Abstract
Stone pine (Pinus pinea L.) is a Mediterranean forest species used for the commercial harvesting of its edible seeds, known as pine nuts. Plant regeneration by somatic embryogenesis on semi-solid medium has been achieved in stone pine, but suspension culture systems for the large-scale production of somatic embryos need to be developed. Suspension cultures were established from several embryogenic lines. Growth parameters as settled cell volume (SCV) and dry weight (DW) in suspension cultures were influenced by genotype, initial inoculum density, orbiting speed and type of carbohydrate in the culture medium. A higher linear growth (DW) up to week four was obtained using 20 mg mL−1 inoculum. The orbiting speed affected the SCV as well as the morphology of embryogenic cultures. Growth parameters decreased when orbiting speed was increased from 50 rpm to 150 rpm. Cultures at 50 rpm most commonly grew as clusters of embryonal cell masses, whereas at 150 rpm there were large numbers of single cells. Cultures grew slowly in medium with maltose instead of sucrose, but growth was not affected when plant growth regulators (PGRs) concentration was decreased. A sustainable suspension culture could be maintained by transferring 250 mg of filtered cell mass to 25 mL of liquid medium at 3-week intervals. Culture scaling-up in liquid medium resulted in a 23-fold DW increase in 6 weeks, compared with 12 fold increase on semi-solid medium. Cotyledonary embryos were obtained on semi-solid maturation medium from lines proliferated in suspension cultures. Increased somatic embryo production was obtained when proliferation was performed at 100 rpm orbiting speed in liquid medium with low PGRs concentration and sucrose instead of maltose.




Similar content being viewed by others
References
Abrahamsson M, Valladares S, Merino I, Larsson E, von Arnold S (2017) Degeneration pattern in somatic embryos of Pinus sylvestris L. Vitro Cell Dev Biol—Plant 53:86–96. https://doi.org/10.1007/s11627-016-9797-y
Adams GW, Kunze HA, McCartney A, Millican S, Park YS (2016) An industrial perspective on the use of advanced reforestation stock technologies. In: Park YS, Bonga JM, Moon HK (eds) Vegetative propagation of forest trees. National Institute of Forest Science (NIFoS). Seoul, pp 323–334. https://www.iufro.org/science/divisions/division-2/20000/20900/20902/publications/ Online Edition:
Andrade GM, Nairn CJ, Le HT, Merkle SA (2009) Sexually mature transgenic American chestnut trees via embryogenic suspension-based transformation. Plant Cell Rep 28:1385–1397. https://doi.org/10.1007/s00299-009-0738-7
Aquea F. Poupin MJ, Matus JT, Gebauer M, Medina C, Arce-Johnson P (2008) Synthetic seed production from somatic embryos of Pinus radiata. Biotechnol Lett 30:1847–1852. https://doi.org/10.1007/s10529-008-9754-x
Astarita LV, Guerra MP (2000) Conditioning of the culture medium by suspension cells and formation of somatic proembryo in Araucaria angustifolia (Coniferae). In Vitro Cell Dev Biol—Plant 36:194–200. https://doi.org/10.1007/s11627-000-0036-0
Awan HUM, Pettenella D (2017) Pine Nuts: a review of recent sanitary conditions and market development. Forests 8(10):367. https://doi.org/10.3390/f8100367
Azevedo H, Dias A, Tavares RM (2008) Establishment and characterization of Pinus pinaster suspension cell cultures. Plant Cell Tiss Organ Cult 93:115–121. https://doi.org/10.1007/s11240-008-9349-1
Blanc G, Lardet L, Martin A, Jacob JL, Carron MP (2002) Differential carbohydrate metabolism conducts morphogenesis in embryogenic callus of Hevea brasiliensis (Müll. Arg.). J Exp Bot 53:1453–1462. https://doi.org/10.1093/jexbot/53.373.1453
Breton D, Harvengt L, Trontin JF, Bouvet A, Favre JM (2005) High subculture frequency, maltose-based and hormone-free medium sustained early development of somatic embryos in Maritime pine. In Vitro Cell Dev Biol—Plant 41:494–504. https://doi.org/10.1079/IVP2005671
Carneros E, Celestino C, Klimaszewska K, Park Y-S, Toribio M, Bonga JM (2009) Plant regeneration in Stone pine (Pinus pinea L.) by somatic embryogenesis. Plant Cell Tiss Organ Cult 98:165–178. https://doi.org/10.1007/s11240-009-9549-3
Carneros E, Hernández I, Toribio M, Díaz-Sala C, Celestino C (2017a) Effect of different cryoprotectant procedures on the recovery and maturation ability of cryopreserved Pinus pinea embryogenic lines of different ages. In Vitro Cell Dev Biol—Plant 53:469–477. https://doi.org/10.1007/s11627-017-9833-6
Carneros E, Toribio M, Celestino C (2017b) Effect of ABA, the auxin antagonist PCIB and partial desiccation on stone pine somatic embryo maturation. Plant Cell Tiss Organ Cult 131:445–458. https://doi.org/10.1007/s11240-017-1296-2
Choudhury H, Tandon P (2014) Non-destructive assessment of growth performance of embryogenic suspension culture of Pinus kesiya (Royle ex. Gord.) in shake-flask and self-designed bubble bioreactor and successful regeneration of plantlets from the culture systems. Proc Natl Acad Sci India Sect B Biol Sci 84:771–777. https://doi.org/10.1007/s40011-013-0253-z
Deb CR, Tandon P (2004) Establishment of an embryogenic suspension culture of Pinus kesiya (Khasi pine) from various explants. Indian J Biotech 3:445–448
dos Santos ALW, Silveira V, Steiner N, Maraschin M, Guerra MP (2010) Biochemical and morphological changes during the growth kinetics of Araucaria angustifolia suspension cultures. Braz Arch Biol Technol 53:497–504. https://doi.org/10.1590/S1516-89132010000300001
Ducos JP, Lambot C, Pétiard V (2007) Bioreactors for coffee mass propagation by somatic embryogenesis. Int J Plant Dev Biol 1:1–12.
Egertsdotter U, von Arnold S (1998) Development of somatic embryos in Norway spruce. J Exp Bot 49:155–162. https://doi.org/10.1093/jxb/49.319.155
Find JI, Kristensen MM, Nørgaard JV, Krogstrup P (1998a) Effect of culture period and cell density on regrowth following cryopreservation of embryogenic suspension cultures of Norway Spruce and Sitka spruce. Plant Cell Tiss Organ Cult 53:27–33. https://doi.org/10.1023/A:1006070729489
Find JI, Nørgaard JV, Krogstrup P (1998b) Growth parameters, nutrient uptake and maturation capacity of two cell-lines of Norway spruce (Picea abies) in suspension culture. J Plant Physiol 152:510–517. https://doi.org/10.1016/S0176-1617(98)80271-X
Finer JJ, Kriebel HB, Becwar MR (1989) Initiation of embryogenic callus and suspension cultures of eastern white pine (Pinus strobus L.). Plant Cell Rep 8:203–206. https://doi.org/10.1007/BF00778532
Gorbatenko O, Hakman I (2001) Desiccation-tolerant somatic embryos of Norway spruce (Picea abies) can be produced in liquid cultures and regenerated into plantlets. Int J Plant Sci 162:1211–1218. https://doi.org/10.1086/323416
Gupta PK (1996) Method for reproducing conifers by somatic embryogenesis using a maltose enriched maintenance medium. US Patent No 5,563,061, Washington, DC: U.S. Patent and Trademark Office
Gupta PK, Timmis R (2005) Mass propagation of conifer trees in liquid cultures–progress towards commercialization. Plant Cell Tiss Organ Cult 81:339–346. https://doi.org/10.1007/s11240-004-6654-1
Hargreaves C (2017) The development and application of conifer tissue culture and somatic embryogenesis protocols in New Zealand: The Pinus radiata D. Don story. In: Bonga JM, Park Y-S, Trontin J-F (eds). In Proc 4th Int Conf IUFRO Unit 2.09.02 on “Development and application of vegetative propagation technologies in plantation forestry to cope with a changing climate and environment”. September 19–23, 2016. La Plata, Argentina, pp 235–244. http://www.iufro.org/science/divisions/division-2/20000/20900/20902/publications/
Hřib J, Vooková B, Neděla V (2015) Imaging of native early embryogenic tissue of Scots pine (Pinus sylvestris L.) by ESEM. Open Life Sci 10:285–290. https://doi.org/10.1515/biol-2015-0028
Ibaraki Y, Kurata K (2001) Automation of somatic embryo production. Plant Cell Tiss Organ Cult 65:179–199. https://doi.org/10.1023/A:1010636525315
Ingram B, Mavituna F (2000) Effect of bioreactor configuration on the growth and maturation of Picea sitchensis somatic embryo cultures. Plant Cell Tiss Organ Cult 61:87–96. https://doi.org/10.1023/A:1006333907907
Iraqi D, Tremblay FM (2001) Analysis of carbohydrate metabolism enzymes and cellular contents of sugars and proteins during spruce somatic embryogenesis suggests a regulatory role of exogenous sucrose in embryo development. J Exp Bot 52:2301–2311. https://doi.org/10.1093/jexbot/52.365.2301
Jayawickrama KJ, McKeand SE, Jett JB (1997) Rootstock effects on scion growth and reproduction in 8-year-old grafted loblolly pine. Can J For Res 27:1781–1787. https://doi.org/10.1139/x97-152
Jiménez JA, Alonso-Blázquez N, López-Vela D, Celestino C, Toribio M, Alegre J (2011) Influence of culture vessel characteristics and agitation rate on gaseous exchange, hydrodynamic stress and growth of embryogenic cork oak (Quercus suber L.) cultures. In Vitro Cell Dev Biol—Plant 47:578–588. https://doi.org/10.1007/s11627-011-9399-7
Kaul K (1995) Somatic embryogenesis in eastern white pine (Pinus strobus L.). In: Jain S, Gupta P, Newton R (eds) Somatic embryogenesis in woody plants, vol 3. Kluwer Academic Publishers, Dordrecht, pp 257–268. https://doi.org/10.1007/978-94-011-0960-4_16
Klimaszewska K, Hargreaves C, Lelu-Walter M-A, Trontin J-F (2016) Advances in conifer somatic embryogenesis since year 2000. In: Germanà MA, Lambardi M (eds) In vitro embryogenesis in higher plants. Methods in molecular biology, vol 1359. Springer, New York, pp 131–166. https://doi.org/10.1007/978-1-4939-3061-6_7
Klöckner W, Büchs J (2012) Advances in shaking technologies. Trends Biotechnol 30:307–314. https://doi.org/10.1016/j.tibtech.2012.03.001
Krogstrup P (1990) Effect of culture densities on cell proliferation and regeneration from embryogenic cell suspensions of Picea sitchensis. Plant Sci 72:115–123. https://doi.org/10.1016/0168-9452(90)90193-R
Kubeš M, Drážná N, Konrádová H, Lipavská H (2014) Robust carbohydrate dynamics based on sucrose resynthesis in developing Norway spruce somatic embryos at variable sugar supply. In Vitro Cell Dev Biol—Plant 50:45–57. https://doi.org/10.1007/s11627-013-9589-6
Le-Feuvre R, Triviño C, Sabja AM, Bernier-Cardou M, Moynihan MR, Klimaszewska K (2013) Organic nitrogen composition of the tissue culture medium influences Agrobacterium tumefaciens growth and the recovery of transformed Pinus radiata embryonal masses after cocultivation. In Vitro Cell Dev Biol—Plant 49:30–40. https://doi.org/10.1007/s11627-013-9492-1
Lelu-Walter M-A, Thompson D, Harvengt L, Sanchez L, Toribio M, Pâques LE (2013) Somatic embryogenesis in forestry with a focus on Europe: state-of-the-art, benefits, challenges and future direction. Tree Genet Genomes 9:883–899. doi: s11295-013-0620-1
Lelu-Walter MA, Klimaszewska K, Miguel C, Aronen T, Hargreaves C, Teyssier C, Trontin JF (2016) Somatic embryogenesis for more effective breeding and deployment of improved varieties in Pinus spp.: bottlenecks and recent advances. In: Loyola-Vargas V, Ochoa-Alejo N (eds) Somatic embryogenesis: fundamental aspects and applications. Springer, Cham, pp 319–365. https://doi.org/10.1007/978-3-319-33705-0_19
Li XY, Huang FH, Murphy JB, Gbur EE (1998) Polyethylene glycol and maltose enhance somatic embryo maturation in loblolly pine (Pinus taeda L.). In Vitro Cell Dev Biol—Plant 34:22–26. https://doi.org/10.1007/BF02823118
Lipavská H, Konrádová H (2004) Somatic embryogenesis in conifers: the role of carbohydrate metabolism. In Vitro Cell Dev Biol—Plant 40:23–30. https://doi.org/10.1079/IVP2003482
Litvay JD, Verma DC, Johnson MA (1985) Influence of loblolly pine (Pinus taeda L.). culture medium and its components on growth and somatic embryogenesis of the wild carrot (Daucus carota L.). Plant Cell Rep 4:325–328. https://doi.org/10.1007/BF00269890
Mustafa NR, de Winter W, van Iren F, Verpoorte R (2011) Initiation, growth and cryopreservation of plant cell suspension cultures. Nat Protoc 6:715–742. https://doi.org/10.1038/nprot.2010.144
Mutke S, Gordo J, Gil L (2000) The stone pine (Pinus pinea L.) breeding programme in Castile-Leon (Central Spain). FAO—CIHEAM Nucis Newslett 9:50–55
Mutke S, Gordo J, Gil L (2005) Variability of Mediterranean Stone pine cone production: yield loss as response to climate change. Agric For Meteorol 132:263–272. https://doi.org/10.1016/j.agrformet.2005.08.002
Neděla V, Hřib J, Havel L, Hudec J, Runštuk J (2016) Imaging of Norway spruce early somatic embryos with the ESEM, Cryo-SEM and laser scanning microscope. Micron 84:67–71. https://doi.org/10.1016/j.micron.2016.02.011
Park Y-S (2014) Conifer somatic embryogenesis and multi-varietal forestry. In: Fenning T (ed) Challenges and opportunities for the world’s forests in the 21st century. Forests sciences, vol 81. Springer, Netherlands, pp 425–439. https://doi.org/10.1007/978-94-007-7076-8_17
Petrek J, Zitka O, Adam V, Bartusek K, Anjum NA, Pereira E, Havel L, Kizek R (2015) Are early somatic embryos of the Norway spruce (Picea abies (L.) Karst.). organised? PLoS ONE 10(12):e0144093. https://doi.org/10.1371/journal.pone.0144093
Pilarska M, Knox JP, Konieczny R (2013) Arabinogalactan-protein and pectin epitopes in relation to an extracellular matrix surface network and somatic embryogenesis and callogenesis in Trifolium nigrescens Viv. Plant Cell Tiss Organ Cult 115:35–44. https://doi.org/10.1007/s11240-013-0337-8
Preil W (2005) General introduction: a personal reflection on the use of liquid media for in vitro culture. In: Hvoslef-Eide AK, Preil W (eds) Liquid culture systems for in vitro plant propagation. Springer, Dordrecht, pp 1–18. https://doi.org/10.1007/1-4020-3200-5_1
Pullman GS, Skryabina A (2007) Liquid medium and liquid overlays improve embryogenic tissue initiation in conifers. Plant Cell Rep 26:873–887. https://doi.org/10.1007/s00299-006-0296-1
Salaj T, Blehova A, Salaj J (2007) Embryogenic suspension cultures of Pinus nigra Arn.: growth parameters and maturation ability. Acta Physiol Plant 29:225–231. https://doi.org/10.1007/s11738-007-0028-3
Silveira V, Floh EIS, Handro W, Guerra MP (2004) Effect of plant growth regulators on the cellular growth and levels of intracellular protein, starch and polyamines in embryogenic suspension cultures of Pinus taeda. Plant Cell Tiss Organ Cult 76:53–60. https://doi.org/10.1023/A:102584751
Sokal RR, Rohlf FJ (1969) Biometry. The principles and practice of statistics in biological research. W.H. Freeman, San Francisco
Strong WB (2006) Seasonal changes in seed reduction in lodgepole pine cones caused by feeding of Leptoglossus occidentalis (Hemiptera: Coreidae). Can Entomol 138:888–896. https://doi.org/10.4039/n05-092
Sun H, Aidun CK, Egertsdotter U (2010) Effects from shear stress on morphology and growth of early stages of Norway spruce somatic embryos. Biotechnol Bioeng 105:588–598. https://doi.org/10.1002/bit.22554
Sun H, Aidun CK, Egertsdotter U (2011) Possible effect from shear stress on maturation of somatic embryos of norway spruce (Picea abies). Biotechnol Bioeng 108:1089–1099. https://doi.org/10.1002/bit.23040
Tautorus TE, Lulsdorf MM, Kikcio SI, Dunstan DI (1992) Bioreactor culture of Picea mariana Mill. (black spruce) and the species complex Picea glauca-engelmannii (interior spruce) somatic embryos. Growth parameters. Appl Microbiol Biotechnol 38:46–51. https://doi.org/10.1007/BF00169417
Trontin JF, Teyssier C, Morel A, Harvengt L, Lelu-Walter MA (2016) Prospects for new variety deployment through somatic embryogenesis in maritime pine. In: Park Y-S, Bonga JM, Moon H-K (eds) Vegetative propagation of forest trees. National Institute of Forest Science (NIFoS), Seoul, pp 572–606. Online Edition: https://www.iufro.org/science/divisions/division-2/20000/20900/20902/publications/
Umehara M, Ogita S, Sasamoto H, Kamada H (2004) Inhibitory factor(s) of somatic embryogenesis regulated suspensor differentiation in suspension culture of Japanese larch (Larix leptolepis GORDON). Plant Biotechnol 21:87–94. https://doi.org/10.5511/plantbiotechnology.21.87
Warschefsky EJ, Klein LL, Frank MH, Chitwood DH, Londo JP, von Wettberg EJ, Miller AJ (2016) Rootstocks: diversity, domestication, and impacts on shoot phenotypes. Trends Plant Sci 21:418–437. https://doi.org/10.1016/j.tplants.2015.11.008
Acknowledgements
Funds were provided by the Spanish Ministry of Education and Science (AGL2010-22292-C03-01); the Madrid Regional R&D program S2009AMB-1668 (REGENFOR-CM) and IMIDRA grant to N. Gonzalez Cabrero. The participation of Dr. D. López-Vela and Dr. N. Alonso-Blázquez in some experiments is highly appreciated. Authors thank the comments and suggestions of two anonymous reviewers, which served to improve the manuscript. This work is part of the requirements to fulfill the N. Gonzalez-Cabrero Ph.D. degree.
Author information
Authors and Affiliations
Contributions
NG-C and MR-G participated in the experimental design, conducted experiments and helped to draft the manuscript. JA performed the statistical analyses and helped to draft the manuscript. MT wrote the final version of the manuscript. CC conceived the study, designed the experiments and drafted the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Maurizio Lambardi.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11240_2018_1467_MOESM1_ESM.pdf
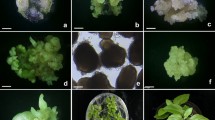
Supplementary material 1—Fig s1 Maturation and conversion into plants of somatic embryos from suspension cultures of Pinus pinea. a Maturing somatic embryos of 2F47 line cultured on 121 µM ABA-containing medium. Bar 1 mm. b Cotyledonary embryo after 12 weeks of culture. Bar 1 mm. c Germinating somatic embryos following 4 weeks on germination medium. Bar 1 cm. d Somatic seedlings growing in forest containers after 4 months of acclimatization in a mist chamber. Bar 5 cm (PDF 113 KB)
Rights and permissions
About this article
Cite this article
González-Cabrero, N., Ruiz-Galea, M., Alegre, J. et al. Growth, morphology and maturation ability of Pinus pinea embryogenic suspension cultures. Plant Cell Tiss Organ Cult 135, 331–346 (2018). https://doi.org/10.1007/s11240-018-1467-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-018-1467-9