Abstract
Purpose
Little is known about the interactive effects of temperature, nitrogen (N) supply, litter quality, and decomposition time on the turnover of carbon (C) and N of forest litter. The objective of this study was to investigate the interactive effects of warming, N addition and tree species on the turnover of C and N during the early decomposition stage of litters in a temperate forest.
Materials and methods
A 12-week laboratory incubation experiment was carried out. The leaf litters including two types of broadleaf litters (Quercus mongolica and Tilia amurensis), a needle litter (Pinus koraiensis), and a mixed litter of them were collected from a broad-leaved Korean pine mixed forest ecosystem in northeastern China in September 2009. Nine treatments were conducted using three temperatures (15, 25, and 35 °C) combined with three doses of N addition (equal to 0, 75, and 150 kg · ha−1 a−1, respectively, as NH4NO3).
Results and discussion
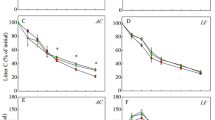
After 12 weeks of incubation, the mass loss ranged between 12 and 35 %. The broadleaf litters had greater mass loss and cumulative CO2–C emission than the needle litter. Temperature and N availability interacted to affect litter mass loss and decomposition rate. The dissolved organic carbon (DOC) and nitrogen (DON) concentrations in litter leachate varied widely with litter types. DOC increased significantly with increased temperature but decreased significantly with increased N availability. DON increased significantly with increased N availability but showed a higher level at the moderate decomposition temperature. The amounts of CO2 and N2O emission were significantly higher at 25 °C than those at 15 and 35 °C, and were significantly increased by the N addition.
Conclusions
The present study indicated relatively intricate temperature and N addition effects on C and N cycling during early stages of litter decomposition, implying that future increases in temperature and N deposition will directly affect C and N cycling in broad-leaved Korean pine mixed forest ecosystem, and may indirectly influence the ecosystem composition, productivity, and functioning in NE China. It is, therefore, important to understand the interactive effects of biotic and abiotic factors on litter decomposition in field conditions in order to assess and predict future ecosystem responses to environmental changes in NE China.





Similar content being viewed by others
References
Aber JD (1992) Nitrogen cycling and nitrogen saturation in temperate forest ecosystems. Trends Ecol Evol 7(7):220–224
Ågren G, Bosatta E, Magill A (2001) Combining theory and experiment to understand effects of inorganic nitrogen on litter decomposition. Oecologia 128(1):94–98
Austin AT, Vitousek PM (2000) Precipitation, decomposition and litter decomposability of Metrosideros polymorpha in native forests on Hawai’i. J Ecol 88(1):129–138
Berg B, Matzner E (1997) Effect of N deposition on decomposition of plant litter and soil organic matter in forest systems. Environ Rev 5(1):1–25
Braker G, Schwarz J, Conrad R (2010) Influence of temperature on the composition and activity of denitrifying soil communities. FEMS Microbiol Ecol 73(1):134–148
Breeuwer A, Heijmans M, Robroek BJM, Limpens J, Berendse F (2008) The effect of increased temperature and nitrogen deposition on decomposition in bogs. Oikos 117(8):1258–1268
Butterbach-Bahl K, Breuer L, Gasche R, Willibald G, Papen H (2002) Exchange of trace gases between soils and the atmosphere in Scots pine forest ecosystems of the northeastern German lowlands: 1. Fluxes of N2O, NO/NO2 and CH4 at forest sites with different N-deposition. For Ecol Manag 167(1–3):123–134
Butterbach-Bahl K, Gasche R, Huber C, Kreutzer K, Papen H (1998) Impact of N-input by wet deposition on N-trace gas fluxes and CH4-oxidation in spruce forest ecosystems of the temperate zone in Europe. Atmos Environ 32(3):559–564
Carreiro MM, Sinsabaugh RL, Repert DA, Parkhurst DF (2000) Microbial enzyme shifts explain litter decay responses to simulated nitrogen deposition. Ecology 81(9):2359–2365
Chapman PJ, Williams BL, Hawkins A (2001) Influence of temperature and vegetation cover on soluble inorganic and organic nitrogen in a spodosol. Soil Biol Biochem 33(7–8):1113–1121
Cleveland CC, Neff JC, Townsend AR, Hood E (2004) Composition, dynamics, and fate of leached dissolved organic matter in terrestrial ecosystems: results from a decomposition experiment. Ecosystems 7(3):175–285
Currie W, Aber J, McDowell W, Boone R, Magill A (1996) Vertical transport of dissolved organic C and N under long-term N amendments in pine and hardwood forests. Biogeochemistry 35(3):471–505
Davidson EA, Chorover J, Dail DB (2003) A mechanism of abiotic immobilization of nitrate in forest ecosystems: the ferrous wheel hypothesis. Global Change Biol 9(2):228–236
Deng XW, Liu Y, Han SJ (2009) Carbon and nitrogen dynamics in early stages of forest litter decomposition as affected by nitrogen addition. J For Res 20(2):111–116
Fang H, Mo JM, Peng SL, Li Z, Wang H (2007) Cumulative effects of nitrogen additions on litter decomposition in three tropical forests in southern China. Plant Soil 297(1):233–242
Fierer N, Craine JM, McLauchlan K, Schimel JP (2005) Litter quality and the temperature sensitivity of decomposition. Ecology 86(2):320–326
Gödde M, Conrad R (1999) Immediate and adaptational temperature effects on nitric oxide production and nitrous oxide release from nitrification and denitrification in two soils. Biol Fert Soils 30(1):33–40
Galloway JN, Cowling EB (2002) Reactive nitrogen and the world: 200 years of change. AMBIO: A Journal of the Human Environment 31(2):64–71
Guo LB, Sims REH (2001) Eucalypt litter decomposition and nutrient release under a short rotation forest regime and effluent irrigation treatments in New Zealand: I. External effects Soil Biol Biochem 33(10):1381–1388
Hammrich A (2008) Effects of warming and nitrogen enrichment on carbon turnover in a littoral wetland. ETH Zürich, citizen of Germany
Hobbie SE (1996) Temperature and plant species control over litter decomposition in Alaskan Tundra. Ecol Monogr 66(4):503–522
Hobbie SE (2000) Interactions between litter lignin and nitrogenitter lignin and soil nitrogen availability during leaf litter decomposition in a Hawaiian montane forest. Ecosystems 3(5):484–494
Hobbie SE, Vitousek PM (2000) Nutrient limitation of decomposition in Hawaiian forests. Ecology 81(7):1867–1877
Iiyama K, Wallis AFA (1990) Determination of lignin in herbaceous plants by an improved acetyl bromide procedure. J Sci Food Agr 51(2):145–161
Keeney DR, Nelson DW (1982) Nitrogen-inorganic forms. In: Page AL, Miller RH, Keeney DR (eds) Methods of soil analysis. Part 2, 2nd edn. Series Agronomy 9. ASA, SSSA, Madison
Knorr M, Frey SD, Curtis PS (2005) Nitrogen additions and litter decomposition: a meta-analysis. Ecology 86(12):3252–3257
Kuiters AT, Mulder W (1993) Water-soluble organic matter in forest soils. Plant Soil 152(2):215–224
Lü C, Tian H (2007) Spatial and temporal patterns of nitrogen deposition in China: synthesis of observational data. J Geophys Res 112(D22): D22S05
Li MH, Kräuchi N, Gao SP (2006) Global warming: can existing reserves really preserve current levels of biological diversity? J Invest Plant Bio 48(3):255–259
Liski J, Nissinen ARI, Erhard M, Taskinen O (2003) Climatic effects on litter decomposition from arctic tundra to tropical rainforest. Global Change Biol 9(4):575–584
Magill AH, Aber JD (1998) Long-term effects of experimental nitrogen additions on foliar litter decay and humus formation in forest ecosystems. Plant Soil 203(2):301–311
Magill AH, Aber JD (2000) Dissolved organic carbon and nitrogen relationships in forest litter as affected by nitrogen deposition. Soil Biol Biochem 32(5):603–613
Manning P, Saunders M, Bardgett RD, Bonkowski M, Bradford MA, Ellis RJ, Kandeler E, Marhan S, Tscherko D (2008) Direct and indirect effects of nitrogen deposition on litter decomposition. Soil Biol Biochem 40(3):688–698
Muhammad W, Vaughan S, Dalal R, Menzies N (2011) Crop residues and fertilizer nitrogen influence residue decomposition and nitrous oxide emission from a Vertisol. Biol Fert Soils 47(1):15–23
Nadelhoffer KJ, Colman BP, Currie WS, Magill A, Aber JD (2004) Decadal-scale fates of 15N tracers added to oak and pine stands under ambient and elevated N inputs at the Harvard Forest (USA). Forest Ecol Manag 196(1):89–107
Osono T (2007) Ecology of ligninolytic fungi associated with leaf litter decomposition. Ecol Res 22(6):955–974
Perakis SS, Hedin LO (2001) Fluxes and fates of nitrogen in soil of an unpolluted old-growth temperate forest, southern Chile. Ecology 82(8):2245–2260
Root TL, Price JT, Hall KR, Schneider SH, Rosenzweig C, Pounds JA (2003) Fingerprints of global warming on wild animals and plants. Nature 421(6918):57–60
Salah YMS, Scholes MC (2011) Effect of temperature and litter quality on decomposition rate of Pinus patula needle litter. Procedia Environmental Sciences 6:180–193
Scheffer RA, Van Logtestijn RSP, Verhoeven JTA (2001) Decomposition of carex and sphagnum litter in two mesotrophic fens differing in dominant plant species. Oikos 92(1):44–54
Schlesinger WH, Lichter J (2001) Limited carbon storage in soil and litter of experimental forest plots under increased atmospheric CO2. Nature 411(6836):466–469
Scowcroft PG, Turner DR, Vitousek PM (2000) Decomposition of Metrosideros polymorpha leaf litter along elevational gradients in Hawaii. Global Change Biol 6(1):73–85
Seely B, Lajtha K (1997) Application of a 15N tracer to simulate and track the fate of atmospherically deposited N in the coastal forests of the Waquoit Bay Watershed, Cape Cod, Massachusetts. Oecologia 112(3):393–402
Sinsabaugh RL, Carreiro MM, Repert DA (2002) Allocation of extracellular enzymatic activity in relation to litter composition, N deposition, and mass loss. Biogeochemistry 60(1):1–24
Song CC, Zhang JB (2009) Effects of soil moisture, temperature, and nitrogen fertilization on soil respiration and nitrous oxide emission during maize growth period in northeast China. Acta Agr Scand B-S P 59(2):97–106
Staaf H, Berg B (1982) Accumulation and release of plant nutrients in decomposing Scots pine needle litter. Long-term decomposition in a Scots pine forest II Can J Bot 60(8):1561–1568
Tang JW, Cao M, Zhang JH, Li MH (2010) Litterfall production, decomposition and nutrient use efficiency varies with tropical forest types in Xishuangbanna, SW China: a 10-year study. Plant Soil 335(1):271–288
Taylor B, Parkinson D (1988a) A new microcosm approach to litter decomposition studies. Can J Bot 66(10):1933–1939
Taylor BR, Parkinson D (1988b) Aspen and pine leaf litter decomposition in laboratory microcosms. II. Interactions of temperature and moisture level. Can J Bot 66(10):1966–1973
Treseder KK (2008) Nitrogen additions and microbial biomass: a meta-analysis of ecosystem studies. Ecol Lett 11(10):1111–1120
Vestgarden LS (2001) Carbon and nitrogen turnover in the early stage of Scots pine (Pinus sylvestris L.) needle litter decomposition: effects of internal and external nitrogen. Soil Biol Biochem 33(4–5):465–474
Waldrop MP, Zak DR, Sinsabaugh RL, Gallo M, Lauber C (2004) Nitrogen deposition modifies soil carbon storage through changes in microbial enzymatic activity. Ecol Appl 14(4):1172–1177
Wetterstedt JÅM, Persson T, ÅGren GI (2010) Temperature sensitivity and substrate quality in soil organic matter decomposition: results of an incubation study with three substrates. Global Change Biol 16(6):1806–1819
Zech W, Guggenberger G, Schulten H-R (1994) Budgets and chemistry of dissolved organic carbon in forest soils: effects of anthropogenic soil acidification. Sci Total Environ 152(1):49–62
Zhang CY, Zhao XH, Zheng JM (2006) A study on soil properties in forest gaps and under canopy in broad-leaved Pinus koriensis forest in Changbai Mountain. Forest Res 19(3):347–352
Zhang M, Guan DX, Han SJ (2005) Climatic dynamics of broadleaved Korean pine forest in Changbai Mountain during the last 22years. Chinese J Ecol 24(9):1007–1012
Acknowledgments
We acknowledge the National Basic Research Program of China (no. 2011CB403200), National Natural Science Foundation of China (no. 40930107), and Knowledge Innovation Program of Chinese Academy of Sciences (no. KSCX-YW-Z-1022) for their financial support. We are highly indebted to Prof. Zhihong Xu of the School of Biomolecular and Physical Sciences for his helpful comments. We are also grateful to Prof. Gongfu Ye of Fujian Province Forest Institute for providing experimental apparatus.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Thomas H. DeLuca
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
DOC 42 kb
Rights and permissions
About this article
Cite this article
Guo, R., Zheng, J., Han, S. et al. Carbon and nitrogen turnover in response to warming and nitrogen addition during early stages of forest litter decomposition—an incubation experiment. J Soils Sediments 13, 312–324 (2013). https://doi.org/10.1007/s11368-012-0630-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-012-0630-x