Abstract
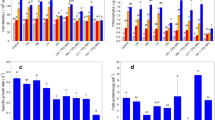
Nitrogen significantly regulated (p < 0.05) the effects of spiramycin on the growth and antioxidant responses of Microcystis aeruginosa as well as the biodegradation of spiramycin by M. aeruginosa during a 7-day exposure test. At a nitrogen level of 0.5 mg L−1, the activities of superoxide dismutase and catalase were stimulated by 100–400 ng L−1 of spiramycin to protect algal cells from oxidative damage, resulting in alleviated toxicity of spiramycin and low malondialdehyde content in M. aeruginosa. The catalase activity was inhibited by 400 ng L−1 of spiramycin at higher nitrogen levels of 5–50 mg L−1, leading to significant growth inhibition (p < 0.05) and higher malondialdehyde content through accumulation of hydrogen peroxide. Stimulated glutathione content and glutathione S-transferase activity were coupled to the biodegradation of spiramycin in M. aeruginosa. The 7-day biodegradation percentage of spiramycin varied from 8.9 to 29.6 %, which was enhanced by increased nitrogen concentration and decreased spiramycin concentration. Due to the regulation of algal growth, the toxicity of M. aeruginosa were significantly enhanced (p < 0.05) by 100 ng L−1 of spiramycin at a nitrogen concentration of 0.5 mg L−1 while significantly reduced (p < 0.05) by 400 ng L−1 of spiramycin at nitrogen levels of 5–50 mg L−1, according to the luminescent bacteria test. Low concentration of coexisting spiramycin contaminant should be considered during the control of M. aeruginosa bloom, especially under nitrogen deficient condition.





Similar content being viewed by others
References
Ahmed, A. (1968). Mechanism of inhibition of protein synthesis by spiramycin. Biochim Biophys Acta, 166(1), 205–217.
Alscher, R. G., Erturk, N., & Heath, L. S. (2002). Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. J Exp Bot, 53(372), 1331–1341.
Baptista, M. S., Stoichev, T., Basto, M. C. P., Vasconcelos, V. M., & Vasconcelos, M. T. S. D. (2009). Fate and effects of octylphenol in a Microcystis aeruginosa culture medium. Aquatic Toxicology, 92(2), 59–64.
Belchik, S. M., & Xun, L. (2011). S-glutathionyl-(chloro) hydroquinone reductases: a new class of glutathione transferases functioning as oxidoreductases. Drug Metab Rev, 43(2), 307–316.
Bhargava, P., Atri, N., Srivastava, A. K., & Rai, L. C. (2007). Cadmium mitigates ultraviolet-B stress in Anabaena doliolum: enzymatic and non-enzymatic antioxidants. Biol Plant, 51(3), 546–550.
Brisson-Noël, A., Trieu-Cuot, P., & Courvalin, P. J. (1988). Mechanism of action of spiramycin and other macrolides. J Antimicrob Chemother, 22(Suppl B), 13–23.
Churro, C., Fernandes, A. S., Alverca, E., Sam-Bento, F., Paulino, S., Figueira, V. C., Bento, A. J., Prabhakar, S., Lobo, A. M., Martins, L. L., Mourato, M. P., & Pereira, P. (2010). Effects of tryptamine on growth, ultrastructure, and oxidative stress of cyanobacteria and microalgae cultures. Hydrobiologia, 649(1), 195–206.
Downing, T. G., Sember, C. S., Gehringer, M. M., & Leukes, W. (2005). Medium N:P ratios and specific growth rate comodulate microcystin and protein content in Microcystis aeruginosa PCC7806 and M. aeruginosa UV027. Microb Ecol, 49(3), 468–473.
Elbaz, A., Wei, Y. Y., Meng, Q., Zheng, Q., & Yang, Z. M. (2010). Mercury-induced oxidative stress and impact on antioxidant enzymes in Chlamydomonas reinhardtii. Ecotoxicology, 19(7), 1285–1293.
Graham, J. L., Loetin, K. A., Meyer, M. T., & Ziegler, A. A. (2010). Cyanotoxin mixtures and taste-and-odor compounds in cyanobacteria blooms from the Midwestern United States. Environ Sci Technol, 44(19), 7361–7368.
Jiang, Y., Ji, B., Wong, R. N. S., & Wong, M. H. (2008). Statistical study on the effects of environmental factors on the growth and microcystins production of bloom-forming cyanobacterium-Microcystis aeruginosa. Harmful Algae, 7(2), 127–136.
Jin, Z. P., Luo, K., Zhang, S., Zheng, Q., & Yang, H. (2012). Bioaccumulation and catabolism of prometryne in green algae. Chemosphere, 87(3), 278–284.
Kong, Q. X., Zhu, L. Z., & Shen, X. Y. (2010). The toxicity of naphthalene to marine Chlorella vulgaris under different nutrient conditions. J Hazard Mater, 178(1–3), 282–286.
Liu, Y., Gao, B. Y., Yue, Q. Y., Guan, Y. T., Wang, Y., & Huang, L. H. (2012a). Influences of two antibiotic contaminants on the production, release and toxicity of microcystins. Ecotoxicol Environ Saf, 77, 79–87.
Liu, Y., Guan, Y. T., Gao, B. Y., & Yue, Q. Y. (2012b). Antioxidant responses and degradation of two antibiotic contaminants in Microcystis aeruginosa. Ecotoxicol Environ Saf, 86, 23–30.
Liu, Z. Q., Cui, F. Y., Ma, H., Fan, Z. Q., Zhao, Z. W., Hou, Z. L., & Liu, D. M. (2014). The transformation mechanism of nitrobenzene in the present of a species of cyanobacteria Microcystis aeruginosa. Chemosphere, 95, 234–240.
Ma, J. (2005). Differential sensitivity of three cyanobacterial and five green algal species to organotins and pyrethroids pesticides. Sci Total Environ, 341(1–3), 109–117.
Ni, L. X., Acharya, K., Hao, X. Y., & Li, S. Y. (2013). Antioxidant and metabolism responses to polyphenol stress in cyanobacterium Microcystis aeruginosa. J Environ Sci Health B, 48(2), 153–161.
Paerl, H. W., & Huisman, J. (2009). Climate change: a catalyst for global expansion of harmful cyanobacterial blooms. Environ Microbiol Rep, 1(1), 27–37.
Polyak, Y., Zaytseva, T., & Medvedeva, N. (2013). Response of toxic cyanobacterium Microcystis aeruginosa to environmental pollution. Water Air Soil Pollut, 224, 1494–1508.
Pradhan, S., & Rai, L. C. (2001). Copper removal by immobilized Microcystis aeruginosa in continuous flow columns at different bed heights: study of the adsorption/desorption cycle. World J Microbiol Biotechnol, 17(9), 829–832.
Qian, H. F., Li, J. J., Pan, X. J., Sun, Z. Q., Ye, C. B., Jin, G. Q., & Fu, Z. W. (2012). Effects of streptomycin on growth of algae Chlorella vulgaris and Microcystis aeruginosa. Environ Toxicol, 27(4), 229–237.
Riedel, G. F., Sanders, J. G., & Breitburg, D. L. (2003). Seasonal variability in response of estuarine phytoplankton communities to stress: linkage between toxic trace elements and nutrient enrichment. Estuaries, 26(2A), 323–338.
Rzymski, P., Poniedzialek, B., Niedzielski, P., Tabaczewski, P., & Wiktorowicz, K. (2014). Cadmium and lead toxicity and bioaccumulation in Microcystis aeruginosa. Front Environ Sci Eng, 8(3), 427–432.
Shao, J. H., Wu, Z. X., Yu, G. L., Peng, X., & Li, R. H. (2009). Allelopathic mechanism of pyrogallol to Microcystis aeruginosa PCC7806 (Cyanobacteria): From views of gene expression and antioxidant system. Chemosphere, 75(9), 924–928.
Stoichev, T., Baptista, M. S., Basto, M. C. P., Vasconcelos, V. M., & Vasconcelos, M. T. S. D. (2011). Effects of minocycline and its degradation products on the growth of Microcystis aeruginosa. Ecotoxicol Environ Saf, 74(3), 219–224.
Van der Grinten, E., Pikkemaat, M. G., Vanden Brandhof, E. J., Stroomberg, G. J., & Kraak, M. H. S. (2010). Comparing the sensitivity of algal, cyanobacterial and bacterial bioassays to different groups of antibiotics. Chemosphere, 80(1), 1–6.
Wang, J. X., & Xie, P. (2007). Antioxidant enzyme activities of Microcystis aeruginosa in response to nonylphenols and degradation of nonylphenols by M. aeruginosa. Environ Geochem Health, 29(5), 375–383.
Wang, Z. H., Luo, Z. X., & Yan, C. Z. (2013). Accumulation, transformation, and release of inorganic arsenic by the freshwater cyanobacterium Microcystis aeruginosa. Environ Sci Pollut Res, 20(10), 7286–7295.
Wright, G. D. (2005). Bacterial resistance to antibiotics: Enzymatic degradation and modification. Adv Drug Deliv Rev, 57(10), 1451–1470.
Wu, Z. X., Shi, J. Q., & Yang, S. Q. (2013). The effect of pyrogallic acid on growth, oxidative stress, and gene expression in Cylindrospermopsis raciborskii (Cyanobacteria). Ecotoxicology, 22(2), 271–278.
Xu, D. M., Li, C. D., Chen, H., & Shao, B. (2013). Cellular response of freshwater green algae to perfluorooctanoic acid toxicity. Ecotoxicol Environ Saf, 88, 103–107.
Yang, W. W., Tang, Z. P., Zhou, F. Q., Zhang, W. H., & Song, L. R. (2013). Toxicity studies of tetracycline on Microcystis aeruginosa and Selenastrum capricornutum. Environ Toxicol Pharmacol, 35(2), 320–324.
Zhang, S. L., Zhang, B., Dai, W., & Zhang, X. M. (2011). Oxidative damage and antioxidant responses in Microcystis aeruginosa exposed to the allelochemical berberine isolated from golden thread. J Plant Physiol, 168(7), 639–643.
Zhou, W. B., Juneau, P., & Qiu, B. S. (2006). Growth and photosynthetic responses of the bloom-forming cyanobacterium Microcystis aeruginosa to elevated levels of cadmium. Chemosphere, 65(10), 1738–1746.
Zhu, X. Z., Kong, H. L., Gao, Y. Z., Wu, M. F., & Kong, F. X. (2012). Low concentrations of polycyclic aromatic hydrocarbons promote the growth of Microcystis aeruginosa. J Hazard Mater, 237–238, 371–375.
Acknowledgments
This work was supported by National Natural Science Foundation of China (51209125) and partly by Promotive Research Foundation of Shandong Province (2013BSE27073).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, Y., Chen, S., Zhang, J. et al. Nitrogen-Regulated Interactions Between Microcystis aeruginosa and Spiramycin Contaminant. Water Air Soil Pollut 226, 135 (2015). https://doi.org/10.1007/s11270-015-2412-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-015-2412-8