Abstract
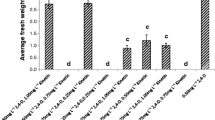
Isoflavonoids are naturally occurring polyphenolic compounds that have various biochemical and pharmacological properties. The tuberous roots of Pueraria candollei var. mirifica contain isoflavonoids and are widely used as dietary supplements and in therapeutics. The present study was aimed at producing reliable sources of isoflavonoids from single Pueraria cells and at optimizing a culture medium with increased biomass and isoflavonoid content. The medium optimized for prolonged maintenance of callus was MS medium supplemented with 200 mg/L KH2PO4, 1 mg/L thiamine HCl, 100 mg/L of myo-inositol with 0.2 mg/L 2,4-dichlorophenoxyacetic acid (2,4-D) and subculturing for 30 days for the highest growth. The amount of isoflavonoids varied among calli obtained from different explants. The highest amounts of puerarin and daidzein were quantified as 0.63 and 1.80 mg/g of dry weight (DW) in the examined suspension cell culture of single clones, whereas daidzein (0.50 mg/g) and genistein (0.03 mg/g) were present in the suspension cell culture obtained from a mixture of cells. The suspension of cell cultures from the same origin gave comparable amounts of isoflavonoids, but cells from different sources showed varied amounts of isoflavonoids. The amount of isoflavonoid also varied depending on the cell age. The present study provided the first evidence of Pueraria cell suspension from single cells and highlighted the importance of screening the starting cells from a single cell line and the cell age prior to the scale-up process to select desired sources of isoflavonoid compounds.






Similar content being viewed by others
Change history
03 January 2020
The Acknowledgements section in the original article was incomplete. The complete section is shown below. Acknowledgements This research was supported by Rachadapisek Sompote Fund for Postdoctoral Fellowship and for New Faculty Member Chulalongkorn University, Bangkok and Thailand Research Fund Project No. MRG5680090. TM was a recipient of the Research assistant scholarship from the Graduate School, Chulalongkorn University.
03 January 2020
The Acknowledgements section in the original article was incomplete. The complete section is shown below. Acknowledgements This research was supported by Rachadapisek Sompote Fund for Postdoctoral Fellowship and for New Faculty Member Chulalongkorn University, Bangkok and Thailand Research Fund Project No. MRG5680090. TM was a recipient of the Research assistant scholarship from the Graduate School, Chulalongkorn University.
Abbreviations
- BA:
-
6-benzylaminopurine
- NAA:
-
α-naphthaleneacetic acid
- TDZ:
-
Thidiazuron
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- MS:
-
Murashige and Skoog
- DW:
-
Dry weight
References
Abdollahi MR, Rashidi S (2017) Production and conversion of haploid embryos in chickpea (Cicer arietinum L.) anther cultures using high 2,4-D and silver nitrate containing media. Plant Cell Tiss Org Cult. https://doi.org/10.1007/s11240-017-1359-4
Bhojwani SS, Dantu PK (2013) Plant tissue culture: an introductory text. Springer, London. https://doi.org/10.1007/978-81-322-1026-9_3
Boonsnongcheep P, Korsangruang S, Soonthornchareonnon N, Chintapakorn Y, Saralamp P, Prathanturarug S (2010) Growth and isoflavonoid accumulation of Pueraria candollei var. candollei and P. candollei var. mirifica cell suspension cultures. Plant Cell Tiss Org Cult 101:119–126. https://doi.org/10.1007/s11240-010-9668-x
Cain JC (1960) Miroestrol: an oestrogen from the plant Pueraria mirifica. Nature 188:774–777
Cherdshewasart W, Subtang S, Dahlan W (2007) Major isoflavonoid contents of the phytoestrogen rich-herb Pueraria mirifica in comparison with Pueraria lobata. J Pharm Biomed Anal 43(2):428–434. https://doi.org/10.1016/j.jpba.2006.07.013
Davies KM, Deroles SC (2014) Prospects for the use of plant cell cultures in food biotechnology. Curr Opin Biotechnol 26:133–140. https://doi.org/10.1016/j.copbio.2013.12.010
Fidemann T, de Araujo Pereira GA, Heluy TR et al (2017) Handling culture medium composition for optimizing plant cell suspension culture in shake flasks. Plant Cell Tiss Organ Cult. https://doi.org/10.1007/s11240-017-1368-3
Karwasara VS, Dixit VK (2012) Culture medium optimization for improved puerarin production by cell suspension cultures of Pueraria tuberosa (Roxb. ex Willd.) DC. In Vitro Cell Dev Biol-Plant 48:189–199. https://doi.org/10.1007/s11627-011-9415-y
Karwasara VS, Dixit VK (2013) Culture medium optimization for camptothecin production in cell suspension cultures of Nothapodytes nimmoniana (J. Grah.) Mabberley. Plant Biotechnol Rep 7:357–369. https://doi.org/10.1007/s11816-012-0270-z
Malaivijitnond S (2012) Medical applications of phytoestrogens from the Thai herb Pueraria mirifica. Front Med 6(1):8–21. https://doi.org/10.1007/s11684-012-0184-8
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Murthy HN, Lee EJ, Paek KY (2014) Production of secondary metabolites from cell and organ cultures: strategies and approaches for biomass improvement and metabolite accumulation. Plant Cell Tiss Organ Cult. https://doi.org/10.1007/s11240-014-0467-7
Nagella P, Murthy HN (2011) Effects of macroelements and nitrogen source on biomass accumulation and withanolide—a production from cell suspension cultures of Withania somnifera (L.) Dunal. Plant Cell Tiss Org Cult 104:119–124. https://doi.org/10.1007/s11240-010-9799-0
Narayani M, Chadha A, Srivastava S (2017) Callus and cell suspension culture of Viola odorata as in vitro production platforms of known and novel cyclotides. Plant Cell Tiss Organ Cult 130:289–299. https://doi.org/10.1007/s11240-017-1223-6
Nebe B, Peters A, Duske K, Richter DU, Briese V (2006) Influence of phytoestrogens on the proliferation and expression of adhesion receptors in human mammary epithelial cells in vitro. Eur J Cancer Prev 15:405–415
Pope GS, Grundy HM, Jones HEH, Tait SAS (1958) The estrogenic substance (miroestrol) from the tuberous roots of Pueraria mirifica. J Endocrinol 17(1):15–16
Rao SR, Ravishankar GA (2002) Plant cell cultures: chemical factories of secondary metabolites. Biotechnol Adv 20:10–153. https://doi.org/10.1016/S0734-9750(02)00007-1
Schabath MB, Hernandez LM, Wu X, Pillow PC, Spitz MR (2005) Dietary phytoestrogens and lung cancer risk. JAMA 294:1493–1504. https://doi.org/10.1001/jama.294.12.1493
Sepehr MF, Ghorbanli M (2002) Effects of nutritional factors on the formation of Anthraquinones in callus cultures of Rheum ribes. Plant Cell Tissue Org Cult 68:171–175. https://doi.org/10.1023/A:1013837232047
Suntara A (1931) The remedy pamphlet of Kwao Krua tuber of luang anusarnsuntarakromkarnphiset. Chiang Mai Upatipongsa Press, Chiang Mai
Thanonkeo S, Panichajakul S (2006) Production of isofalvones, daidzein and genistein in callus cultures of Pueraria candollei Wall. ex Benth. var. mirifica. Songklanakarin J Sci Technol 28:45–53
Udomsuk L, Jarukamjorn K, Tanaka H, Putalun W (2009) Production of isoflavonoids in callus cultures of Pueraria candollei var. mirifica. Z Naturforsch C 64:239–243
Udomsuk L, Juengwattanatrakul T, Jarukamjorn K, Putalun W (2012) Increased miroestrol, deoxymiroestrol and isoflavonoid accumulation in callus and cell suspension cultures of Pueraria candollei var. mirifica. Acta Physiol Plant 34:1093–1100. https://doi.org/10.1007/s11738-011-0906-6
Van der Maesen LJG (2002) Pueraria: botanical characteristics. In: Keung WM (ed) Pueraria: the genus Pueraria. Taylor & Francis, London, pp 1–28
Wanadorn W (1933) A reputed rejuvenator. J Siam Soc Nat His 9:145–147 (in Thai)
Wilson SA, Roberts SC (2012) Recent advances towards development and commercialization of plant cell culture processes for the synthesis of biomolecules. Plant Biotechnol J 10:249–268. https://doi.org/10.1111/j.1467-7652.2011.00664.x
Zhou X, Zhong J-J (2010) Plant cell culture, secondary product accumulation. In: Flickinger MC (ed) Encyclopedia of industrial bio- technology: bioprocess, bioseparation, and cell technology. Wiley, Hoboken, pp 1–29
Acknowledgements
This research was supported by Rachadapisek Sompote Fund for Postdoctoral Fellowship and for New Faculty Member, Chulalongkorn University, Bangkok and Thailand Research Fund Project No. MRG5680090.
Author information
Authors and Affiliations
Contributions
DR performed the experiments and wrote the manuscript. TM and KK helped in the experiments. SV designed the experiment and wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Sergio J. Ochatt.
Rights and permissions
About this article
Cite this article
Rani, D., Meelaph, T., Kobtrakul, K. et al. Optimizing Pueraria candollei var. mirifica cell suspension culture for prolonged maintenance and decreased variation of isoflavonoid from single cell lines. Plant Cell Tiss Organ Cult 134, 433–443 (2018). https://doi.org/10.1007/s11240-018-1433-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-018-1433-6