Abstract
This study demonstrates the combination of wastewater treatment and green microalgae cultivation for the low-cost production of lipids as a feedstock for biodiesel production. Three green microalgal species were used: Chlamydomonas reinhardtii, Monoraphidium braunii, and Scenedesmus obliquus. Nutrient, heavy metals and minerals removal, biomass productivity, carbohydrate, protein, proline, lipid, and fatty acids methyl ester (FAMEs) contents besides biodiesel properties were evaluated. The results showed that all algal species were highly efficient and had the potential to reduce nitrate, ammonia, phosphate, sulfate, heavy metals (Zn2+, Cu2+, Mn2+, and Fe2+), calcium, magnesium, sodium, and potassium after 10 days of algal treatment compared to initial concentrations. The removal efficiency of these parameters ranged from 12 to 100%. The growth rates of M. braunii and S. obliquus cultivated in wastewater were significantly decreased compared to the control (synthetic medium). In contrast, C. reinhardtii showed the highest growth rate when cultivated in sewage water. Wastewater could decrease the soluble carbohydrates and protein content in all tested algae and increase the proline content in M. braunii and S. obliquus. In wastewater culture, M. braunii had the highest lipid productivity of 5.26 mg L−1 day−1. The fatty acid profiles of two studied species (C. reinhardtii and M. braunii) revealed their suitability as a feedstock for biodiesel production due to their high content of saturated fatty acids, representing 80.91% and 68.62% of the total fatty acid content, respectively, when cultivated in wastewater. This study indicated that wastewater could be used to modify biomass productivity, lipid productivity, and the quantity of individual fatty acids in some algae that affect biodiesel quality to achieve international biodiesel standards.
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
Introduction
Bioremediation is the process of treating wastewater with naturally existing bacteria and other elements of the environment. Additionally, it is cheaper than alternative technologies for disposing of hazardous waste (Vidali 2001). The environmentally friendly and economically practical approach of nutrient removal and biomass generation is offered by microalgae in wastewater treatment (Fawzy and Issa 2016).
Many studies showed that using wastewater can produce not only wastewater reuse but also a great amount of biomass transformation and production, particularly in the microalgae biofuels production and other uses with excellent application prospects (Zhou et al. 2018; Hernández-García et al. 2019). Microalgae engross many nutrients since they need a high amount of phosphorus and nitrogen to synthesize 45–60% of microalgae dry-weight proteins, nucleic acids, and phospholipids.
Sewage and wastewater must be treated since heavy metals are toxic to flora and fauna, and their occurrence in the environment is a significant problem (Adefila et al. 2010). The use of microalgae in heavy metal phycoremediation is a growing trend because of their numerous advantages, including their widespread availability, low costs, high metal removal efficiency, and eco-friendly nature (Leong and Chang 2020; Touliabah et al. 2022; Znad et al. 2022). According to Wang and Davidson (2010), algae may absorb organic contaminants entering cells, such as lipids and carbohydrates, resulting in an environmentally friendly pollution reduction.
In wastewater, some types of algae grow slowly. The wastewater’s chemical makeup or excessive heavy metal levels may cause an inhibiting effect (El-Sheekh et al. 2005, 2016b). The primary characteristic of the technology is the capacity of algae to grow in wastewater to efficiently assimilate organic carbons and inorganic nutrients and achieve maximal algal biomass, effective nutrient removal, and lipid accumulation, which can then be turned into biodiesel (Perez-Garcia et al. 2011). Microalgae grown in wastewater can accumulate lipids ranging from low (10% dry weight) to moderate (25–30% DW) and can provide up to 80% of DW (Abou-Shanab et al. 2013).
Algae have a yearly oil yield that is 7 to 31 times more than palm oil due to their ability to accumulate lipids and their high photosynthetic production (Hu et al. 2008; El-Sheekh et al. 2017a). A potential renewable energy source that could replace fossil fuel without compromising the availability of food for people is biodiesel produced from microalgae (Chisti 2007). One alternative fuel is biodiesel (monoalkyl esters), produced by transesterifying triglyceride oil with monohydric alcohols. Based on previous reports, biodiesel made from canola, soybean, palm, sunflower, and algae can be used as a diesel fuel substitute (Mostafa et al. 2012).
The total biodiesel yield depends on the algal strain’s lipid content and growth rate. In producing biodiesel, the economic biomass production of microalgae must be considered; hence, microalgal species with a high lipid content and cell growth are utilized (Lv et al. 2010).
Factors influencing fuel qualities such as viscosity, specific gravity (SG), cetane number (CN), cloud point (CP), iodine value (IV), and greater heating value (HHV) may be related to fatty acid (FA) chain length and the unsaturation degree in fatty acid methyl ester (FAME) (Knothe 2011; Hoekman et al. 2012). As the chain length increases, CN, Vis, and CP increase, and as unsaturation increases, they decrease (Francisco et al. 2010; Tan and Lin 2011; Hoekman et al. 2012).
On the contrary, SG, IV, and HHV decrease with the increase of chain length and increase with the increase of unsaturation (Refaat 2009). Long chain length and low unsaturation are preferred to achieve low-temperature performance and oxidative stability in biodiesel fuel (Refaat 2009; Hoekman et al. 2012).
The current study demonstrates the bioremoval capacity of three green microalgae: Chlamydomonas reinhardtii, Monoraphidium braunii, and Scenedesmus obliquus from sewage wastewater with simultaneous lipid and fatty acid (FA) accumulation to provide low-cost and environmentally friendly biodiesel production technology.
Materials and methods
Algal isolation and culturing
The unicellular green microalgae, Chlamydomonas reinhardtii, Monoraphidium braunii, and Scenedesmus obliquus, were isolated from a water sample collected from South Valley University. Liquid and solid Beijerinck’s nutritive media were used for the cultivation and isolation of C. reinhardtii and M. braunii (Stein 1966), and blue-green 11 medium was used for Scenedesmus obliquus according to Song et al. (2013). The growth of algal species was evaluated and identified using a compound microscope (Leica DM500) (Prescott-Allen and Prescott-Allen 1982).
Algal growth and culture conditions
In separate 500-mL culture flasks with a nutritive media, Chlamydomonas reinhardtii, Monoraphidium braunii, and Scenedesmus obliquus axenic algal samples were cultured. The cultures were exposed to 100 µmol photons m−2 s−1 of cool-white fluorescent light while being incubated at 25 °C ± 1 °C. The dry air was delivered to the algal cultures (Lorenzen 1964) to maintain algae suspension without mechanical stress, provide CO2 needed for photosynthesis, and prevent cells from settling at the bottom of the containers (Persoone et al. 1980). The algae were routinely sub-cultured and preserved in ideal circumstances.
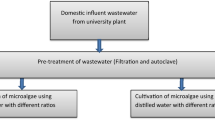
Green microalgae treatment of sewage wastewater sample
Water sampling and experimental design
A sample of sewage water was taken from the sewage station in Qena, Egypt, called Al-Salhya. The algal cells were inoculated in 10-L glass tanks containing a nutritive medium. Sewage water samples were inoculated with algae at a concentration of 5% by adding algal cell cultures. Sewage wastewater was treated with algae for 10 days under illumination and aeriation. The algal mats were precipitated by centrifuging the treated sewage water. The properties of the sewage water were determined using a supernatant. The precipitate algal mats were used to investigate the effect of sewage water on the tested algae.
Chemical analysis of sewage wastewater
A preliminary analysis of sewage water was performed before the inoculation of algae. The total content was filtered to eliminate algae at the final step in each flask and then used to measure various parameters, including nitrate, ammonia, sulfate, and phosphate based on standard methods (American Public Health Association (APHA) 2005). Heavy metals (Fe2+, Cu2+, Zn2+, and Mn2+), calcium, magnesium, sodium, potassium, biological oxygen demand (BOD), and chemical oxygen demand (COD) were evaluated by atomic absorption (spectrometer: MESLO). The following equation was used to determine the percentage of metal ion biosorption.
where Ci is the initial concentration and Cf is the final concentration.
Effect of sewage wastewater on the growth and some metabolites of green microalgae
The effect of sewage water on the growth and metabolites of the tested algae was investigated using precipitate algal mats (algal cells).
Biomass assay
The optical density of the culture for microalgal growth was measured at 560 nm (OD560) using a spectrophotometer and by determining the algal cellular dry weight (CDW). Biomass productivity was determined according to Abomohra et al. (2013).
where CDWE indicates the CDW (g L−1) at days of the early exponential phase (tE) and CDWL at days of the late exponential phase (tL).
Biochemical assessment of cell constituents
Using a spectrophotometer, pigment fractions (Chl. a, Chl. b, and carotenoids) were estimated in accordance with the method of Metzner et al. (1965).
Pigment fractions were measured by µg/mL algal suspension. Soluble saccharides were calorimetrically determined using the anthrone sulfuric acid method, according to Badour (1959). The soluble proteins were calculated using a calibration curve made using serum albumin and quantified using a spectrophotometer in accordance with the procedure outlined by Lowry et al. (1951). According to the Bates et al. (1973) method, proline was measured spectrophotometrically at 520 nm. Absorbance was calculated on a dry weight basis as mg proline/gm DW using a standard curve.
Determination of lipid content
The modified protocol of El-Sheekh et al. (2017b) was used to extract total lipids from algae. In brief, cells were homogenized with chloroform/methanol (2/1). After dispersion, the mixture was stirred for 20 min at room temperature in an orbital shaker.
A 0.2 volume of 0.9% NaCl solution was used to wash the solvent. Centrifugation was used to divide the homogenate into two phases. Under an argon stream, the lipid extracts were dried in pre-weighted glass vials for 30 min at 80 °C, cooled in a desiccator, and then weighed.
Lipid productivity calculation.
Lipid productivity was determined based on Andrade and Costa (2007) and modified by Abomohra et al. (2013).
where L0 and LD represent the total lipid (mg L−1) of cultivation on the first day (T0) and the desired days phase (TD), respectively.
Fatty acids profile
The lipids and fatty acids methyl esters (FAMEs) transmethylation were extracted as described by Radwan (1978) using the GC–MS system (Agilent Technologies) equipped with a gas chromatograph (7890B) and mass spectrometer detector (5977A) at Central Laboratories Network, National Research Centre, Cairo, Egypt. The GC was equipped with a DB-WAX column (30 m × 250 μm internal diameter and 0.25 μm film thickness). By contrasting the spectrum fragmentation pattern with those found in the Wiley and NIST Mass Spectral Library data, the various constituents were distinguished.
Assessment of biodiesel properties
The produced biodiesel quality was estimated from the fatty acid profile by chemical and physical properties calculations, including the average degree of unsaturation (ADU, %), cetane number (CN), iodine value (IV, g I2 100 g−1 oil), kinematic viscosity (KV, mm2 s−1), specific gravity (SG, kg−1), cloud point (CP, °C) higher heating value (HHV, MJ kg−1), long chain saturation factor (LCSF, %wt.), and cold filter plugging point (CFPP, C) from Eqs. (1)–(9) (Hoekman et al. 2012; Ma et al. 2013).
where the number of carbon–carbon double bonds in the corresponding fatty acid is N, the mass fraction of each fatty acid is Mf, and the weight percentage of each corresponding fatty acid are C16:0, C18:0, C20:0, C22:0, and C24:0.
Statistical analysis
The data were statistically examined using the SPSS statistical computer program and tested using a one-way analysis of variance (version 23).
Results and discussion
Reduction of nutrients from wastewater by green microalgae
The obtained data demonstrated that after algal treatment of the sewage wastewater, nutrient concentrations such as nitrate (NO3−), ammonia (NH4+), phosphate (PO43−), and sulfate (SO42−) significantly reduced (Table 1).
The reduction efficiency of nitrate, ammonia, phosphate, and sulfate reached 57%, 69%, 7%, and 22% in the cultures of C. reinhardtii grown in the wastewater. A reduction of 35%, 71%, 62%, and 12% was detected by using S. obliquus. The removal efficiency by M. braunii accounted for 34%, 53%, 73%, and 17%, compared to the control (initial concentrations).
A similar observation was recorded by Kshirsagar (2014) and El-Sheekh et al. (2016a), who showed that the amount of nutrients consumed during algal growth or absorbed into tissues of algae affected the removal efficiency of nutrients in wastewater. Nitrogen is a significant macro element in microalgae, accounting for 1 to 10% of total biomass, and it is also an important factor in lipid content regulation within algal cells (Chisti 2008). The common bioavailable nitrogen compounds assimilated by microalgae are ammonium (NH4+) and nitrate (NO3−). Ammonium is preferred as a source of nitrogen for the microalgae, and when this is available, no other nitrogen source will be used (Abdel-Raouf et al. 2012). The use of phosphorus for algal growth during phycoremediation results in phosphorus removal. Phosphorus, which is used for the formation of phospholipids, adenosine triphosphates, and nucleic acids in algal cells, is absorbed as inorganic orthophosphate, and this active process requires energy (Emparan et al. 2019). Microalgae can absorb excess phosphorus, which are then granulated into polyphosphate (volutin) and stored within the cells. In the absence of available phosphorus, these reserves can be sufficient for a prolonged growth (Woertz et al. 2009).
Removal of heavy metals from sewage wastewater by green microalgae
The data in Table 1 exhibited that heavy metal concentrations were significantly reduced after biotreatment. From the data, M. braunii culture could remove 100% (Fe2+, Cu2+, and Zn2+) and 86% (Mn2+) from wastewater. The reduction accounting for 56% (Zn2+) and 100% (Fe2+, Cu2+, and Mn2+) were observed using S. obliquus as biosorbent. The removal efficiency of Fe2+, Cu2+, Zn2+, and Mn2+ reached 87%, 68%, 60%, and 100%, respectively, in C. reinhardtii cultures as compared to the control (initial concentration).
In this study, green microalgae were highly effective in removing heavy metals from sewage water. The results demonstrate that M. braunii and S. obliquus were more capable of bioremoval of heavy metals than C. reinhardtii. In addition, the types of heavy metals and algae utilized affected the removal effectiveness of heavy metals. The surface of cell adsorption and cytoplasmic attaching to compounds such as phytochelatins, metallothioneins, as well as intracellular ligands may be responsible for the metal ion accumulation (Peña-Castro et al. 2004). The large surface area and high binding affinity of algae contribute to their efficiency in removing heavy metals from wastewater (Sattayawat et al. 2021).
Algae cell walls contain a variety of functional groups, including sulphydryl, carboxyl, amino, phosphoryl, and hydroxyl, which give the cell surface a negative charge. Cationic metal ions make up the majority of water ions; thus, they are adsorbed onto the surface of cells (Romera et al. 2006). The different microalgal species used in this study indicated the ability to remove heavy metals from a sewage water sample. This observation has been emphasized by RATH (2012), who stated that algal cells accumulated higher metal concentrations when grown in media with high metal levels.
Reduction of some minerals from sewage wastewater by green microalgae
Table 1 showed that mineral content decreased markedly in the sewage sample because of algal treatments. The reduction efficiency of calcium, magnesium, sodium, and potassium reached 60%, 41%, 28%, and 43%, respectively, compared to the control in C. reinhardtii cultures grown in the wastewater. In the case of S. obliquus treatment, the removal efficiency of Ca2+, Mg2+, Na+, and K+ from wastewater was 63%, 23%, 62%, and 49%, respectively, compared to the control (initial concentration).
The reduction of Ca2+ (46%), Mg2+ (31%), Na+ (79%), and K+ (80%) was observed by using M. braunii as a biosorbent. This finding is consistent with that obtained by Azab (2002) and El-Sheekh et al. (2016a), who stated that the algal application for wastewater treatment resulted in varying percentages of mineral reduction. In this regard, Rao et al. (2011) found that the utilization of C. vulgaris during phycoremediation significantly reduced magnesium levels and moderately decreased potassium levels. However, they observed a decrease in sodium levels and a significant decrease in calcium.
Removal of biological oxygen demand and chemical oxygen demand
Treated wastewater resulted in a significant reduction in BOD and COD levels. Substances that can be degraded biologically and consume dissolved oxygen in the treatment during the cultivation period were expressed as the BOD indicator. The BOD level was reduced to 90%, 86%, and 81.33% by C. reinhardtii, S. obliquus, and M. braunii, respectively (Table 1). El-Sheekh et al. (2016a) claim that the value of BOD represents the degree of toxicity of wastewater and that utilizing Chlorella species resulted in a 90% reduction in BOD of sewage water. BOD in wastewater represents the respiratory requirements of the algae and bacteria that absorb the organic matter, and an excess of BOD typically reduces the dissolved oxygen (Abdel-Raouf et al. 2012). Zhang et al. (2008), Azarpira et al. (2014), and El-Sheekh et al. (2016a) stated very high BOD reductions using various algal species, proving that microalgae are the best choice for improving physicochemical parameters and wastewater purification. The COD level was also reduced to 88%, 72%, and 69% by C. reinhardtii, S. obliquus, and M. braunii, respectively (Table 1). Similar observations were recorded by Azarpira et al. (2014) and El-Sheekh et al. (2016a). The chemical oxidation of carbon in organic pollutants, which releases carbon dioxide, lowers COD levels. Additionally, faster biodegradation and bioconversion of organic matter because algae may also have a role. In the present study, the BOD and COD levels were higher, reduced to 90% and 88% by C. reinhardtii, showing COD and BOD’s best removal capacity from wastewater.
Influence of wastewater on the growth rate of green microalgae
The effect of wastewater on the growth rate of green microalgae was investigated using the density of algal cells (measured as optical density, OD560) and cellular dry weight (CDW) (biomass productivity) after 10 days of incubation. The optical density of M. braunii and S. obliquus was significantly reduced in wastewater; it was reduced by 31% and 29% when compared to the control (synthetic medium), respectively (Fig. 1). As opposed to that, the optical density of C. reinhardtii considerably increased in wastewater compared to the control; it was increased by 23% of control The productivity of biomass of M. braunii and S. obliquus in the late exponential phase decreased from 90.0 and 27.2 mg L−1 d−1 in synthetic medium to 73.75 and 16.45 mg L−1d−1 in wastewater, respectively (Fig. 1). In contrast, C. reinhardtii demonstrated the highest biomass productivity in wastewater (48.62 mg L−1 day−1) corresponding to (40.2 mg L−1 day−1) in synthetic medium (control).
This finding is consistent with El-Sheekh et al. (2005), who found that microalgae grew more rapidly in sewage wastewater than in Allen’s medium. Available organic matter, phosphorus, and nitrogenous compounds may cause microbial growth. According to Park et al. (2018), the growth of microalgae and biomass are closely related to the photosynthesis occurrence and the subsequent production of energy; thus, they may serve as a biomarker of stress and an important way to express the ecological success of algal strains in adapting to its natural inhabitants. This result also agrees with that of Galgale et al. (2014) connected to the inhibition of wastewater effect to its high salinity levels, which may result in plasmolysis and loss of algal cell activity. Furthermore, a factor that prevented algae growth was a high ammonia concentration in sewage water. The growth of studied algae may be limited due to significant levels of heavy metals in wastewater. Heavy metals were identified as a source of toxicity in wastewater in this context (Tiruneh et al. 2014).
Influence of sewage wastewater on the pigments of green microalgae
Figure 2 revealed that the wastewater was responsible for the increased Chl. a, Chl. b, and carotenoids in C. reinhardtii reach 40%, 14%, and 23%, respectively, compared to the control (standard medium). In the case of S. obliquus and M. braunii, cultures observed that Chl. a, Chl. b, and carotenoid values are decreased markedly when cultivated in wastewater. The greatest inhibition effect of Chl. a and carotenoid contents of S. obliquus were 47% and 41% of the control (synthetic media), respectively (Fig. 2).
The reduction in pigment contents in S. obliquus and M. braunii revealed that these algae used the standard medium as a source of nutrition rather than sewage water because sewage water’s chemical composition differs from synthetic media. The quantity of internal metals and the amount of metals bonded to the surface of the algal cells were connected to the growth inhibition (Ma et al. 2003). This result might be attributed to the effect of wastewater on the photosynthetic system, which degrades the pigment content of cells. This observation was consistent with the findings of El-Sheekh et al. (2016b) and El-Otify et al. (2011), who linked the influence of heavy metals to the chlorophyll damage on the thylakoid membranes.
Influence of sewage wastewater on soluble carbohydrate, soluble protein, and proline contents of green microalgae
Treatments with sewage water exerted a significant decrease in the content of soluble carbohydrates and proteins of C. reinhardtii, S. obliquus, and M. braunii, reaching 69.9%, 4.12%, 45.08%, 24.23%, 35.65, and 51.8% as compared to the control cultures grown in synthetic media (Table 2). The results shown in Table 2 cleared that proline content in M. braunii and S. obliquus grown in sewage water significantly increased by 92% and 153% more than that in synthetic medium. In contrast, sewage water decreased proline content in C. reinhardtii by 21.23% compared to synthetic medium (control). In general, the carbohydrate content of C. reinhardtii cells was more affected by sewage water treatment than other microalgae.
Similar to this, El-Sheekh et al. (2016b) found that wastewater caused green microalgae cultures to contain less carbohydrates than control cultures grown in synthetic media. This decrease is a result of heavy metals’ inhibition of photosynthetic activity and increased nitrogen metabolism brought on by the use of carbohydrates as substrates. Kathryn et al. (2013) reported that the difference in the chemical composition between the well-balanced medium and wastewater caused reductions in carbohydrate and protein contents. According to Tripathi and Gaur (2006), the toxicity of these metals on enzymes responsible for protein synthesis causes the accumulation of protein inhibited by metal concentrations. Soluble protein is one of the indicators of both reversible and irreversible changes in an organism’s metabolism, which is known to react to a number of stressors, including natural and xenobiotics. Proline helps to cleanse sewage water, acts as an antioxidant, and scavenges free radicals in the presence of metal stress (Sharma and Dietz 2009). Proline’s enhanced production in plants indicates that it is responding to antioxidants by acting as a protein stabilizer, an O2 scavenger, and an inhibitor of -OH− (Sharma and Dietz 2009). According to Fatma et al. (2007), higher metabolism and reduced oxidative loss lead to increased proline content as a result of metal stress-oriented toxicity.
Influence of sewage wastewater on the total lipid content and lipid productivity of green microalgae
The data in Fig. 3 showed that when these green microalgae were cultured in sewage wastewater, the total lipid content and lipid productivity of the three microalgae significantly increased compared to the synthetic medium (control). M. braunii had the highest lipid content and lipid productivity increase, which was more than twofold higher than the control (synthetic medium). This result could be due to differences in biotic stress responses among algal species and a severe lack of all required elements in secondary treated sewage water, particularly nitrogen, which increased the accumulation of lipids (Rodríguez-Palacio et al. 2022). The findings of this study were consistent with those of Widjaja (2009) and Mostafa et al. (2012), who revealed that the green microalgae (C. vulgaris) accumulated a high lipid content when grown in nitrogen-depleted conditions (0.02 mg/L nitrate). Furthermore, the current findings were in accordance with those of Shalaby et al. (2010) that the overproduction of carbon skeleton altered algal metabolism under salt stress conditions (with normal nitrate concentrations in culture media), which was partly directed toward the production of substances with beneficial roles in algal tolerance or defense mechanisms such as polyols, methylated amino acids, carbohydrate, and protein. One major criterion for selecting microalgae strains as a renewable source for biofuel production is high lipid content (El-Sheekh et al. 2017a). Xin et al. (2010) found that Scenedesmus sp. has high lipid levels under stress conditions, but that the lipid productivity and the microalgal biomass productivity were reduced due to the low growth rate.
According to Francisco et al. (2010), lipid content and biomass productivity are inversely related. In the current study, using the results of Francisco et al. (2010), at the late exponential phase, the lipid content and biomass output in the tested species C. reinhardtii were inversely correlated. The correlation study did not include S. obliquus and M. braunii because they showed relatively low biomass production and high lipid content in the wastewater.
Fatty acids profile
The fatty acid composition of the three green microalgae cultivated in wastewater and the synthetic medium is displayed in Table 3. The results indicated that the two studied species (C. reinhardtii and M. braunii) have a high percentage of saturated fatty acids (80.91% and 68.62% of FAs, respectively) when grown in wastewater compared to synthetic medium, whereas there was a reduction in saturated fatty acid in S. obliquus (63.47% of TFAs) in wastewater corresponding to 72.24% of TFAs in the synthetic medium mainly composed of palmitic acid (C16:0), stearic acid (C18:0), and myristic acid (C14:0) which are the most abundant fatty acid. On the other hand, the largest concentration of monosaturated fatty acids (MUFAS), 17.71% of TFAs, was recorded in M. braunii grown in a synthetic medium, corresponding to 12.52% TFAs in wastewater due to the existence of palmitoleic acid (C16:1) and elaidic acid (C18:1). Finally, the total polyunsaturated fatty acids (PUFAS) in C. reinhardtii, M. braunii, and S. obliquus grown in wastewater increased to 12.0%, 13.27%, and 16.75% of TFAs, respectively, as compared to those in synthetic media (control) due to the presence of benzene propanoic acid (C17:3), linoleic acid (C18:2), and linolenic acid (C18:3).
These findings indicated a range in saturated and unsaturated fatty acid composition and a variation in fatty acid chain length between C-14 and C-19. Our findings were consistent with previous research demonstrating the removal of nutrients (TN and TP) by algal biomass and algal lipid content after cultivation in various municipal wastewater effluents (Abou-Shanab et al. 2014). The characteristics of biodiesel would be impacted by the chain length and saturation of fatty acids. Because of their high oxidation stability and avoiding cold flow, products with a high percentage of unsaturated fatty acids are not preferred in terms of quality (Krzemińska and Oleszek 2016).
Therefore, the transesterification process and biodiesel quality are positively affected by the large proportion of saturated fatty acids (Hu et al. 2008). According to the results, all microalgal species contain a high proportion of saturated fatty acids with C16–C18 carbon chains, which are highly recommended for the production of biodiesel, and only a small amount of linoleic acid (C18:3) is below 12%; their biomass is also suitable for the production of biodiesel from these microalgae species (Rodríguez-Palacio et al. 2022). Additionally, the high C16–C18 percentage enhances the performance and quality of biodiesel (Elshobary et al. 2020; Huo et al. 2020). Therefore, the fatty acid profile (FAMEs) produced by all species is suitable for synthesizing biodiesel based on the current research.
Biodiesel quality
For sustainable biodiesel production, selecting microalgal species requires high lipid productivity and acceptable characteristics of the produced fatty acid methyl esters (FAMEs). Table 4 shows the main physicochemical properties of biodiesel of the green microalgae cultivated in wastewater and synthetic medium compared to the international standards (ASTM D6751 in the US and EN 14,214 in Europe). The KV, CN, IV, SV, and ρ values are estimated in C. reinhardtii, S. obliquus, and M. braunii stayed within the bounds of the international standards, low KV (1.13 mm2 s−1), the relatively high CN (60.15), low IV value (43.12 g I2 100 g−1 oil) and high SV (199.83 mg KOH g−1) detected in C. reinhardtii. The KV, CN, IV, and SV in S. obliquus and M. braunii with a small difference between those in C. reinhardtii and all estimated results were in line with the international standards recommendations.
The average degree of unsaturation of S. obliquus (0.77) resulted in a higher iodine number but not more than 120 for high-quality biodiesel. Because the climate conditions in the USA and Europe differ, there are no clear guidelines for the cloud point (CP) (Knothe 2011). Optimum CP is the temperature that leads to wax cloud formation in the fuel. C. reinhardtii exhibited high CPs around 18.62 °C, enhancing biodiesel suitability under colder conditions. Fatty acid profile significantly affected the biodiesel quality (Ashour et al. 2019; Wang et al. 2018). The content of high saturated and low unsaturated fatty acids in our study was preferable to achieve good-quality biodiesel because saturated fatty acids have an advantageous effect on the transesterification and the quality of biodiesel (Hu et al. 2008).
In addition, a high CN value is preferable for biodiesel production, where a high value is proportionate with the ignition quality and oxidative stability and achievable at a lower unsaturation degree (Ma et al. 2013). On the contrary, a lower IV value is favorable for biodiesel production, and the lower IV value improves oxidative stability during prolonged storage (Yodsuwan et al. 2017). The SV values can reflect the highest fatty acid content.
Moreover, the high content of saturated fatty acids may decrease the CFPP properties of biodiesel because SFAs have significantly higher melting points than those of USFAs (Francisco et al. 2010). Therefore, C. reinhardtii, S. obliquus, and M. braunii demonstrated appropriate biodiesel properties in accordance with international standards, which could compete with fossil diesel.
Conclusion
Based on the results, wastewater recycling and reuse can be accomplished through bioremediation using various algal species such as Chlamydomonas reinhardtii, Scenedesmus obliquus, and Monoraphidium braunii. It is also concluded that because all algal species showed higher biomass and lipid productivities, they are a feedstock for biodiesel production. Research in the future should focus on the bioengineering of microalgae for the high lipid production and accumulation of fatty acid because it would be of highly promising way to meet the demand for energy using microalgae as a feedstock for third-generation biofuel.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Abdel-Raouf N, Al-Homaidan AA, Ibraheem IBM (2012) Microalgae and wastewater treatment. Saudi J Biol Sci 19:257–275. https://doi.org/10.1016/j.sjbs.2012.04.005
Abomohra AE-F, Wagner M, El-Sheekh M, Hanelt D (2013) Lipid and total fatty acid productivity in photoautotrophic fresh water microalgae: screening studies towards biodiesel production. J Appl Phycol 25:931–936. https://doi.org/10.1007/s10811-012-9917-y
Abou-Shanab RAI, Ji M-K, Kim H-C (2013) Microalgal species growing on piggery wastewater as a valuable candidate for nutrient removal and biodiesel production. J Environ Manage 115:257–264. https://doi.org/10.1016/j.jenvman.2012.11.022
Abou-Shanab RAI, El-Dalatony MM, El-Sheekh MM (2014) Cultivation of a new microalga, Micractinium reisseri, in municipal wastewater for nutrient removal, biomass, lipid, and fatty acid production. Biotechnol Bioprocess Eng 19:510–518. https://doi.org/10.1007/s12257-013-0485-z
Adefila EO, Onwordi CT, Ogunwande IA (2010) Level of heavy metals uptake on vegetables planted on poultry droppings dumpsite. Arch Appl Sci Res 2:347–353. www.scholarsresearchlibrary.com
American Public Health Association (APHA) (2005) Standard methods for the examination of water and wastewater, 21st edn. American Public Health Association, Washington, DC
Andrade MR, Costa JAV (2007) Mixotrophic cultivation of microalga Spirulina platensis using molasses as organic substrate. Aquaculture 264:130–134. https://doi.org/10.1016/j.aquaculture.2006.11.021
Ashour M, Elshobary ME, El-Shenody R (2019) Evaluation of a native oleaginous marine microalga Nannochloropsis oceanica for dual use in biodiesel production and aquaculture feed. Biomass Bioenerg 120:439–447. https://doi.org/10.1016/j.biombioe.2018.12.009
Azab YA (2002) The use of immobilized algae and algal biofilters for the treatment of industrial heavy metals pollution proceedings, plant and industrial pollution. Egypt Bot Soc Cairo 135–148
Azarpira H, Behdarvand P, Dhumal K, Pondhe G (2014) Comparative studies on phycoremediation of sewage water by using blue green algae. Int J Bio Sci 4:58–64
Badour SSA (1959) Analytisch–chemische Untersuchung des Kaliummangels bei Chlorella im Vergleich mit anderen Mangelzuständen. [Analytical-chemical investigation of potassium deficiency in Chlorella in comparison with other deficiencies]. Ger Göttingen Univ, Göttingen
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Chisti Y (2007) Biodiesel from microalgae. Biotechnol Adv 25:294–306. https://doi.org/10.1016/j.biotechadv.2007.02.001
Chisti Y (2008) Biodiesel from microalgae beats bioethanol. https://doi.org/10.1016/j.tibtech.2007.12.002
El-Otify AM, El-Sheekh M, Saber H (2011) Detrimental effects of industrial effluents on the growth and metabolic products of the Nile planktonic Cyanobacteria. Fresen Environ Bull 20(11):2911–2919
El-Sheekh MM, El-Shouny WA, Osman MEH, El-Gammal EWE (2005) Growth and heavy metals removal efficiency of Nostoc muscorum and Anabaena subcylindrica in sewage and industrial wastewater effluents. Environ Toxicol Pharmacol 19:357–365. https://doi.org/10.1016/j.etap.2004.09.005
El-Sheekh MM, Farghl AA, Galal HR, Bayoumi HS (2016a) Bioremediation of different types of polluted water using microalgae. Rend Lincei 27:401–410. https://doi.org/10.1007/s12210-015-0495-1
El-Sheekh M, Abomohra AE-F, Abd El-Azim M, Abou-Shanab R (2017a) Effect of temperature on growth and fatty acids profile of the biodiesel producing microalga Scenedesmus acutus. Biotechnol Agron Soc Environ 21(4):233–239. https://doi.org/10.25518/1780-4507.15291
El-Sheekh MM, Allam NG, Shabana SA, Azab MM (2017b) Efficiency of lipid accumulating Actinomycetes isolated from soil for biodiesel production: comparative study with microalgae. Energy Sources, Part A Recover Util Environ Eff 39:883–892. https://doi.org/10.1080/15567036.2016.1273279
El-Sheekh MM, Farghl AA, Bayoumi HS (2016b) Cultivation of Chlorella species in sewage and different water samples mixtures. Egypt J Exp Biol (Bot) 12(1): 97–106. https://www.bibliomed.org/?mno=229647
Elshobary ME, Essa DI, Attiah AM (2020) Algal community and pollution indicators for the assessment of water quality of Ismailia canal, Egypt. Stoch Environ Res Risk Assess 34:1089–1103. https://doi.org/10.21203/rs.3.rs-305271/v1
Emparan Q, Harun R, Danquah MK (2019) Role of phycoremediation for nutrient removal from wastewaters. Appl Ecol Environ Res 17(1):889–915. https://aloki.hu/pdf/1701_889915.pdf
Fatma T, Khan MA, Choudhary M (2007) Impact of environmental pollution on cyanobacterial proline content. J Appl Phycol 19:625–629. https://doi.org/10.1007/s10811-007-9195-2
Fawzy MA, Issa AA (2016) Bioremoval of heavy metals and nutrients from sewage plant by Anabaena oryzae and Cyanosarcina fontana. Int J Phytoremediation 18:321–328. https://doi.org/10.1080/15226514.2015.1094448
Francisco EC, Neves DB, Jacob Lopes E, Franco TT (2010) Microalgae as feedstock for biodiesel production: carbon dioxide sequestration, lipid production and biofuel quality. J Chem Technol Biotechnol 85:395–403. https://doi.org/10.1002/jctb.2338
Galgale AD, Shah NB, Shah NG (2014) Treatment of wastewater containing high concentration of phenol and total dissolved solids in moving bed biofilm reactor. IJIRSET 3(4):10924–10930. https://doi.org/10.1016/S0032-9592(02)00195-4
Hernández-García A, Velásquez-Orta SB, Novelo E (2019) Wastewater-leachate treatment by microalgae: biomass, carbohydrate and lipid production. Ecotoxicol Environ Saf 174:435–444. https://doi.org/10.1016/j.ecoenv.2019.02.052
Hoekman SK, Broch A, Robbins C (2012) Review of biodiesel composition, properties, and specifications. Renew Sustain Energy Rev 16:143–169. https://doi.org/10.1016/j.rser.2011.07.143
Hu Q, Sommerfeld M, Jarvis E (2008) Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J 54:621–639. https://doi.org/10.1111/j.1365-313X.2008.03492.x
Huo S, Basheer S, Liu F (2020) Bacterial intervention on the growth, nutrient removal and lipid production of filamentous oleaginous microalgae Tribonema sp. Algal Res 52:102088. https://doi.org/10.1016/j.algal.2020.102088
Kathryn E, Crystal D, Whitney G, Patrick G, McGinn J (2013) Nutrient remediation rates in municipal wastewater and their effect on biochemical composition of the microalga Scenedesmus sp. AMDD Algal Res 2:127–134. https://doi.org/10.1016/j.algal.2013.01.009
Knothe G (2011) A technical evaluation of biodiesel from vegetable oils vs. algae. Will algae-derived biodiesel perform? Green Chem 13:3048–3065. https://doi.org/10.1039/C0GC00946F
Krzemińska I, Oleszek M (2016) Glucose supplementation-induced changes in the Auxenochlorella protothecoides fatty acid composition suitable for biodiesel production. Bioresour Technol 218:1294–1297. https://doi.org/10.1016/j.biortech.2016.07.104
Kshirsagar AD (2014) Remediation of domestic wastewater by using algal and fungal mixed culture: an experimental study. Int Interdiscip Res J 4:166–173
Leong YK, Chang J-S (2020) Bioremediation of heavy metals using microalgae: recent advances and mechanisms. Bioresour Technol 303:122886. https://doi.org/10.1016/j.biortech.2020.122886
Lorenzen H (1964) Synchronization of Chlorella with light dark changes and periodical dilution to a standard cell number. Inersci Publ, New York, p 571
Lowry O, Rosebrough N, Farr AL, Randall R (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275. https://doi.org/10.1016/s0021-9258(19)52451-6
Lv J-M, Cheng L-H, Xu X-H (2010) Enhanced lipid production of Chlorella vulgaris by adjustment of cultivation conditions. Bioresour Technol 101:6797–6804. https://doi.org/10.1016/j.biortech.2010.03.120
Ma M, Zhu W, Wang Z, Witkamp GJ (2003) Accumulation, assimilation and growth inhibition of copper on freshwater alga (Scenedesmus subspicatus 86.81 SAG) in the presence of EDTA and fulvic acid. Aquat Toxicol 63:221–228. https://doi.org/10.1016/S0166-445X(02)00179-0
Ma Y, Wang Z, Zhu M (2013) Increased lipid productivity and TAG content in Nannochloropsis by heavy-ion irradiation mutagenesis. Bioresour Technol 136:360–367. https://doi.org/10.1016/j.biortech.2013.03.020
Metzner H, Rau H, Senger H (1965) Untersuchungen zur synchronisierbarkeit einzelner pigmentmangel-mutanten von Chlorella. Planta 65:186–194. https://doi.org/10.1007/BF00384998
Mostafa SSM, Shalaby EA, Mahmoud GI (2012) Cultivating microalgae in domestic wastewater for biodiesel production. Not Sci Biol 4: 56–65. https://www.notulaebiologicae.ro/index.php/nsb
Park H, Jung D, Lee J, Kim P, Cho Y, Jung I, Kim Z-H, Lim S-M, Lee C-G (2018) Improvement of biomass and fatty acid productivity in ocean cultivation of Tetraselmis sp. using hypersaline medium. J Appl Phyco 30:2725–2735. https://doi.org/10.1007/s10811-018-1388-3
Peña-Castro JM, Martínez-Jerónimo F, Esparza-García F, Cañizares-Villanueva RO (2004) Phenotypic plasticity in Scenedesmus incrassatulus (Chlorophyceae) in response to heavy metals stress. Chemosphere 57:1629–1636. https://doi.org/10.1016/j.chemosphere.2004.06.041
Perez-Garcia O, Escalante FME, De-Bashan LE, Bashan Y (2011) Heterotrophic cultures of microalgae: metabolism and potential products. Water Res 45:11–36. https://doi.org/10.1016/j.watres.2010.08.037
Persoone G, Morales J, Verlet H, De Pauw N (1980) Air-lift pumps and the effect of mixing on algal growth. Algal Biomass 11:505–522
Prescott-Allen R, Prescott-Allen C (1982) What’s wildlife worth? Economic contributions of wild plants and animals to developing countries. Earthscan, p 92
Radwan SS (1978) Coupling of two-dimensional thin-layer chromatography with gas chromatography for the quantitative analysis of lipid classes and their constituent fatty acids. J Chromatogr Sci 16:538–542. https://doi.org/10.1093/chromsci/16.11.538
Rao P, Kumar RR, Raghavan BG et al (2011) Application of phycoremediation technology in the treatment of wastewater from a leather-processing chemical manufacturing facility. Water Sa 37(1):2011. https://doi.org/10.4314/wsa.v37i1.64099
Rath B (2012) Microalgal bioremediation: current practices and perspectives. J Biochem Technol 3:299–304
Refaat AA (2009) Correlation between the chemical structure of biodiesel and its physical properties. Int J Environ Sci Technol 6:677–694. https://doi.org/10.1007/BF03326109
Rodríguez-Palacio MC, Cabrera-Cruz RBE, Rolón-Aguilar JC, Tobias-Jaramillo R, Martinez-Hernandez M, Lozano-Ramirez C (2022) The cultivation of five microalgae species and their potential for biodiesel production. Energ Sustain Soc 12:1–7. https://doi.org/10.1186/s13705-022-00337-5
Romera E, Gonzalez F, Ballester A (2006) Biosorption with algae: a statistical review. Crit Rev Biotechnol 26:223–235. https://doi.org/10.1080/07388550600972153
Sattayawat P, Yunus IS, Noirungsee N (2021) Synthetic biology-based approaches for microalgal bio-removal of heavy metals from wastewater effluents. Front Environ Sci 562. https://doi.org/10.3389/fenvs.2021.778260
Shalaby EA, Shanab SMM, Singh V (2010) Salt stress enhancement of antioxidant and antiviral efficiency of Spirulina platensis. J Med Plants Res 4:2622–2632. https://doi.org/10.5897/JMPR09.300
Sharma SS, Dietz K-J (2009) The relationship between metal toxicity and cellular redox imbalance. Trends Plant Sci 14:43–50. https://doi.org/10.1016/j.tplants.2008.10.0
Song M, Pei H, Hu W, Ma G (2013) Evaluation of the potential of 10 microalgal strains for biodiesel production. Bioresour Technol 141:245–251. https://doi.org/10.1016/j.biortech.2013.02.024
Stein JR (1966) Growth and mating of Gonium pectorale (volvocales) in defined media 1. J Phycol 2:23–28. https://doi.org/10.1111/j.1529-8817.1966.tb04587.x
Tan Y, Lin J (2011) Biomass production and fatty acid profile of a Scenedesmus rubescens-like microalga. Bioresour Technol 102:10131–10135. https://doi.org/10.1016/j.biortech.2011.07.091
Tiruneh AT, Fadiran AO, Mtshali JS (2014) Evaluation of the risk of heavy metals in sewage sludge intended for agricultural application in Swaziland. Int J Environ Sci 5(1):197–216. https://doi.org/10.6088/ijes.2014050100017
Touliabah HE, El-Sheekh MM, Ismail MM, El-kassas H (2022) A review of microalgae-and cyanobacteria-based biodegradation of organic pollutants. Molecules 27:1141. https://doi.org/10.3390/molecules27031141
Tripathi BN, Gaur JP (2006) Physiological behavior of Scenedesmus sp. during exposure to elevated levels of Cu and Zn and after withdrawal of metal stress. Protoplasma 229:1–9. https://doi.org/10.1007/s00709-006-0196-9
Vidali M (2001) Bioremediation. An overview. Pure Appl Chem 73:1163–1172. https://doi.org/10.1351/pac200173071163
Wang Y, Davidson MCG (2010) A review of micro-analyses of tourist expenditure. Curr Issues Tour 13:507–524. https://doi.org/10.1080/13683500903406359
Wang S, Jiang D, Cao B (2018) Study on the interaction effect of seaweed bio-coke and rice husk volatiles during co-pyrolysis. J Anal Appl Pyrolysis 132:111–122. https://doi.org/10.1016/j.jaap.2018.03.009
Widjaja A (2009) Lipid production from microalgae as a promising candidate for biodiesel production. Makara J Technol 13:9. https://doi.org/10.7454/mst.v13i1.496
Woertz I, Feffer A, Lundquist T, Nelson Y (2009) Algae grown on dairy and municipal wastewater for simultaneous nutrient removal and lipid production for biofuel feedstock. J Environ Eng 135(11):1115–1122. https://doi.org/10.1061/(ASCE)EE.1943-7870.0000129
Xin L, Hong-Ying H, Ke G, Ying-Xue S (2010) Effects of different nitrogen and phosphorus concentrations on the growth, nutrient uptake, and lipid accumulation of a freshwater microalga Scenedesmus sp. Bioresour Technol 101:5494–5500. https://doi.org/10.1016/j.biortech.2010.02.016
Yodsuwan N, Sawayama S, Sirisansaneeyakul S (2017) Effect of nitrogen concentration on growth, lipid production and fatty acid profiles of the marine diatom Phaeodactylum tricornutum. Agric Nat Resour 51:190–197. https://doi.org/10.1016/j.anres.2017.02.004
Zhang E, Wang B, Wang Q, Zhang S, Zhao B (2008) Ammonia–nitrogen and orthophosphate removal by immobilized Scenedesmus sp. isolated from municipal wastewater for potential use in tertiary treatment. Bioresour Technol 99:3787–3793. https://doi.org/10.1016/j.biortech.2007.07.011
Zhou Q, Zhang H, Fu C (2018) The distribution and morphology of microplastics in coastal soils adjacent to the Bohai Sea and the Yellow Sea. Geoderma 322:201–208. https://doi.org/10.1016/j.geoderma.2018.02.015
Znad H, Awual MR, Martini S (2022) The utilization of algae and seaweed biomass for bioremediation of heavy metal-contaminated wastewater. Molecules 27:1275. https://doi.org/10.3390/molecules27041275
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
Mostafa El-Sheekh supervised the work and wrote and revised the final manuscript; Hamdy Galal supervised the work; Amal Mousa carried out the practical work and the final tables and figures; Abla Farghl wrote the first draft of the manuscript and prepared the figures and statistical analysis of the results. All authors have read and approved the manuscript”.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Ta Yeong Wu
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
El-Sheekh, M.M., Galal, H.R., Mousa, A.S.H. et al. Coupling wastewater treatment, biomass, lipids, and biodiesel production of some green microalgae. Environ Sci Pollut Res 30, 35492–35504 (2023). https://doi.org/10.1007/s11356-023-25628-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-25628-y