Abstract
Introduction
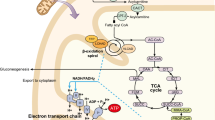
The m.3243A > G mitochondrial DNA mutation is one of the most common mitochondrial disease-causing mutations, with a carrier rate as high as 1:400. This point mutation affects the MT-TL1 gene, ultimately affecting the oxidative phosphorylation system and the cell’s energy production. Strikingly, the m.3243A > G mutation is associated with different phenotypes, including mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes (MELAS), maternally inherited diabetes and deafness (MIDD) and myopathy.
Objectives
We investigated urine metabolomes of MELAS, MIDD and myopathy patients in order to identify affected metabolic pathways and possible treatment options.
Methods
A multiplatform metabolomics approach was used to comprehensively analyze the metabolome and compare metabolic profiles of different phenotypes caused by the m.3243A > G mutation. Our analytical array consisted of NMR spectroscopy, LC-MS/MS and GC-TOF-MS.
Results
The investigation revealed phenotypic specific metabolic perturbations, as well as metabolic similarities between the different phenotypes. We show that glucose metabolism is highly disturbed in the MIDD phenotype, but not in MELAS or myopathy, remodeled fatty acid oxidation is characteristic of the MELAS patients, while one-carbon metabolism is strongly modified in both MELAS and MIDD, but not in the myopathy group. Lastly we identified increased creatine in the urine of the myopathy patients, but not in MELAS or MIDD.
Conclusion
We conclude by giving novel insight on the phenotypes of the m.3243A > G mutation from a metabolomics point of view. Directives are also given for future investigations that could lead to better treatment options for patients suffering from this debilitating disease.







Similar content being viewed by others
Abbreviations
- 2-HG:
-
2-Hydroxyglutaric acid
- ADP:
-
Adenosine diphosphate
- ANOVA:
-
Analysis of variance
- ATP:
-
Adenosine triphosphate
- AUC:
-
Area under the curve
- BCAA:
-
Branched-chain amino acid
- Complex V:
-
ATP synthase
- FAD:
-
Oxidized flavin adenine dinucleotide
- FADH2 :
-
Reduced flavin adenine dinucleotide
- FAS:
-
Fatty acids synthesis
- GC–TOF–MS:
-
Gas chromatography time-of-flight mass spectrometry
- KSS:
-
Kearns–Sayre syndrome
- LC–MS/MS:
-
Liquid chromatography tandem mass spectrometry
- LS:
-
Leigh syndrome
- MCFA:
-
Medium chain fatty acids
- MD:
-
Mitochondrial disease
- MELAS:
-
Mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes
- MERRF:
-
Myoclonus epilepsy with ragged red fibers
- MIDD:
-
Maternally inherited diabetes and deafness
- MS:
-
Mass spectrometry
- mtDNA:
-
Mitochondrial DNA
- mTOR:
-
mammalian target of rapamycin
- MT-TL1:
-
Mitochondria-encoded tRNA leucine 1
- NAD+ :
-
Oxidized nicotine amide adenine dinucleotide
- NADH:
-
Reduced nicotine amide adenine dinucleotide
- NMR:
-
Proton nuclear magnetic resonance spectroscopy
- OXPHOS:
-
Oxidative phosphorylation
- PCA:
-
Principal component analysis
- PCr:
-
Phosphocreatine
- PEO:
-
progressive external ophthalmoplegia
- ROC:
-
Receiver operating characteristic
- SCFA:
-
Short chain fatty acids
- SI:
-
Supplementary information
- TCA:
-
Tricarboxylic acid
References
Baertling, F., Rodenburg, R. J., Schaper, J., Smeitink, J. A., Koopman, W. J., Mayatepek, E., et al. (2014). A guide to diagnosis and treatment of Leigh syndrome. Journal of Neurology, Neurosurgery, and Psychiatry, 85(3), 257–265.
Bao, X. R., Ong, S. E., Goldberger, O., Peng, J., Sharma, R., Thompson, D. A., et al. (2016). Mitochondrial dysfunction remodels one-carbon metabolism in human cells. eLife., 5, e10575.
Brière, J., Chrétien, D., Bénit, P., & Rustin, P. (2004). Respiratory chain defects: What do we know for sure about their consequences in vivo? Biochimica Biophysica Acta (BBA). 1659 (2), 172–177.
Buzkova, J., Nikkanen, J., Ahola, S., Hakonen, A. H., Sevastianova, K., Yki-Järvinen, H. T., et al. (2018). Metabolomes of mitochondrial diseases and inclusion body myositis patients: Treatment targets and biomarkers. EMBO Molecular Medicine, 10, e9091.
Carbonneau, M., Gagné, M., Lalonde, L., Germain, M. E., Motorina, M. A., Guiot, A., et al. (2016). The oncometabolite 2-hydroxyglutarate activates the mTOR signalling pathway. Nature Communications, 7, 12700.
Chin, R. M., Fu, X., Pai, M. Y., Vergnes, L., Hwang, H., Deng, G., et al. (2014). The metabolite α-ketoglutarate extends lifespan by inhibiting ATP synthase and TOR. Nature., 510(7505), 397–401.
El-Hattab, A. W., Adesina, A. M., Jones, J., & Scaglia, F. (2015). MELAS syndrome: Clinical manifestations, pathogenesis, and treatment options. Molecular Genetics and Metabolism, 116(1), 4–12.
Esterhuizen, K., Lindeque, J. Z., Mason, S., van der Westhuizen, F. H., Suomalainen, A., Hakonen, A. H., et al. (2019). A urinary biosignature for mitochondrial myopathy, encephalopathy, lactic acidosis and stroke like episodes (MELAS). Mitochondrion., 45, 38–45.
Esterhuizen, K., van der Westhuizen, F. H., & Louw, R. (2017). Metabolomics of mitochondrial disease. Mitochondrion., 35, 97–110.
Fiehn, O. (2002). Metabolomics—the link between genotypes and phenotypes. Plant Molecular Biology, 48, 155–171.
Finsterer, J. (2007). Genetic, pathogenetic, and phenotypic implications of the mitochondrial A3243G tRNALeu (UUR) mutation. Acta Neurologica Scandinavica, 116(1), 1–14.
Fu, X., Chin, R. M., Vergnes, L., Hwang, H., Deng, G., Xing, Y., et al. (2015). 2-Hydroxyglutarate inhibits ATP synthase and mTOR signalling. Cell Metabolism, 22(3), 508–515.
Goto, Y., Nonaka, I., & Horai, S. (1990). A mutation in the tRNA (Leu (UUR)) gene associated with the MELAS subgroup of mitochondrial encephalomyopathies. Nature., 348(6302), 651–653.
Hall, A. M., Vilasi, A., Garcia-Perez, I., Lapsley, M., Alston, C. L., Pitceathly, R. D., et al. (2015). The urinary proteome and metabonome differ from normal in adults with mitochondrial disease. Kidney International, 87(3), 610–622.
Hernández-Luis, C., García-Morán, E., Rubio-Sanz, J., & Fernández-Avilés, F. (2007). Kearns- Sayre syndrome: Recurrent syncope and atrial flutter. Revista Española de Cardiología (English Edition)., 60(1), 89–90.
Hiltunen, J. K., Schonauer, M. S., Autio, K. J., Mittelmeier, T. M., Kastaniotis, A. J., & Dieckmann, C. L. (2009). Mitochondrial fatty acid synthesis type II: More than just fatty acids. The Journal of Biological Chemistry, 284(14), 9011–9015.
Holm, S. (1979). A simple sequentially rejective multiple test procedure. Scandinavian Journal of Statistics, 6(2), 65–70.
Johnson, S. C., Yanos, M. E., Kayser, E. B., Quintana, A., Sangesland, M., Castanza, A., et al. (2013). mTOR inhibition alleviates mitochondrial disease in a mouse model of Leigh syndrome. Science, 342(6165), 1524–1528.
Kami, K., Fujita, Y., Igarashi, S., Koike, S., Sugawara, S., Ikeda, S., et al. (2012). Metabolomic profiling rationalized pyruvate efficacy in cybrid cells harboring MELAS mitochondrial DNA mutations. Mitochondrion., 12(6), 644–653.
Kassel, D., Martin, M., Schall, W., & Sweeley, C. (1986). Urinary metabolites of L-threonine in type 1 diabetes determined by combined gas chromatography/chemical ionization mass spectrometry. Biological Mass Spectrometry, 13(10), 535–540.
Kaufmann, P., Engelstad, K., Wei, Y., Kulikova, R., Oskoui, M., Battista, V., et al. (2009). Protean phenotypic features of the A3243G mitochondrial DNA mutation. Archives of Neurology, 66(1), 85–91.
Kennedy, A. D., Miller, M. J., Beebe, K., Wulff, J. E., Evans, A. M., Miller, L. A. D., et al. (2016). Metabolomic profiling of human urine as a screen for multiple inborn errors of metabolism. Genetic Testing and Molecular Biomarkers, 20(9), 485–495.
Khamis, M. M., Holt, T., Awad, H., El-Aneed, A., & Adamko, D. J. (2018). Comparative analysis of creatinine and osmolality as urine normalization strategies in targeted metabolomics for the differential diagnosis of asthma and COPD. Metabolomics, 14(9), 115.
Khan, N. A., Auranen, M., Paetau, I., Pirinen, E., Euro, L., Forsström, S., et al. (2014). Effective treatment of mitochondrial myopathy by nicotinamide riboside, a vitamin B3. EMBO Molecular Medicine., 6(6), 721–731.
Lorenzoni, P. J., Scola, R. H., Kay, C. S. K., Silvado, C. E. S., & Werneck, L. C. (2014). When should MERRF (myoclonus epilepsy associated with ragged-red fibers) be the diagnosis? Arquivos de Neuro-Psiquiatria, 72(10), 803–811.
Lorenzoni, P. J., Werneck, L. C., Kay, C. S. K., Silvado, C. E. S., & Scola, R. H. (2015). When should MELAS (Mitochondrial myopathy, Encephalopathy, Lactic Acidosis, and Stroke-like episodes) be the diagnosis? Arquivos de Neuro-Psiquiatria., 73(11), 959–967.
Löwik, M. M., Hol, F. A., Steenbergen, E. J., Wetzels, J. F. M., & van den Heuvel, L. P. W. J. (2005). Mitochondrial tRNALeu(UUR) mutation in a patient with steroid-resistant nephrotic syndrome and focal segmental glomerulosclerosis. Nephrology Dialysis Transplantation., 20(2), 336–341.
Manwaring, N., Jones, M. M., Wang, J. J., Rochtchina, E., Howard, C., Mitchell, P., et al. (2007). Population prevalence of the MELAS A3243G mutation. Mitochondrion, 7(3), 230–233.
Marshall, D. D., & Powers, R. (2017). Beyond the paradigm: Combining mass spectrometry and nuclear magnetic resonance for metabolomics. Progress in Nuclear Magnetic Resonance Spectroscopy, 100, 1–16.
McLaughlin, M. J., & Sainani, K. L. (2014). Bonferroni, Holm, and Hochberg corrections: Fun names, serious changes to p values. Physical Medicine and Rehabilitation, 6(6), 544–546.
Mignarri, A., Cenciarelli, S., Da Pozzo, P., Cardaioli, E., Malandrini, A., Federico, A., et al. (2015). Mitochondrial recessive ataxia syndrome: A neurological rarity not to be missed. Journal of the Neurological Sciences, 349(1–2), 254–255.
Muoio, D. M., & Newgard, C. B. (2008). Fatty acid oxidation and insulin action: When less is more. Diabetes, 57(6), 1455–1456.
Murphy, R., Turnbull, D., Walker, M., & Hattersley, A. (2008). Clinical features, diagnosis and management of maternally inherited diabetes and deafness (MIDD) associated with the 3243A > G mitochondrial point mutation. Diabetic Medicine, 25(4), 383–399.
Naviaux, R. K. (2014). Metabolic features of the cell danger response. Mitochondrion, 16, 7–17.
Nikkanen, J., Forsström, S., Euro, L., Paetau, I., Kohnz, R. A., Wang, L., et al. (2016). Mitochondrial DNA replication defects disturb cellular dNTP pools and remodel one-carbon metabolism. Cell Metabolism, 23(4), 635–648.
Padmalayam, I., Hasham, S., Saxena, U., & Pillarisetti, S. (2009). Lipoic acid synthase (LASY): A novel role in inflammation, mitochondrial function, and insulin resistance. Diabetes, 58(3), 600–608.
Park, S. Y., Kim, S. H., & Lee, Y. (2017). Molecular diagnosis of myoclonus epilepsy associated with ragged-red fibers syndrome in the absence of ragged red fibers. Frontiers in Neurology, 8, 520.
Pinto, M., & Moraes, C. T. (2014). Mitochondrial genome changes and neurodegenerative diseases. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease, 1842(8), 1198–1207.
Pisters, P. W., Cersosimo, E., Rogatko, A., & Brennan, M. F. (1992). Insulin action on glucose and branched-chain amino acid metabolism in cancer cachexia: Differential effects of insulin. Surgery, 111(3), 301–310.
Reinecke, C. J., Koekemoer, G., van der Westhuizen, F. H., Louw, R., Lindeque, J. Z., Mienie, L. J., et al. (2012). Metabolomics of urinary organic acids in respiratory chain deficiencies in children. Metabolomics., 8(2), 264–283.
Rosen Vollmar, A. K., Rattray, N. J. W., Cai, Y., Santos-Neto, A. J., Deziel, N. C., Jukic, A. M. Z., & Johnson, C. H. (2019). Normalizing untargeted periconceptional urinary metabolomics data: A comparison of approaches. Metabolites, 9(10), 198.
Rowland, L. P. (1993). Progressive external ophthalmoplegia. Handbook of Clinical Neurology, 22, 177–202.
Sage-Schwaede, A., Engelstad, K., Salazar, R., Curcio, A., Khandji, A., Garvin, J. H. Jr., et al. (2019). Exploring mTOR inhibition as treatment for mitochondrial disease. Annals of Clinical and Translational Neurology, 6(9), 1877–1881.
Santa, K. M. (2010). Treatment options for mitochondrial myopathy, encephalopathy, lactic acidosis, and stroke-like episodes (MELAS) syndrome. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy, 30(11), 1179–1196.
Scaglia, F., & Northrop, J. L. (2006). The mitochondrial myopathy encephalopathy, lactic acidosis with stroke-like episodes (MELAS) syndrome: A review of treatment options. CNS Drugs., 20(6), 443–464.
Schymanski, E. L., Jeon, J., Gulde, R., Fenner, K., Ruff, M., Singer, H. P., et al. (2014). Identifying small molecules via high resolution mass spectrometry: Communicating confidence. Environmental Science & Technology, 48, 2097–2098.
Smeitink, J. A., Zeviani, M., Turnbull, D. M., & Jacobs, H. T. (2006). Mitochondrial medicine: A metabolic perspective on the pathology of oxidative phosphorylation disorders. Cell Metabolism, 3(1), 9–13.
Smuts, I., van der Westhuizen, F. H., Louw, R., Mienie, L. J., Engelke, U. F., Wevers, R. A., et al. (2013). Disclosure of a putative biosignature for respiratory chain disorders through a metabolomics approach. Metabolomics., 9(2), 379–391.
Xia, J., & Wishart, D. S. (2016). Using MetaboAnalyst 3.0 for comprehensive metabolomics data analysis. Current Protocols in Bioinformatics, 55, 10–14.
Yang, J., Chen, T., Sun, L., Zhao, Z., Qi, X., Zhou, K., et al. (2013) Potential metabolite markers of schizophrenia. Molecular Psychiatry, 18, 67–78.
Yi, X., & Maeda, N. (2005). Endogenous production of lipoic acid is essential for mouse development. Molecular and Cellular Biology, 25(18), 8387–8392.
Zhang, A., Sun, H., Wang, P., Han, Y., & Wang, X. (2012). Modern analytical techniques in metabolomics analysis. The Analyst, 137(2), 293–300.
Acknowledgements
We would like to thank Prof. L.J. Mienie from the North-West University, Potchefstroom, South Africa, for valuable help with the interpretation of the metabolic data. The research was supported by a grant from the Technology Innovation Agency (TIA, Grant Number Metabol. 01), South Africa. The research facilities were provided by the National Metabolomics Platform, North-West University, Potchefstroom Campus, Potchefstroom, South Africa.
Author information
Authors and Affiliations
Contributions
RL, MJ, ZL, FVDW and JS planned and oversaw the study. KE, ZL and SM performed all analytical work. Data mining was performed by RL, ZL, SM and KE. The manuscript was written by KE, RL and ZL, assisted by all the authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests. Jan Smeitink is the founding CEO of Khondrion.
Ethical approval
The study complied with all Institutional guidelines and terms of the Declaration of Helsinki of 1975 (as revised in 2013) for investigation of human participants. Ethical approval was obtained from the Health Research Ethics Committee (HREC) of the North-West University, South Africa (NWU-00170-13-S1) and the Ethics Committee of the Nijmegen-Arnhem region of the Netherlands (2010/183; NL32683.091.10). All the patients, as well as the controls, provided written informed consent for their urine samples to be used for these research purposes.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Esterhuizen, K., Lindeque, J.Z., Mason, S. et al. One mutation, three phenotypes: novel metabolic insights on MELAS, MIDD and myopathy caused by the m.3243A > G mutation. Metabolomics 17, 10 (2021). https://doi.org/10.1007/s11306-020-01769-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-020-01769-w