Abstract
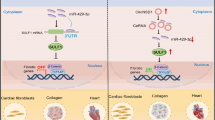
Cardiac fibrosis is one of the crucial pathological factors in the heart, and various cardiac conditions associated with excessive fibrosis can eventually lead to heart failure. However, the exact molecular mechanism of cardiac fibrosis remains unclear. In the present study, we show that a novel lncRNA that we named cardiac fibrosis-associated regulator (CFAR) is a profibrotic factor in the heart. CFAR was upregulated in cardiac fibrosis and its knockdown attenuated the expression of fibrotic marker genes and the proliferation of cardiac fibroblasts, thereby ameliorating cardiac fibrosis. Moreover, CFAR acted as a ceRNA sponge for miR-449a-5p and derepressed the expression of LOXL3, which we experimentally established as a target gene of miR-449a-5p. In contrast to CFAR, miR-449a-5p was found to be significantly downregulated in cardiac fibrosis, and artificial knockdown of miR-449a-5p exacerbated fibrogenesis, whereas overexpression of miR-449a-5p impeded fibrogenesis. Furthermore, we found that LOXL3 mimicked the fibrotic factor TGF-β1 to promote cardiac fibrosis by activating mTOR. Collectively, our study established CFAR as a new profibrotic factor acting through a novel miR-449a-5p/LOXL3/mTOR axis in the heart and therefore might be considered as a potential molecular target for the treatment of cardiac fibrosis and associated heart diseases.
Similar content being viewed by others
References
Abbas, N., Perbellini, F., and Thum, T. (2020). Non-coding RNAs: emerging players in cardiomyocyte proliferation and cardiac regeneration. Basic Res Cardiol 115, 52.
Cai, B., Ma, W., Ding, F., Zhang, L., Huang, Q., Wang, X., Hua, B., Xu, J., Li, J., Bi, C., et al. (2018). The long noncoding RNA CAREL controls cardiac regeneration. J Am College Cardiol 72, 534–550.
Che, H., Wang, Y., Li, H., Li, Y., Sahil, A., Lv, J., Liu, Y., Yang, Z., Dong, R., Xue, H., et al. (2020). Melatonin alleviates cardiac fibrosis via inhibiting lncRNA MALAT1/miR-141-mediated NLRP3 inflammasome and TGF-β1/Smads signaling in diabetic cardiomyopathy. FASEB J 34, 5282–5298.
Chen, W., Yang, A., Jia, J., Popov, Y.V., Schuppan, D., and You, H. (2020). Lysyl oxidase (LOX) family members: rationale and their potential as therapeutic targets for liver fibrosis. Hepatology 72, 729–741.
Findlay, A.D., Foot, J.S., Buson, A., Deodhar, M., Jarnicki, A.G., Hansbro, P.M., Liu, G., Schilter, H., Turner, C.I., Zhou, W., et al. (2019). Identification and optimization of mechanism-based fluoroallylamine inhibitors of lysyl oxidase-like 2/3. J Med Chem 62, 9874–9889.
Glenfield, C., and McLysaght, A. (2018). Pseudogenes provide evolutionary evidence for the competitive endogenous RNA hypothesis. Mol Biol Evol 35, 2886–2899.
Guan, X., Wang, L., Liu, Z., Guo, X., Jiang, Y., Lu, Y., Peng, Y., Liu, T., Yang, B., Shan, H., et al. (2016). miR-106a promotes cardiac hypertrophy by targeting mitofusin 2. J Mol Cell Cardiol 99, 207–217.
Halushka, P.V., Goodwin, A.J., and Halushka, M.K. (2019). Opportunities for microRNAs in the crowded field of cardiovascular biomarkers. Annu Rev Pathol Mech Dis 14, 211–238.
Hinderer, S., and Schenke-Layland, K. (2019). Cardiac fibrosis—a short review of causes and therapeutic strategies. Adv Drug Deliver Rev 146, 77–82.
Jones, M.G., Andriotis, O.G., Roberts, J.J., Lunn, K., Tear, V.J., Cao, L., Ask, K., Smart, D.E., Bonfanti, A., Johnson, P., et al. (2018). Nanoscale dysregulation of collagen structure-function disrupts mechanohomeostasis and mediates pulmonary fibrosis. eLife 7, e36354.
Jourdan-Le Saux, C., Tomsche, A., Ujfalusi, A., Jia, L., and Csiszar, K. (2001). Central nervous system, uterus, heart, and leukocyte expression of the LOXL3 gene, encoding a novel lysyl oxidase-like protein. Genomics 74, 211–218.
Kraft-Sheleg, O., Zaffryar-Eilot, S., Genin, O., Yaseen, W., Soueid-Baumgarten, S., Kessler, O., Smolkin, T., Akiri, G., Neufeld, G., Cinnamon, Y., et al. (2016). Localized LoxL3-dependent fibronectin oxidation regulates myofiber stretch and integrin-mediated adhesion. Dev Cell 36, 550–561.
Liu, Y., Wang, Y., Fu, X., and Lu, Z. (2018). Long non-coding RNA NEAT1 promoted ovarian cancer cells’ metastasis through regulation of miR-382-3p/ROCK1 axial. Cancer Sci 109, 2188–2198.
Lorenzen, J.M., and Thum, T. (2016). Long noncoding RNAs in kidney and cardiovascular diseases. Nat Rev Nephrol 12, 360–373.
Micheletti, R., Plaisance, I., Abraham, B.J., Sarre, A., Ting, C.C., Alexanian, M., Maric, D., Maison, D., Nemir, M., Young, R.A., et al. (2017). The long noncoding RNA Wisper controls cardiac fibrosis and remodeling. Sci Transl Med 9, eaai9118.
Morgan, J.T., Fink, G.R., and Bartel, D.P. (2019). Excised linear introns regulate growth in yeast. Nature 565, 606–611.
Morine, K.J., Qiao, X., York, S., Natov, P.S., Paruchuri, V., Zhang, Y., Aronovitz, M.J., Karas, R.H., and Kapur, N.K. (2018). Bone morphogenetic protein 9 reduces cardiac fibrosis and improves cardiac function in heart failure. Circulation 138, 513–526.
Nie, X., Fan, J., Li, H., Yin, Z., Zhao, Y., Dai, B., Dong, N., Chen, C., and Wang, D.W. (2018). miR-217 promotes cardiac hypertrophy and dysfunction by targeting PTEN. Mol Ther-Nucleic Acids 12, 254–266.
Perl, A. (2016). Activation of mTOR (mechanistic target of rapamycin) in rheumatic diseases. Nat Rev Rheumatol 12, 169–182.
Piccoli, M.T., Gupta, S.K., Viereck, J., Foinquinos, A., Samolovac, S., Kramer, F.L., Garg, A., Remke, J., Zimmer, K., Batkai, S., et al. (2017). Inhibition of the cardiac fibroblast-enriched lncRNA Meg3 prevents cardiac fibrosis and diastolic dysfunction. Circ Res 121, 575–583.
Ponnusamy, M., Liu, F., Zhang, Y.H., Li, R.B., Zhai, M., Liu, F., Zhou, L. Y., Liu, C.Y., Yan, K.W., Dong, Y.H., et al. (2019). Long noncoding RNA CPR (cardiomyocyte proliferation regulator) regulates cardiomyocyte proliferation and cardiac repair. Circulation 139, 2668–2684.
Ricupero, D.A., Poliks, C.F., Rishikof, D.C., Cuttle, K.A., Kuang, P.P., and Goldstein, R.H. (2001). Phosphatidylinositol 3-kinase-dependent stabilization of α1 (I) collagen mRNA in human lung fibroblasts. Am J Physiol-Cell Physiol 281, C99–C105.
Santamaría, P.G., Floristán, A., Fontanals-Cirera, B., Vázquez-Naharro, A., Santos, V., Morales, S., Yuste, L., Peinado, H., García-Gómez, A., Portillo, F., et al. (2018). Lysyl oxidase-like 3 is required for melanoma cell survival by maintaining genomic stability. Cell Death Differ 25, 935–950.
Sarropoulos, I., Marin, R., Cardoso-Moreira, M., and Kaessmann, H. (2019). Developmental dynamics of lncRNAs across mammalian organs and species. Nature 571, 510–514.
Saw, P.E., Xu, X., Chen, J., and Song, E.W. (2021). Non-coding RNAs: the new central dogma of cancer biology. Sci China Life Sci 64, 22–50.
Schwarzenbach, H., Nishida, N., Calin, G.A., and Pantel, K. (2014). Clinical relevance of circulating cell-free microRNAs in cancer. Nat Rev Clin Oncol 11, 145–156.
Seo, H.H., Lee, S., Lee, C.Y., Lee, J., Shin, S., Song, B.W., Kim, I.K., Choi, J.W., Lim, S., Kim, S.W., et al. (2019). Multipoint targeting of TGF-β/Wnt transactivation circuit with microRNA 384–5p for cardiac fibrosis. Cell Death Differ 26, 1107–1123.
Shegogue, D., and Trojanowska, M. (2004). Mammalian target of rapamycin positively regulates collagen type I production via a phosphatidylinositol 3-kinase-independent pathway. J Biol Chem 279, 23166–23175.
Shih, Y.C., Chen, C.L., Zhang, Y., Mellor, R.L., Kanter, E.M., Fang, Y., Wang, H.C., Hung, C.T., Nong, J.Y., Chen, H.J., et al. (2018). Endoplasmic reticulum protein TXNDC5 augments myocardial fibrosis by facilitating extracellular matrix protein folding and redoxsensitive cardiac fibroblast activation. Circ Res 122, 1052–1068.
Tee, A.R., and Blenis, J. (2005). mTOR, translational control and human disease. Semin Cell Dev Biol 16, 29–37.
Thum, T., and Condorelli, G. (2015). Long noncoding RNAs and microRNAs in cardiovascular pathophysiology. Circ Res 116, 751–762.
Viereck, J., Bührke, A., Foinquinos, A., Chatterjee, S., Kleeberger, J.A., Xiao, K., Janssen-Peters, H., Batkai, S., Ramanujam, D., Kraft, T., et al. (2020). Targeting muscle-enriched long non-coding RNA H19 reverses pathological cardiac hypertrophy. Eur Heart J 41, 3462–3474.
Xue, Y., Chen, R., Qu, L., and Cao, X. (2020). Noncoding RNA: from dark matter to bright star. Sci China Life Sci 63, 463–468.
Yang, M., Woolfenden, H.C., Zhang, Y., Fang, X., Liu, Q., Vigh, M.L., Cheema, J., Yang, X., Norris, M., Yu, S., et al. (2020). Intact RNA structurome reveals mRNA structure-mediated regulation of miRNA cleavage in vivo. Nucleic Acids Res 48, 8767–8781.
Yuan, J., Liu, H., Gao, W., Zhang, L., Ye, Y., Yuan, L., Ding, Z., Wu, J., Kang, L., Zhang, X., et al. (2018). MicroRNA-378 suppresses myocardial fibrosis through a paracrine mechanism at the early stage of cardiac hypertrophy following mechanical stress.. Theranostics 8, 2565–2582.
Zhang, F., Fu, X., Kataoka, M., Liu, N., Wang, Y., Gao, F., Liang, T., Dong, X., Pei, J., Hu, X., et al. (2021). Long noncoding RNA Cfast regulates cardiac fibrosis. Mol Ther-Nucleic Acids 23, 377–392.
Zhang, M., Jiang, Y., Guo, X., Zhang, B., Wu, J., Sun, J., Liang, H., Shan, H., Zhang, Y., Liu, J., et al. (2019). Long non-coding RNA cardiac hypertrophy-associated regulator governs cardiac hypertrophy via regulating miR-20b and the downstream PTEN/AKT pathway. J Cell Mol Med 23, 7685–7698.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82070240, 82073844, 82070236, 82270246), University Nursing Program for Young Scholars with Creative Talents in Heilongjiang Province (UNPYSCT-2020169), and Harbin Medical University Marshal Initiative Funding (HMUMIF-21026).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Compliance and ethics The authors declare that they have no conflict of interest.
Supplementsry Figures
Rights and permissions
About this article
Cite this article
Zhang, M., Zhang, B., Wang, X. et al. LncRNA CFAR promotes cardiac fibrosis via the miR-449a-5p/LOXL3/mTOR axis. Sci. China Life Sci. 66, 783–799 (2023). https://doi.org/10.1007/s11427-021-2132-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-021-2132-9