Abstract
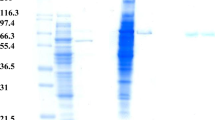
A novel 1,170 bp β-galactosidase gene sequence from Halomonas sp. S62 (BGalH) was identified through whole genome sequencing and was submitted to GenBank (Accession No. JQ337961). The BGalH gene was heterologously expressed in Escherichia coli BL21(DE3) cells, and the enzymatic properties of recombinant BGalH were studied. According to the polyacrylamide gel electrophoresis results and the sequence alignment analysis, BGalH is a dimeric protein and cannot be classified into one of the known β-galactosidase families (GH1, GH2, GH35, GH42). The optimal pH and temperature were determined to be 7.0 and 45 °C, respectively; the K m and K cat were 2.9 mM and 390.3 s−1, respectively, for the reaction with the substrate ortho-nitrophenyl-β-d-galactopyranoside. At 0–20 °C, BGalH exhibited 50–70 % activity relative to its activity under the optimal conditions. BGalH was stable over a wide range of pHs (6.0–8.5) after a 1 h incubation (>93 % relative activity) and was thermostable at 50 °C and below (>60 % relative activity). The enzyme hydrolyzes lactose completely in milk over 24 h at 7 °C. The characteristics of this novel β-galactosidase suggest that BGalH may be a good candidate for medical researches and food industry applications.





Similar content being viewed by others
References
Białkowska AM, Cieśliński H, Nowakowska KM, Kur J, Turkiewicz M (2009) A new β-galactosidase with a low temperature optimum isolated from the Antarctic Arthrobacter sp. 20B: gene cloning, purification and characterization. Arch Microbiol 191:825–835
Campbell AK, Waud JP, Matthews SB (2009) The molecular basis of lactose intolerance. Sci Prog 92:241–287
Cieśliński H, Kur J, Białkowska A, Baran I, Makowski K, Turkiewicz M (2005) Cloning, expression, and purification of a recombinant cold-adapted β-galactosidase from Antarctic bacterium Pseudoalteromonas sp. 22b. Protein Expr Purif 39:27–34
Coker JA, Brenchley JE (2006) Protein engineering of a cold-active β-galactosidase from Arthrobacter sp. SB to increase lactose hydrolysis reveals new sites affecting low temperature activity. Extremophiles 10:515–524
Coombs JM, Brenchley JE (1999) Biochemical and phylogenetic analyses of a cold-active β-galactosidase from the lactic acid bacterium Carnobacterium piscicola BA. Appl Environ Microbiol 65:5443–5450
Daabrowski S, Sobiewska G, Maciuńska J, Synowiecki J, Kur J (2000) Cloning, expression, and purification of the His6-tagged thermostable β-galactosidase from Pyrococcus woesei in Escherichia coli and some properties of the isolated enzyme. Protein Expr Purif 19:107–112
Feller G, Gerday C (2003) Psychrophilic enzymes: hot topics in cold adaptation. Nat Rev Microbiol 1:200–208
Fernandes S, Geueke B, Delgado O, Coleman J, Hatti-Kaul R (2002) β-galactosidase from a cold-adapted bacterium: purification, characterization and application for lactose hydrolysis. Appl Microbiol Biotechnol 58:313–321
Fujimoto H, Miyasato M, Ito Y, Sasaki T, Ajisaka K (1998) Purification and properties of recombinant β-galactosidase from Bacillus circulans. Glycoconj J 15:155–160
Gutshall KR, Trimbur DE, Kasmir JJ, Brenchley JE (1995) Analysis of a novel gene and β-Galactosidase isozyme from a psychrotrophic Arthrobacter isolate. J Bacteriol 177:1981–1988
Harrington LK, Mayberry JF (2008) A re-appraisal of lactose intolerance. Int J Clin Pract 62:1541–1546
Henrissat B, Bairoch A (1996) Updating the sequence-based classification of glycosyl hydrolases. Biochem J 316:695–696
Henrissat B, Davies G (1997) Structural and sequence-based classification of glycoside hydrolases. Curr Opin Struct Biol 7:637–644
Hildebrandt P, Wanarska M, Kur J (2009) A new cold-adapted β-D-galactosidase from the Antarctic Arthrobacter sp. 32c-gene cloning, overexpression, purification and properties. BMC Microbiol 9:151–161
Hoyoux A, Jennes I, Dubois P, Genicot S, Dubail F, François JM, Baise E, Feller G, Gerday C (2001) Cold-adapted β-galactosidase from the Antarctic psychrophile Pseudoalteromonas haloplanktis. Appl Environ Microbiol 67:1529–1535
Hu JM, Li H, Cao LX, Wu PC, Zhang CT, Sang SL, Zhang XY, Chen MJ, Lu JQ, Liu YH (2007) Molecular cloning and characterization of the gene encoding cold-active β-galactosidase from a psychrotrophic and halotolerant Planococcus sp. L4. J Agric Food Chem 55:2217–2224
Jacobson RH, Zhang XJ, DuBose RF, Matthews BW (1994) Three-dimensional structure of β-galactosidase from E.coli. Nature 369:761–766
Jeong JK, Kwon O, Lee YM, Oh DB, Lee JM, Kim S, Kim EH, Le TN, Rhee DK, Kang HA (2009) Characterization of the Streptococcus pneumoniae BgaC protein as a novel surface β-galactosidase with specific hydrolysis activity for the Galβ1-3GlcNAc moiety of oligosaccharides. J Bacteriol 191:3011–3023
Karasová-Lipovová P, Strnad H, Spiwok V, Malá Š, Králová B, Russell NJ (2003) The cloning, purification and characterisation of a cold-active β-galactosidase from the psychrotolerant Antarctic bacterium Arthrobacter sp. C2–2. Enzyme Microb Technol 33:836–844
Lam KS (2006) Discovery of novel metabolites from marine actinomycetes. Curr Opin Microbiol 9:245–251
Lee HS, Kwon KK, Kang SG, Cha SS, Kim SJ, Lee JH (2010) Approaches for novel enzyme discovery from marine environments. Curr Opin Microbio 21:353–357
Lu X, Liu X, Long C, Wang G, Gao Y, Liu J, Jiao B (2011) A preliminary study of the microbial resources and their biological activities of the East China Sea. Evid Based Complement Alternat Med 2011:806485–806492
Matthews BW (2005) The structure of E. coli β-galactosidase. C R Biol 328:549–556
Matthews SB, Waud JP, Roberts AG, Campbell AK (2005) Systemic lactose intolerance: a new perspective on an old problem. Postgrad Med J 81:167–173
Nakagawa T, Fujimoto Y, Ikehata R, Miyaji T, Tomizuka N (2006) Purification and molecular characterization of cold-active β-galactosidase from Arthrobacter psychrolactophilus strain F2. Appl Microbiol Biotechnol 72:720–725
Nakagawa T, Ikehata R, Myoda T, Miyaji T, Tomizuka N (2007) Overexpression and functional analysis of cold-active β-galactosidase from Arthrobacter psychrolactophilus strain F2. Protein Expr Purif 54:295–299
Sachadyn P, Stanislawska-Sachadyn A, Kur J (2007) A bifunctional chimeric protein consisting of MutS and beta-galactosidase. J Biotechnol 127:229–234
Sarkar S, Pramanik A, Mitra A, Mukherjee J (2010) Bioprocessing data for the production of marine enzymes. Mar Drugs 8:1323–1372
Sheridan PP, Brenchley JE (2000) Characterization of a salt-tolerant family 42 β-galactosidase from a psychrophilic Antarctic Planococcus Isolate. Appl Environ Microbiol 66:2438–2444
Sørensen HP, Porsgaard TK, Kahn RA, Stougaard P, Mortensen KK, Johnsen MG (2006) Secreted β-galactosidase from a Flavobacterium sp. isolated from a low-temperature environment. Appl Microbiol Biotechnol 70:548–557
Trimbur DE, Gutshall KR, Prema P, Brenchley JE (1994) Characterization of a psychrotrophic Arthrobacter gene and its cold-active β-galactosidase. Appl Environ Microbiol 60:4544–4552
Turkiewicz M, Kur J, Białkowska A, Cieśliński H, Kalinowska H, Bielecki S (2003) Antarctic marine bacterium Pseudoalteromonas sp. 22b as a source of cold-adapted β-galactosidase. Biomol Eng 20:317–324
Wierzbicka-Woś A, Cieśliński H, Wanarska M, Kozłowska-Tylingo K, Hildebrandt P, Kur J (2011) A novel cold-active β-D-galactosidase from the Paracoccus sp. 32d—gene cloning, purification and characterization. Microb Cell Fact 10:108–119
Acknowledgments
This work was supported by a National Hi-tech R&D Program grant to Dr. X. Liu (No. 2011AA09070302).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, Gx., Gao, Y., Hu, B. et al. A novel cold-adapted β-galactosidase isolated from Halomonas sp. S62: gene cloning, purification and enzymatic characterization. World J Microbiol Biotechnol 29, 1473–1480 (2013). https://doi.org/10.1007/s11274-013-1311-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-013-1311-7