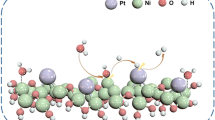
Abstract
We report the in-situ investigation of the production of H2O2 in 5,10,15,20-tetra(4-methoxyphenyl)-21H,23H-porphyrin cobalt(II) (CoTMPP)-catalyzed oxygen reduction reaction (ORR) in neutral electrolytes by electrochemical scanning tunneling microscopy (ECSTM) at the molecular scale. The adsorption of OOH− on active sites can be observed in STM images and is found to be correlated with the pH value of the electrolyte. The thermodynamic parameters of the formation of CoTMPP—OOH− complex are extracted by the quantitative analysis of the STM images. Two stages of the ORR including the formation of H2O2 and further reduction of H2O2 at different reduction potentials can be revealed by electrochemical measurements. In-situ ECSTM experiments unambiguously identify the formation of the CoTMPP—OOH− complex as the high contrast species and its reduction and oxidation process. This work provides the direct evidence for understanding the formation and transformation process of H2O2 at the molecular scale, which benefits the rational design of the high-efficiency electrocatalysts for ORR and H2O2 production.

Similar content being viewed by others
References
Debe MK. Nature, 2012, 486: 43–51
Cullen DA, Neyerlin KC, Ahluwalia RK, Mukundan R, More KL, Borup RL, Weber AZ, Myers DJ, Kusoglu A. Nat Energy, 2021, 6: 462–474
Leng Y, Yang B, Zhao Y, Xiang Z. J Energy Chem, 2022, 73: 549–555
Tang T, Ding L, Jiang Z, Hu JS, Wan LJ. Sci China Chem, 2020, 63: 1517–1542
Santoro C, Bollella P, Erable B, Atanassov P, Pant D. Nat Catal, 2022, 5: 473–484
Zhu Y, Wang X, Shi J, Gan L, Huang B, Tao L, Wang S. Sci China Chem, 2022, 65: 1445–1452
Liu J, Jin Z, Wang X, Ge J, Liu C, Xing W. Sci China Chem, 2019, 62: 669–683
Lu Z, Chen G, Siahrostami S, Chen Z, Liu K, Xie J, Liao L, Wu T, Lin D, Liu Y, Jaramillo TF, Nørskov JK, Cui Y. Nat Catal, 2018, 1: 156–162
Zhang Y, Wu C, Jiang H, Lin Y, Liu H, He Q, Chen S, Duan T, Song L. Adv Mater, 2018, 30: 1707522
Feng YC, Wang X, Wang YQ, Yan HJ, Wang D. J Electrochem, 2022, 28: 2108531
Tang H, Yin H, Wang J, Yang N, Wang D, Tang Z. Angew Chem Int Ed, 2013, 52: 5585–5589
Jurow M, Schuckman AE, Batteas JD, Drain CM. Coord Chem Rev, 2010, 254: 2297–2310
Lindsey JS, Bocian DF. Acc Chem Res, 2011, 44: 638–650
Svane KL, Reda M, Vegge T, Hansen HA. ChemSusChem, 2019, 12: 5133–5141
Li W, Yu A, Higgins DC, Llanos BG, Chen Z. J Am Chem Soc, 2010, 132: 17056–17058
Chen Z, Higgins D, Yu A, Zhang L, Zhang J. Energy Environ Sci, 2011, 4: 3167–3192
Baranton S, Coutanceau C, Roux C, Hahn F, Léger JM. J Electroanal Chem, 2005, 577: 223–234
Liu H, Zhang L, Zhang J, Ghosh D, Jung J, Downing BW, Whittemore E. J Power Sources, 2006, 161: 743–752
Samanta S, Sengupta K, Mittra K, Bandyopadhyay S, Dey A. Chem Commun, 2012, 48: 7631–7633
Perry SC, Pangotra D, Vieira L, Csepei LI, Sieber V, Wang L, Ponce de León C, Walsh FC. Nat Rev Chem, 2019, 3: 442–458
Mahata A, Pathak B. Nanoscale, 2017, 9: 9537–9547
Pang Y, Xie H, Sun Y, Titirici MM, Chai GL. J Mater Chem A, 2020, 8: 24996–25016
Bhattarai A, Marchbanks-Owens K, Mazur U, Hipps KW. J Phys Chem C, 2016, 120: 18140–18150
Bhattarai A, Mazur U, Hipps KW. J Am Chem Soc, 2014, 136: 2142–2148
Yoshimoto S, Inukai J, Tada A, Abe T, Morimoto T, Osuka A, Furuta H, Itaya K. J Phys Chem B, 2004, 108: 1948–1954
Yoshimoto S, Tada A, Suto K, Itaya K. J Phys Chem B, 2003, 107: 5836–5843
Cai ZF, Wang X, Wang D, Wan LJ. ChemElectroChem, 2016, 3: 2048–2051
Facchin A, Zerbetto M, Gennaro A, Vittadini A, Forrer D, Durante C. ChemElectroChem, 2021, 8: 2825–2835
Facchin A, Durante C. Adv Sustain Syst, 2022, 6: 2200111
Facchin A, Kosmala T, Gennaro A, Durante C. ChemElectroChem, 2020, 7: 1431–1437
Wang X, Wang YQ, Feng YC, Wang D, Wan LJ. Chem Soc Rev, 2021, 50: 5832–5849
Wang X, Cai ZF, Wang YQ, Feng YC, Yan HJ, Wang D, Wan LJ. Angew Chem Int Ed, 2020, 59: 16098–16103
Pfisterer JHK, Liang Y, Schneider O, Bandarenka AS. Nature, 2017, 549: 74–77
Gu JY, Cai ZF, Wang D, Wan LJ. ACS Nano, 2016, 10: 8746–8750
Clavilier J. J Electroanal Chem Interfacial Electrochem, 1980, 107: 211–216
Tersoff J, Hamann DR. Phys Rev B, 1985, 31: 805–813
Payne MC, Teter MP, Allan DC, Arias TA, Joannopoulos JD. Rev Mod Phys, 1992, 64: 1045–1097
Perdew JP, Burke K, Ernzerhof M. Phys Rev Lett, 1996, 77: 3865–3868
White JA, Bird DM. Phys Rev B, 1994, 50: 4954–4957
Durand Jr. RR, Anson FC. J Electroanal Chem Interfacial Electrochem, 1982, 134: 273–289
Elbaz L, Korin E, Soifer L, Bettelheim A. J Electroanal Chem, 2008, 621: 91–96
Sonkar PK, Prakash K, Yadav M, Ganesan V, Sankar M, Gupta R, Yadav DK. J Mater Chem A, 2017, 5: 6263–6276
Wang G, Ramesh N, Hsu A, Chu D, Chen R. Mol Simul, 2008, 34: 1051–1056
Wang YQ, Wang X, Feng YC, Yan HJ, Wang D, Wan LJ. J Phys Chem C, 2021, 125: 24915–24919
Acknowledgements
This work was supported by the National Key R&D Program of China (2021YFA1501002), the National Natural Science Foundation of China (21725306 and 21972147), the Key Research Program of the Chinese Academy of Sciences (XDPB01), and the National Postdoctoral Program for Innovative Talents (BX20220307) of the Chinese Postdoctoral Science Foundation. The Supercomputing Environment of the Chinese Academy of Sciences is acknowledged for providing computational resources.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of interest
The authors declare no conflict of interest.
Supporting information
The supporting information is available online at chem.scichina.com and link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Feng, YC., Wang, X., Yi, ZY. et al. In-situ ECSTM investigation of H2O2 production in cobalt—porphyrin-catalyzed oxygen reduction reaction. Sci. China Chem. 66, 273–278 (2023). https://doi.org/10.1007/s11426-022-1465-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-022-1465-8