microbiol, 58(4): 330–337
Ooi B G, Mulisa A, Kim H Y, Chong N S (2003). Methods development for the detection of trace metabolites from the biodegradation of polycyclic aromatic hydrocarbons by yeasts. J Tenn Acad Sci, 78: 65–75
Patowary K, Kalita M C, Deka S (2015). Degradation of polyaromatic hydrocarbons (PAHs) employing biosurfactant producing Pseudomonas aeruginosa KS3. Indian J Biotechnol, 14: 208–215
Ren H, Zanma S, Urano N, Endo H, Mineki S, Hayashi T (2004). Pyrene decomposing yeasts collected from sea water of Tokyo Bay. Nippon Suisan Gakkaishi, 70(5): 687–692
Safaei-Ghomi J, Ghasemzadeh M A (2017). Zinc oxide nanoparticle promoted highly efficient one pot three-component synthesis of 2,3-disubstituted benzofurans. Arab J Chem, 10: S1774–S1780
Satpute S K, Bhawsar B D, Dhakephalkar P K, Chopade B A (2008). Assessment of different screening methods for selecting biosurfactant producing marine bacteria. Indian J Mar Sci, 37: 243–250
Seo J S, Keum Y S, Harada R M, Li Q X (2007). Isolation and characterization of bacteria capable of degrading polycyclic aromatic hydrocarbons (PAHs) and organophosphorus pesticides from PAHcontaminated soil in Hilo, Hawaii. J Agric Food Chem, 55(14): 5383–5389
Shin K H, Cha D K (2008). Microbial reduction of nitrate in the presence of nanoscale zero-valent iron. Chemosphere, 72(2): 257–262
Su X M, Bamba A M, Zhang S, Zhang Y G, Hashmi M Z, Lin H J, Ding L X (2018). Revealing potential functions of VBNC bacteria in polycyclic aromatic hydrocarbons biodegradation. Lett Appl Microbiol, 66(4): 277–283
Tuleva B K, Ivanov G R, Christova N E (2002). Biosurfactant production by a new Pseduomonas putida strain. Z. Naturforsch, 57c(3-4): 356–360
Wei H, Le Z, Xiaojun L, Zongqiang G, Yongwei Y, Zhi L (2015). Influence of Mucor mucedo immobilized to corncob in remediation of pyrene contaminated agricultural soil. Environ Eng Res, 20(2): 149–154
Wilson S C, Jones K C (1993). Bioremediation of soil contaminated with polynuclear aromatic hydrocarbons (PAHs): a review. Environ Pollut, 81(3): 229–249
Winquist E, Björklöf K, Schultz E, Räsänen M, Salonen K, Anasonye F, Cajthaml T, Steffen K T, Jørgensen K S, Tuomela M (2014). Bioremediation of PAH-contaminated soil with fungi-from laboratory to field scale. Int Biodeter Biodegr, 86: 238–247
Zaragoza-Ojeda M, Eguía-Aguilar P, Perezpeña-Díazconti M, Arenas-Huertero F (2016). Benzo[ghi]perylene activates the AHR pathway to exert biological effects on the NL-20 human bronchial cell line. Toxicol Lett, 256: 64–76
Zhang X, Zhang N, Fu H, Chen T, Liu S, Zheng S, Zhang J (2017). Effect of zinc oxide nanoparticles on nitrogen removal, microbial activity and microbial community of CANON process in a membrane bioreactor. Bioresour Technol, 243: 93–99
Acknowledgment
The authors are grateful to VIT, Vellore for providing necessary laboratory facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mandal, S.K., Ojha, N. & Das, N. Process optimization of benzo[ghi]perylene biodegradation by yeast consortium in presence of ZnO nanoparticles and produced biosurfactant using Box-Behnken design. Front. Biol. 13, 418–424 (2018). https://doi.org/10.1007/s11515-018-1523-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11515-018-1523-1