Abstract
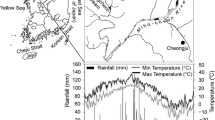
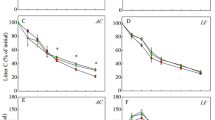
The effects of nitrogen (N) availability and tree species on the dynamics of carbon and nitrogen at early stage of decomposition of forest litter were studied in a 13-week laboratory incubation experiment. Fresh litter samples including needle litter (Pinus koraiensis) and two types of broadleaf litters (Quercus mongolica and Tilia amurensis) were collected from a broadleaf-korean pine mixed forest in the northern slope of Changbai Mountain (China). Different doses of N (equal to 0, 30 and 50 kg·ha−1yr−1, respectively, as NH4NO3) were added to litter during the experiment period. The litter decomposition rate expressed as mass loss and respiration rate increased significantly with increasing N availability. The mass loss and cumulative CO2-C emission were higher in leaf litter compared to that in needle litter. The dissolved organic Carbon (DOC) concentrations in litter leachate varied widely between the species, but were not greatly affected by N treatments. Regardless of the N addition rate, both N treatments and species had no significant effect on dissolved organic N (DON) concentrations in litter leachate. About 52–78% of added N was retained in the litter. The percentage of N retention was positively correlated (R2=0.91, p<0.05) with the litter mass loss. This suggested that a forest floor with easily decomposed litter might have higher potential N sink strength than that with more slowly decomposed litter.
Similar content being viewed by others
References
Adams SN, Cornforth IS. 1973. Some short-term effects of lime and fertilizers on a Sitka spruce plantation. II. Laboratory studies on litter decomposition and nitrogen mineralization. Forestry, 46: 39–47.
Aber JD. 1992. Nitrogen cycling and nitrogen saturation in temperate forest ecosystems. Tree, 7: 220–223.
Berg B, Ekbohm G. 1983. Nitrogen immobilization in decomposing needle litter at variable carbon-nitrogen ratios. Ecology, 64: 63–67.
Berg B, Matzner E. 1997. Effect of N deposition on decomposition of plant litter and soil organic matter in forest systems. Environmental Reviews, 5: 1–25.
Blair JM. 1988. Nitrogen, sulfur and phosphorus dynamics in decomposing deciduous leaf litter in the Southern Appalachians. Soil Biology and Biochemistry, 20: 693–701.
Buchmann N. 2000. Biotic and abiotic factors controlling soil respiration rates in Picea abies stands. Soil Biology and Biochemistry, 32: 1625–1635.
Cleveland CC, Neff JC, Townsend AR, Hood E. 2004. Composition, dynamics, and fate of leached dissolved organic matter in terrestrial ecosystems: Results from a decomposition experiment. Ecosystems, 7: 275–285.
Cornelissen JHC. 1996. An experimental comparison of leaf decomposition rates in a wide variety of temperate plant species and types. Journal of Ecology, 84: 573–582.
Davidson EA, Chorover J, Dail BD. 2003. A mechanism of abiotic immobilization of nitrate in forest ecosystems: the ferrous wheel hypothesis. Global Change Biology, 9: 228–36.
Flanagan PW, Van Cleve K. 1983. Nutrient cycling in relation to decomposition and organic-matter quality in taiga ecosystems. Canadian Journal of Forest Research, 13: 795–817.
Fog K. 1988. The effect of added nitrogen on the rate of decomposition of organic matter. Biological Reviews, 63: 433–462.
Gallardo A, Merino J. 1992. Nitrogen immobilization in leaf litter at two Mediterranean ecosystems of SW Spain. Biogeochemistry, 15: 213–228.
Guggenberger G. 1992. Eigenshaften und Dynamik gelöster organischer Substanzen (DOM) auf unterschiedlich immissionsbelasteten Fichtenstandorten. Bayreuther Bodenkundliche Berichte, 26: 1–164.
Hobbie SE. 2000. Interactions between litter lignin and soil nitrogen availability during leaf litter decomposition in a Hawaiian montane forest. Ecosystems, 3: 484–494.
Hohmann J, Neely RK. 1993. Decomposition of Sparangium eurycarpum under controlled pH and nitrogen regimes. Aquatic Botany, 46: 17–33.
Hughes JW, Fahey TJ, Browne B. 1987. A better seed and litter trap. Canadian Journal of Forest Research, 17: 1623–1624.
Iiyama K, Wallis AFA. 1990. Determination of lignin in herbaceous plants by an improved acetyl bromide procedure. Journal of the Science of Food and Agriculture, 51: 145–161.
Li Xuefeng, Han Shijie, Guo Zhongling, Zhen Xingbo, Song Guozheng, Li Kaoxue. 2006. Decomposition of pine needles and twigs on and under the litter layer in the natural Korean pine broadleaved forests. Journal of Beijing Forestry University, 28(3): 8–13.
Limpens L, Berendse L. 2003. How litter quality affects mass loss and N loss from decomposing Sphagnum. Oikos, 103: 537–547.
Lin Lisha, Han Shijie, Wang Yuesi, Gu Zhijing. 2004. Soil CO2 flux in several typical forests of Mountain Changbai. Chinese Journal of Ecology, 23(5): 42–45.
Magill AH, Aber JD. 2000. Dissolved organic carbon and nitrogen relationships in forest litter as affected by nitrogen deposition. Soil Biology and Biochemistry, 32: 603–613.
McClaugherty CA, Pastor J, Aber JD, 1985. Forest litter decomposition in relation to soil nitrogen dynamics and litter quality. Ecology, 66(1): 266–275.
Melillo JM, Aber JD, Muratore JF. 1982. Nitrogen and lignin control of hardwood leaf litter decomposition dynamics. Ecology, 63(3): 621–626.
Moore TR, Trofymow JA, Taylor B, Prescott C, Camire C, Duschene L, Fyles J, Kozak L, Kranabetter M, Morrison I, Siltanen M, Smith S, Titus B, Visser S, Wein R, Zoltai S. 1999. Litter decomposition rates in Canadian forests. Global Change Biology, 5: 75–82.
Nadelhoffer KJ, Colman BP, Currie WS, Magill AH, Aber JD. 2004. Decadal scale fates of 15N tracers added to oak and pine stands under ambient and elevated N inputs at the Harvard Forest (USA). Forest Ecology and Management, 196: 89–107
Nadelhoffer KJ, Emmett BA, Gundersen P, Kjonass OJ, Koopmans CJ, Schleppi P, Tietema A, Wright RF. 1999. Nitrogen deposition makes a minor contribution to carbon sequestration in temperate forests. Nature, 398: 145–148.
Neff JC, Chapin FS, Vitousek PM. 2003. Breaks in the cycle: dissolved organic nitrogen in terrestrial ecosystems. Frontiers in Ecology and Environment, 1: 205–211.
Park JH, Kalbitz K, Matzner E. 2002. Resource control on the production of dissolved organic C and nitrogen in a deciduous forest floor. Soil Biology and Biochemistry, 34: 813–822.
Perakis SS, Hedin LO. 2001. Fluxes and fates of nitrogen in soil of an unpolluted old-growth temperate forest, southern Chile. Ecology, 82: 2245–2260.
Pregitzer KS, Zak DR, Burton AJ, Ashby JA, MacDonald NW. 2004. Chronic nitrate additions dramatically increase the export of carbon and nitrogen from northern hardwood ecosystems. Biogeochemistry, 68: 179–197.
Prescott CE, Vesterdal L, Preston CM, Simard SW. 2004. Influence of initial chemistry on decomposition of foliar litter in contrasting forest types in British Columbia. Canadian Journal of Forest Research, 34: 1714–1729.
Qualls RG, Haines BL. 1991. Geochemistry of dissolved organic nutrients in water percolating through a forest ecosystem. Soil Science Society of America Journal, 55: 1112–1123.
SAS Institute Inc. 1997. Procedures guide, Release 6.12. Cary, NC, USA. Seely B, Lajtha K. 1997. Application of a 15N tracer to simulate and track the fate of atmospherically-deposited N in the coastal forests of the Waquoit Bay Watershed, Cape Cod, MA. Oecologia, 112: 393–402.
Staaf H, Berg B. 1982. Accumulation and release of plant nutrients in decomposing Scots pine needle litter: long-term decomposition in a Scots pine forest II. Canadian Journal of Botany, 60: 1561–1568.
Taylor BR, Parkinson D, Parsons WFJ. 1989. Nitrogen and lignin content as predictors of litter decay rates: a microcosm test. Ecology, 70(1): 97–104.
Trasar-Cepeda C, Leirós MC, Gil-Sotres F. 2000. Biochemical properties of acid soils under climate vegetation (Atlantic oak wood) in an area of the European temperate humid zone (Galicia, NW Spain): specific parameters. Soil Biology and Biochemistry, 32: 747–755.
Vestgarden LS. 2001. Carbon and nitrogen turnover in the early stage of Scots pine (Pinus sylvestris L.) needle litter decomposition: effects of internal and external nitrogen. Soil Biology Biochemistry, 33: 465–474.
Vitousek PM, Howarth RW. 1991. Nitrogen limitation on land and in the sea: How can it occur? Biogeochemistry, 13: 87–115.
Zech W, Guggenberger G, Schulten HR. 1994. Budgets and chemistry of dissolved organic carbon in forest soils: effects of anthropogenic soil acidification. Science of the Total Environment, 152: 49–62.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation project: This work was supported by the Knowledge Innovation Project of the Chinese Academy of Sciences (KZCX2-YW-416) and the National Natural Science Foundation (90411020)
Biography: DENG Xiao-wen (1976– ), male, Ph.D, Tianjin Academy of Environmental Sciences, Tianjin 300191, P.R. China.
Rights and permissions
About this article
Cite this article
Deng, Xw., Liu, Y. & Han, Sj. Carbon and nitrogen dynamics in early stages of forest litter decomposition as affected by nitrogen addition. Journal of Forestry Research 20, 111–116 (2009). https://doi.org/10.1007/s11676-009-0020-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-009-0020-4