Abstract
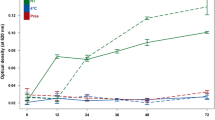
In nutritional metabolomics a large inter- and intra-subject variability exists, and thus, it becomes important to limit the variance introduced by external factors. In a composite controlled study with full provision of all food for the standardized intervention, human urinary metabolite profiles were investigated for different factors, such as handling of urine collections, diet standardization, diet culture, cohabitation and gender. In study A, 8 healthy subjects (4 men; 4 women) collected 24-h urine, splitting each void into two specimens stored either at 4°C or at room temperature. In study B, 16 healthy subjects (7 men; 9 women) collected 24-h urine for three days while being on a standardized diet. Samples were analyzed by 1H NMR and chemometrics. The NMR profiles indicated the presence of metabolites presumably originating from bacterial contamination in 3 out of 16 sample collections stored at room temperature. On the contrary, no changes in the NMR profiles due to contamination occurred in the 24-h urine samples stored at 4°C. The study also showed a trend towards a reduced inter- and intra-individual variation during 3 days of diet standardization. In study A, the urine metabolome showed a clear effect of diet culture and cohabitation, but these effects significantly attenuated after diet standardization (study B). Besides, gender-specific differences were found in both studies. Our results emphasize that best practice for any metabolomic study is a standardized, chilled sample collection procedure, and recommend that diet standardization is performed prior to dietary interventions in order to reduce intra- and inter-subject variability.





Similar content being viewed by others
References
Assfalg, M., Bertini, I., Colangiuli, D., et al. (2008). Evidence of different metabolic phenotypes in humans. Proceedings of the National Academy of Sciences of the United States of America, 105, 1420–1424.
Barton, R. H., Nicholson, J. K., Elliott, P., & Holmes, E. (2008). High-troughput 1H NMR-based metabolic analysis of human serum and urine for large-scale epidemiological studies: validation study. International Journal of Epidemiology, 37, 31–40.
Beckonert, O., Keun, H. C., Ebbels, T. M., et al. (2007). Metabolic profiling, metabolomic and metabonomic procedures for NMR spectroscopy of urine, plasma, serum and tissue extracts. Nature Protocols, 2, 2692–2703.
Bollard, M. E., Stanley, E. G., Lindon, J. C., Nicholson, J. K., & Holmes, E. (2005). NMR-based metabonomic approaches for evaluating physiological influences on biofluid composition. NMR in Biomedicine, 18, 143–162.
Broadhurst, D., & Kell, D. B. (2006). Statistical strategies for avoiding false discoveries in metabolomics and related experiments. Metabolomics, 2, 171–196.
Craig, A., Cloareo, O., Holmes, E., Nicholson, J. K., & Lindon, J. C. (2006). Scaling and normalization effects in NMR spectroscopic metabonomic data sets. Analytical Chemistry, 78, 2262–2267.
Denmark, T. N. F. A. o. (2006). DANKOST. Denmark: Herlev.
Dieterle, F., Ross, A., Schlotterbeck, G., & Senn, H. (2006). Probabilistic quotient normalization as robust method to account for dilution of complex biological mixtures. Application in 1H NMR metabonomics. Analytical Chemistry, 78, 4281–4290.
Dragsted, L. O. (2009). Biomarkers of meat intake and the application of nutrigenomics. Meat Science, 84, 301–307.
Dunn, W. B., Bailey, N. J., & Johnson, H. E. (2005). Measuring the metabolome: current analytical technologies. Analyst, 130, 606–625.
Gika, H. G., Theodoridis, G. A., Wingate, J. E., & Wilson, I. D. (2007). Within-day reproducibility of an HPLC-MS-based method for metabonomic analysis: Application to human urine. Journal of Proteome Research, 6, 3291–3303.
Goodacre, R., Broadhurst, D., Smilde, A. K., et al. (2007). Proposed minimum reporting standards for data analysis in metabolomics. Metabolomics, 3, 231–241.
Holmes, E., Foxall, P. J., Spraul, M., et al. (1997). 750 MHz 1H NMR spectroscopy characterisation of the complex metabolic pattern of urine from patients with inborn errors of metabolism: 2-hydroxyglutaric aciduria and maple syrup urine disease. Journal of Pharmaceutical and Biomedical Analysis, 15, 1647–1659.
Hotelling, H. (1933). Analysis of a complex of statistical variables into principal components. Journal of Educational Psychology, 24, 498–520.
Jenab, M., Slimani, N., Bictash, M., Ferrari, P., & Bingham, S. A. (2009). Biomarkers in nutritional epidemiology: applications, needs and new horizons. Human Genetics, 125, 507–525.
Kochhar, S., Jacobs, D. M., Ramadan, Z., et al. (2006). Probing gender-specific metabolism differences in humans by nuclear magnetic resonance-based metabonomics. Analytical Biochemistry, 352, 274–281.
Lauridsen, M., Hansen, S. H., Jaroszewski, J. W., & Cornett, C. (2007). Human urine as test material in 1H NMR-based metabonomics: recommendations for sample preparation and storage. Analytical Chemistry, 79, 1181–1186.
Lenz, E. M., Bright, J., Wilson, I. D., Morgan, S. R., & Nash, A. F. (2003). A 1H NMR-based metabonomic study of urine and plasma samples obtained from healthy human subjects. Journal of Pharmaceutical and Biomedical Analysis, 33, 1103–1115.
Lenz, E. M., Bright, J., Wilson, I. D., et al. (2004). Metabonomics, dietary influences and cultural differences: a 1H NMR-based study of urine samples obtained from healthy British and Swedish subjects. Journal of Pharmaceutical and Biomedical Analysis, 36, 841–849.
Lindon, J. C., Holmes, E., & Nicholson, J. K. (2006). Metabonomics techniques and applications to pharmaceutical research & development. Pharmaceutical Research, 23, 1075–1088.
Maher, A. D., Zirah, S. F., Holmes, E., & Nicholson, J. K. (2007). Experimental and analytical variation in human urine in 1H NMR spectroscopy-based metabolic phenotyping studies. Analytical Chemistry, 79, 5204–5211.
Nicholls, A. W., Mortishire-Smith, R. J., & Nicholson, J. K. (2003). NMR spectroscopic-based metabonomic studies of urinary metabolite variation in acclimatizing germ-free rats. Chemical Research in Toxicology, 16, 1395–1404.
Nicholson, J. K., Connelly, J., Lindon, J. C., & Holmes, E. (2002). Metabonomics: a platform for studying drug toxicity and gene function. Nature reviews Drug discovery, 1, 153–161.
Nicholson, J. K., Foxall, P. J., Spraul, M., Farrant, R. D., & Lindon, J. C. (1995). 750 MHz 1H and 1H–13C NMR spectroscopy of human blood plasma. Analytical Chemistry, 67, 793–811.
Nicholson, J. K., O’Flynn, M. P., Sadler, P. J., et al. (1984). Proton-nuclear-magnetic-resonance studies of serum, plasma and urine from fasting normal and diabetic subjects. Biochemical Journal, 217, 365–375.
Noah, A., & Truswell, A. S. (2001). There are many Mediterranean diets. Asia Pacific Journal of Clinical Nutrition, 10, 2–9.
Nørgaard, L., Bro, R., Westad, F., & Engelsen, S. B. (2006). A modification of canonical variates analysis to handle highly collinear multivariate data. Journal of Chemometrics, 20, 425–435.
Parry, D. M., & Duerksen, D. R. (2001). Assessment of intestinal permeability with lactulose/mannitol: gum chewing is a potential confounding factor. American Journal of Gastroenterology, 96, 2515–2516.
Pasquali, R., Baraldi, G., Biso, P., et al. (1984). Effect of physiological doses of triiodothyronine replacement on the hormonal and metabolic adaptation to short-term semistarvation and to low-calorie diet in obese patients. Clinical Endocrinology, 21, 357–367.
Pedersen, A. N., Fagt, S., Groth, M. V., et al. (2010). Danskernes kostvaner 2003–2008. Hovedresultater (Dietary habits of the Danes, 2003–2008. Main results) Danish. R. S. G. A/S. Søborg, Demnark, Danish National Food Institute, The Technical University of Denmark.
Saude, E. J., Adamko, D., Rowe, B. H., Marrie, T., & Sykes, B. D. (2007). Variation of metabolites in normal human urine. Metabolomics, 3, 439–451.
Saude, E. J., & Sykes, B. D. (2007). Urine stability for metabolomic studies: effects of preparation and storage. Metabolomics, 3, 19–27.
Savorani, F., Picone, G., Badiani, A., et al. (2010b). Metabolic profiling and aquaculture differentiation of gilthead sea bream by 1H NMR metabonomics. Food Chemistry, 120, 907–914.
Savorani, F., Tomasi, G., & Engelsen, S. B. (2010a). icoshift: A versatile tool for the rapid alignment of 1D NMR spectra. Journal of Magnetic Resonance, 202, 190–202.
Scalbert, A., Brennan, L., Fiehn, O., et al. (2009). Mass-spectrometry-based metabolomics: limitations and recommendations for future progress with particular focus on nutrition research. Metabolomics, 5, 435–458.
Slupsky, C. M., Rankin, K. N., Wagner, J., et al. (2007). Investigations of the effects of gender, diunal variation, and age in human urinary metabolomic profiles. Analytical Chemistry, 79, 6995–7004.
Stella, C., Beckwith-Hall, B., Cloarec, O., et al. (2006a). Susceptibility of human metabolic phenotypes to dietary modulation. Journal of Proteome Research, 5, 2780–2788.
Stella, C., Beckwith-Hall, B., Cloarec, O., et al. (2006b). Susceptibility of human metabolic phenotypes to dietary modulation. Journal of Proteome Research, 5, 2780–2788.
Terentyeva, E. A., Hayakawa, K., Tanae, A., et al. (1997). Urinary biotinidase and alanine excretion in patients with insulin-dependent diabetes mellitus. European Journal of Clinical Chemistry and Clinical Biochemistry, 35, 21–24.
Vlcek, J., & Stemberk, V. (1990). Serum levels and urinary excretion of amino acids during high intensity physical exertion in healthy men. Casopís lékarů ceských, 129, 1141–1146.
Walsh, M. C., Brennan, L., Malthouse, J. P., Roche, H. M., & Gibney, M. J. (2006). Effect of acute dietary standardization on the urinary, plasma, and salivary metabolomic profiles of healthy humans. American Journal of Clinical Nutrition, 84, 531–539.
Walsh, M. C., Brennan, L., Pujos-Guillot, E., et al. (2007). Influence of acute phytochemical intake on human urinary metabolomic profiles. American Journal of Clinical Nutrition, 86, 1687–1693.
Winnike, J. H., Busby, M. G., Watkins, P. B., & O’Connell, T. M. (2009). Effects of a prolonged standardized diet on normalizing the human metabolome. American Journal of Clinical Nutrition, 90, 1496–1501.
Winning, H., Larsen, F. H., Bro, R., & Engelsen, S. B. (2008). Quantitative analysis of NMR spectra with chemometrics. Journal of Magnetic Resonance, 190, 26–32.
Winning, H., Roldan-Marin, E., Dragsted, L. O., et al. (2009). An exploratory NMR nutri-metabonomic investigation reveals dimethyl sulfone as a dietary biomarker for onion intake. Analyst, 134, 2344–2351.
Wishart, D. S., Tzur, D., Knox, C., et al. (2007). HMDB: the human metabolome database. Nucleic Acids Research, 35, D521–D526.
Wisselink, H. W., Weusthuis, R. A., Eggink, G., Hugenholtz, J., & Grobben, G. J. (2002). Mannitol production by lactic acid bacteria: a review. International Dairy Journal, 12, 151–161.
Yang, W. J., Wang, Y. W., Zhou, Q. F., & Tang, H. R. (2008). Analysis of human urine metabolites using SPE and NMR spectroscopy. Science in China Series B-Chemistry, 51, 218–225.
Zivkovic, A. M., & German, J. B. (2009). Metabolomics for assessment of nutritional status. Current Opinion in Clinical Nutrition and Metabolic Care, 12, 501–507.
Acknowledgments
The Danish Ministry of Food, Agriculture and Fisheries is acknowledged for sponsoring the project “Urinary biomarkers for eating patterns using NMR spectroscopy and Chemometrics” (3304-FVFP-060706-01) under which the present research has been developed and the Villum Kann Rasmussen project (Metabonomic Cancer Diagnostic) for continuing the funding for FS. LGR was supported by a research grant from the EU project DiOGenes and a research grant from the project BEST at the University of Copenhagen. LGR conducted the trial and FS conducted the 1H NMR analyses. FS and SBE conducted the chemometric analyses. SBE, FS and LGR were responsible for the interpretation of the results. LGR and FS authored the manuscript with co-authorship from TML, LOD, AA and SBE.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Lone G. Rasmussen, Francesco Savorani contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11306_2010_234_MOESM1_ESM.ppt
“Supplementary data”—online version Supplementary Figure 1. 500 MHz 1H NMR spectrum of human urine split in three regions: high-field, 0.8–2.95 ppm (A); mid-field, 2.95–4.1 ppm (B); low-field, 6.3–8.95 ppm (C). Vertical scale in A and C are respectively magnified 10 and 100 times, with respect to B. The spectrum is a mean of the samples from both study A and B. Assignments of the most significant peaks are numbered and the corresponding metabolites are listed in Supplementary Table 2. Supplementary Figure 2. Cohabitation effect for study B. PCA scores plot in which samples are colored according to couples of subjects living together (cohabitation). No significant clustering effect can be found confirming that standardization of the diet was effective on equalizing urinary metabolomes. Supplementary Figure 3. iECVA spectrum of the 1H NMR urinary data set for study B highlighting the marker alanine, responsible of diet culture differentiation (*): its spectral features are showed in the inset. Supplementary Figure 4. Gender discrimination for study A. A: PCA scores plot showing that the classification according to gender is achieved in PC#1 vs. PC#3. B: iECVA spectrum highlighting the markers responsible of gender differentiation (*): creatinine, citrate and alanine spectral features are illustrated in the above insets. Supplementary Figure 5. Gender discrimination for study B. PCA scores plot of the whole 1H NMR dataset in which samples are marked according to gender. No complete discrimination is achieved but a strong tendency towards gender separation can be found along both PC#1 and PC#5. (PPT 1336 kb)
Rights and permissions
About this article
Cite this article
Rasmussen, L.G., Savorani, F., Larsen, T.M. et al. Standardization of factors that influence human urine metabolomics. Metabolomics 7, 71–83 (2011). https://doi.org/10.1007/s11306-010-0234-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11306-010-0234-7