Abstract
Purpose
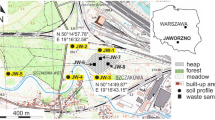
Wastes of unknown composition derived from the production of trivalent chromium (Cr(III)) salts used as tanning agents are deposited in the area of Kanpur, India. The questions of whether these samples are chromite ore processing residue (COPR) and whether Cr occurs in its toxic hexavalent form (Cr(VI)) arise.
Materials and methods
Twenty-one samples from two disposal sites and surrounding soils were analyzed, specifically examining their elemental and mineralogical composition. Additionally, aqueous eluates with different liquid-to-solid ratios were performed and analyzed for Cr(VI).
Results and discussion
The samples were classified in accordance to the sum of silicon and aluminum and the sum of calcium and Cr contents: uncontaminated, moderately contaminated, and highly contaminated material. Highly contaminated material exhibited extremely alkaline pH values up to 12.5 and total Cr contents ranging from 65.7 to 110 g/kg, whereas uncontaminated material had comparatively moderate pH values and Cr contents <1 g/kg. In total, seven crystalline phases commonly found in COPR were identified in the contaminated samples, of which five phases (brownmillerite, hydrocalumite, hydrogarnet, magnesiochromite, and periclase) are known to be able to accommodate Cr whereas hydrogarnet and hydrocalumite are the main host phases for Cr(VI). Batch tests showed that dissolution controlled the Cr(VI) concentrations in the eluates.
Conclusions
Six samples were clearly identified as highly Cr-contaminated COPR. Leaching of Cr(VI) and resulting contamination of soils and water bodies is a key environmental risk arising from these COPR sites especially during the monsoon season. This situation is of particular concern as the local population use the highly Cr(VI)-contaminated water not only for the needs of livestock and irrigation but also as drinking water due to the absence of alternative water resources.






Similar content being viewed by others
References
Alloway BJ (1995) Heavy metals in soils, vol 2. Springer, New York
Ansari AA, Singh IB, Tobschall HJ (2000) Importance of geomorphology and sedimentation processes for metal dispersion in sediments and soils of the Ganga Plain: identification of geochemical domains. Chem Geol 162(3–4):245–266
Avudainayagam S, Megharaj A, Owens G, Kookana RS, Chittleborough D, Naidu R (2003) Chemistry of chromium in soils with emphasis on tannery waste sites. Rev Environ Contam T 178:53–91
Beyersmann D, Hartwig A (2008) Carcinogenic metal compounds: recent insight into molecular and cellular mechanisms. Aech Toxicol 82(8):493–512
Brady NC, Weil RR (2008) The nature and properties of soils, vol 14. Pearson International, Upper Saddle River
Chrysochoou M, Dermatas D (2007) Application of the Rietveld method to assess chromium(VI) speciation in chromite ore processing residue. J Hazard Mater 141(2):370–377
Chrysochoou M, Dermatas D, Grubb DG, Moon DH, Christodoulatos C (2010) Importance of mineralogy in the geoenvironmental characterization and treatment of chromite ore processing residue. J Geotech Geoenviron Eng 136(3):510–521
Darrie G (2001) Commercial extraction technology and process waste disposal in the manufacture of chromium chemicals from ore. Environ Geochem Health 23(3):187–193
Dayan AD, Paine AJ (2001) Mechanisms of chromium toxicity, carcinogenicity and allergenicity: review of the literature from 1985 to 2000. Hum Exp Toxicol 20(9):439–451
Deakin D, West LJ, Stewart DI, Yardley BWD (2001) The leaching characteristics of chromite ore processing residue. Environ Geochem Health 23(3):201–206
Farmer JG, Paterson E, Bewley RJF, Geelhoed JS, Hillier S, Meeussen JCL, Lumsdon DG, Thomas RP, Graham MC (2006) The implications of integrated assessment and modelling studies for the future remediation of chromite ore processing residue disposal sites. Sci Total Environ 360(1–3):90–97
Geelhoed JS, Meeussen JCL, Hillier S, Lumsdon DG, Thomas RP, Farmer JG, Paterson E (2002) Identification and geochemical modeling of processes controlling leaching of Cr(VI) and other major elements from chromite ore processing residue. Geochim Cosmochim Acta 66(22):3927–3942
Geelhoed JS, Meeussen JCL, Roe MJ, Hillier S, Thomas RP, Farmer JG, Paterson E (2003) Chromium remediation or release? Effect of iron(II) sulfate addition on chromium(VI) leaching from columns of chromite ore processing residue. Environ Sci Technol 37(14):3206–3213
Gupta K, Gaumat S, Mishra K (2011) Chromium accumulation in submerged aquatic plants treated with tannery effluent at Kanpur, India. J Environ Biol 32(5):591–597
Hausmann MR (ed) (1990) Engineering principles of ground modification. McGraw-Hill, New York
Hillier S, Roe MJ, Geelhoed JS, Fraser AR, Farmer JG, Paterson E (2003) Role of quantitative mineralogical analysis in the investigation of sites contaminated by chromite ore processing residue. Sci Total Environ 308(1–3):195–210
Hillier S, Lumsdon DG, Brydson R, Paterson E (2007) Hydrogarnet: a host phase for Cr(VI) in chromite ore processing residue (COPR) and other high pH wastes. Environ Sci Technol 41(6):1921–1927
Jaiswal R, Braun B (2010) “Unbemerkte” Verschmutzungsoasen der Weltwirtschaft – das Beispiel der Chromsulfat-Produktion in Nordindien. Geogr Rundsch 62(4):54–55
James BR (1994) Hexavalent chromium solubility and reduction in alkaline soils enriched with chromite ore processing residue. J Environ Qual 23(2):227–233
Kabata-Pendias A, Mukherjee AB (2007) Trace elements from soil to human. Springer, New York
Kumar AR, Riyazuddin P (2010) Chromium speciation in groundwater of a tannery polluted area of Chennai City, India. Environ Monit Assess 160(1–4):579–591
Papp JF (1995) Chromium life cycle study. U.S. Bureau of Mines Information Circular 9411, Reston
Pechova A, Pavlata L (2007) Chromium as an essential nutrient: a review. Veterinarni Medicina 52(1):1–18
Rawat M, Ramanathan A, Subramanian V (2009) Quantification and distribution of heavy metals from small-scale industrial areas of Kanpur City, India. J Hazard Mater 172(2–3):1145–1149
Sass BM, Rai D (1987) Solubility of amorphous chromium(III)-iron(III) hydroxide solid-solutions. Inorg Chem 26(14):2228–2232
Shelnutt SR, Goad P, Belsito DV (2007) Dermatological toxicity of hexavalent chromium. Cr Rev Toxicol 37(5):375–387
Singh RK, Sengupta B, Bali R, Shukla BP, Gurunadharao VVS, Srivatstava R (2009) Identification and mapping of chromium (VI) plume in groundwater for remediation: a case study at Kanpur, Uttar Pradesh. J Geol Soc India 74(1):49–57
Srinivasa Gowd S, Ramakrishna Reddy M, Govil PK (2010) Assessment of heavy metal contamination in soils at Jajmau (Kanpur) and Unnao industrial areas of the Ganga Plain, Uttar Pradesh, India. J Hazard Mater 174(1–3):113–121
Sundar VJ, Rao JR, Muralidharan C (2002) Cleaner chrome tanning—emerging options. J Cleanrt Prod 10(1):69–74
US EPA (1992) SW-846, method 7196A, chromium, hexavalent (colorimetric). US Environmental Protection Agency, Washington
Wazne M, Jagupilla SC, Moon DH, Christodoulatos C, Koutsospyros A (2008) Leaching mechanisms of Cr(VI) from chromite ore processing residue. J Environ Qual 37(6):2125–2134
Whittleston RA, Stewart DI, Mortimer RJG, Tilt ZC, Brown AP, Geraki K, Burke IT (2011) Chromate reduction in Fe(II)-containing soil affected by hyperalkaline leachate from chromite ore processing residue. J Hazard Mater 194:15–23
Acknowledgments
The authors wish to thank the Founder and President of the Kanpur-based NGO “Eco Friends” Mr. R. K. Jaiswal for his constant support both (and especially) on site and from afar. We also appreciate the valuable support from Imran Siddiqui. Finally, special thanks go to Nicole Reps for providing helpful background information about the local situation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Ravi Naidu
Rights and permissions
About this article
Cite this article
Földi, C., Dohrmann, R., Matern, K. et al. Characterization of chromium-containing wastes and soils affected by the production of chromium tanning agents. J Soils Sediments 13, 1170–1179 (2013). https://doi.org/10.1007/s11368-013-0714-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-013-0714-2