Abstract
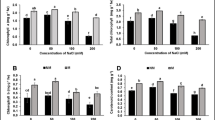
Atractylodes lancea is a traditional Chinese herb, and its quality depends on the main active compounds sesquiterpenoids. In recent years, guaranteeing the quality and yield of A. lancea has become problematic. Our previous results have shown that the endophytic fungus AL12 (Gilmaniella sp.) promotes plant growth and sesquiterpenoid biosynthesis of A. lancea. However, little has been done to uncover the mechanism of this “double-promotion”. In this study, plant biomass and sesquiterpenoid content were analyzed to verify this “double-promotion” effect of AL12 on A. lancea. Physiological tests (such as root viability, element content, rubisco content, and some phytohormones levels) were performed to try to identify the mechanism of improved plant growth. The results showed that growth promotion effect induced by AL12 on A. lancea may depend on increased nutrient content, improved photosynthesis and enhanced cytokinin signaling. Secondary metabolite biosynthesis consumes substance and energy during plant growth. Transcript analysis indicated that AL12 stimulates sesquiterpenoid biosynthesis through the MVA pathway. Physiological tests (such as sesquiterpenoid biosynthesis precursors and organic acids of tricarboxylic acid cycle) were performed to try to identify the mechanism of improved sesquiterpenoid accumulation. The results showed that AL12 improves the primary metabolism of A. lancea including photosynthesis, glycolysis, and the TCA cycle, thus providing sufficient carbon and energy for sesquiterpenoid biosynthesis. To summarize, this work uncover the primary mechanism of AL12 promotion of plant growth and sesquiterpenoid accumulation in A. lancea. This study provides a theoretical basis for obtaining higher yield and quality medicinal plants and also provides a framework to understand plant–endophyte interactions.






Similar content being viewed by others
Abbreviations
- AMF:
-
Arbuscular mycorrhizal fungi
- A. lancea :
-
Atractylodes lancea
- AL12:
-
Endophytic fungus Gilmaniella sp. AL12
- NO:
-
Nitric oxide
- H2O2 :
-
Hydrogen peroxide
- SA:
-
Salicylic acid
- JA:
-
Jasmonic acid
- Br:
-
Brassinosteroid
- Ca2+ :
-
Calcium
- MVA:
-
Mevalonate
- MS medium:
-
Murashige and Skoog medium
- NAA:
-
Naphthaleneacetic acid
- 6-BA:
-
6-Benzyladenine
- PDA:
-
Potato dextrose agar
- GC:
-
Gas chromatograph
- MS:
-
Mass spectrometer
- TTC:
-
Triphenyltetrazolium chloride
- TFA:
-
Trichloroacetic acid
- PVPP:
-
Polyvinylpolypyrrolidone
- HPLC:
-
High-performance liquid chromatography
- PVP:
-
Polyvinylpyrrolidone
- BHT:
-
Butylated hydroxytoluene
- 2-OG:
-
2-Oxpentanedioic acid
- IAA:
-
Indole-3-acetic acid
- GA:
-
Gibberellin
- ABA:
-
Abscisic acid
- CTK:
-
Cytokinin
- RT-qPCR:
-
Real-time quantitative PCR
- EF1α :
-
Elongation factor 1 alpha
- C t :
-
Cycle threshold
- SD:
-
Standard deviations
- MEP:
-
2-C-methyl-d-erythritol-4-phosphate
- HMGR :
-
3-Hydroxy-3-methylgutaryl coenzyme A reductase
- DXR :
-
1-Deoxy-d-xylulose 5-phosphate reductoisomerase
- TCA cycle:
-
Tricarboxylic acid cycle
- ATP:
-
Triphosadenine
- ETH:
-
Ethylene
References
Bonfante P, Genre A (2010) Mechanisms underlying beneficial plant–fungus interactions in mycorrhizal symbiosis. Nat Commun 1:48
Cortleven A, Schmulling T (2015) Regulation of chloroplast development and function by cytokinin. J Exp Bot 66:4999–5013
de Kraker JW, Franssen MC, Joerink M, de Groot A, Bouwmeester HJ (2002) Biosynthesis of costunolide, dihydrocostunolide, and leucodin. Demonstration of cytochrome p450-catalyzed formation of the lactone ring present in sesquiterpene lactones of chicory. Plant Physiol 129:257–268
Duncan DR, Widholm JM (2004) Osmotic induced stimulation of the reduction of the viability dye 2,3,5-triphenyltetrazolium chloride by maize roots and callus cultures. J Plant Physiol 161:397–403
Eljounaidi K et al (2014) Cytochrome P450s from Cynara cardunculus L. CYP71AV9 and CYP71BL5, catalyze distinct hydroxylations in the sesquiterpene lactone biosynthetic pathway. Plant Sci 223:59–68
Fernie AR, Carrari F, Sweetlove LJ (2004) Respiratory metabolism: glycolysis, the TCA cycle and mitochondrial electron transport. Curr Opin Plant Biol 7:254–261
Govindarajulu M et al (2005) Nitrogen transfer in the Arbuscular mycorrhizal symbiosis. Nature 435:819–823
Gutjahr C (2014) Phytohormone signaling in Arbuscular mycorhiza development. Curr Opin Plant Biol 20:26–34
Huang C et al (2015) Rubisco accumulation is important for the greening of the fln2-4 mutant in Arabidopsis. Plant Sci 236:185–194
Jasim B, Geethu PR, Mathew J, Radhakrishnan EK (2015) Effect of endophytic Bacillus sp. from selected medicinal plants on growth promotion and diosgenin production in Trigonella foenum-graecum. Plant Cell Tissue Organ Cult 122:565–572
Kim DS, Hwang BK (2014) An important role of the pepper phenylalanine ammonia-lyase gene (PAL1) in salicylic acid-dependent signalling of the defence response to microbial pathogens. J Exp Bot 65:2295–2306
Kurepin LV, Park JM, Lazarovits G, Bernards MA (2014) Burkholderia phytofirmans-induced shoot and root growth promotion is associated with endogenous changes in plant growth hormone levels. Plant Growth Regul 75:199–207
Kwon YS et al (2010) Proteome analysis of Arabidopsis seedlings exposed to bacterial volatiles. Planta 232:1355–1370
Liu Y, Wang Q, Ding Y, Li G, Xu J, Wang S (2011) Effects of external ABA, GA3 and NAA on the tiller bud outgrowth of rice is related to changes in endogenous hormones. Plant Growth Regul 65:247–254
Ludwig-Muller J (2015) Plants and endophytes: equal partners in secondary metabolite production? Biotechnol Lett 37:1325–1334
Mandal S, Evelin H, Giri B, Singh VP, Kapoor R (2013) Arbuscular mycorrhiza enhances the production of stevioside and rebaudioside-A in Stevia rebaudiana via nutritional and non-nutritional mechanisms. Appl Soil Ecol 72:187–194
Misawa N (2011) Pathway engineering for functional isoprenoids. Curr Opin Biotechnol 22:627–633
Nagegowda DA (2010) Plant volatile terpenoid metabolism: biosynthetic genes, transcriptional regulation and subcellular compartmentation. FEBS Lett 584:2965–2973
Proietti S, Bertini L, Timperio AM, Zolla L, Caporale C, Caruso C (2013) Crosstalk between salicylic acid and jasmonate in Arabidopsis investigated by an integrated proteomic and transcriptomic approach. Mol BioSyst 9:1169–1187
Raj D, Kokotkiewicz A, Drys A, Luczkiewicz M (2015) Effect of plant growth regulators on the accumulation of indolizidine alkaloids in Securinega suffruticosa callus cultures. Plant Cell Tissue Organ Cult 123:39–45
Ren CG, Dai CC (2012) Jasmonic acid is involved in the signaling pathway for fungal endophyte-induced volatile oil accumulation of Atractylodes lancea plantlets. BMC Plant Biol 12:128
Ren CG, Dai CC (2013) Nitric oxide and brassinosteroids mediated fungal endophyte-induced volatile oil production through protein phosphorylation pathways in Atractylodes lancea plantlets. J Integr Plant Biol 55:1136–1146
Ren C-G, Chen Y, Dai C-C (2013) Cross-Talk between calcium–calmodulin and brassinolide for fungal endophyte-induced volatile oil accumulation of Atractylodes lancea plantlets. J Plant Growth Regul 33:285–294
Straub D, Yang H, Liu Y, Tsap T, Ludewig U (2013) Root ethylene signalling is involved in Miscanthus sinensis growth promotion by the bacterial endophyte Herbaspirillum frisingense GSF30(T). J Exp Bot 64:4603–4615
Sweetlove LJ, Beard KF, Nunes-Nesi A, Fernie AR, Ratcliffe RG (2010) Not just a circle: flux modes in the plant TCA cycle. Trends Plant Sci 15:462–470
Vasconsuelo A, Boland R (2007) Molecular aspects of the early stages of elicitation of secondary metabolites in plants. Plant Sci 172:861–875
Wang HX, Liu CM, Liu Q, Gao K (2008) Three types of sesquiterpenes from rhizomes of Atractylodes lancea. Phytochemistry 69:2088–2094
Wang Y, Dai C-C, Zhao Y-W, Peng Y (2011) Fungal endophyte-induced volatile oil accumulation in Atractylodes lancea plantlets is mediated by nitric oxide, salicylic acid and hydrogen peroxide. Process Biochem 46:730–735
Wang Y, Dai CC, Cao JL, Xu DS (2012) Comparison of the effects of fungal endophyte Gilmaniella sp. and its elicitor on Atractylodes lancea plantlets. World J Microb Biot 28:575–584
Wang HW, Zhang W, Su CL, Zhu H, Dai CC (2015a) Biodegradation of the phytoestrogen luteolin by the endophytic fungus Phomopsis liquidambari. Biodegradation 26:197–210
Wang XM, Yang B, Ren CG, Wang HW, Wang JY, Dai CC (2015b) Involvement of abscisic acid and salicylic acid in signal cascade regulating bacterial endophyte-induced volatile oil biosynthesis in plantlets of Atractylodes lancea. Physiol Plant 153:30–42
Wangchuk P, Tobgay T (2015) Contributions of medicinal plants to the gross national happiness and biodiscovery in bhutan. J Ethnobiol Ethnomed 11:48
Watts-Williams SJ, Jakobsen I, Cavagnaro TR, Gronlund M (2015) Local and distal effects of Arbuscular mycorrhizal colonization on direct pathway Pi uptake and root growth in Medicago truncatula. J Exp Bot 66:4061–4073
Wu BH, Genard M, Lescourret F, Gomez L, Li SH (2002) Influence of assimilate and water supply on seasonal variation of acids in peach (cv Suncrest). J Sci Food Agric 82:1829–1836
Yang Y, Wu G (2013) Determination of acetyl coenzyme A in grape berries by high performance liquid chromatography-tandem mass spectrometry. Chin J Chromatogr 31:33–37
Yang CQ, Fang X, Wu XM, Mao YB, Wang LJ, Chen XY (2012a) Transcriptional regulation of plant secondary metabolism. J Integr Plant Biol 54:703–712
Yang X, Zhang X, Yuan D, Jin F, Zhang Y, Xu J (2012b) Transcript profiling reveals complex auxin signalling pathway and transcription regulation involved in dedifferentiation and redifferentiation during somatic embryogenesis in cotton. BMC Plant Biol 12:110
Zhang N, Wang D, Liu Y, Li S, Shen Q, Zhang R (2013) Effects of different plant root exudates and their organic acid components on chemotaxis, biofilm formation and colonization by beneficial rhizosphere-associated bacterial strains. Plant Soil 374:689–700
Zhu XQ, Wang CY, Chen H, Tang M (2014) Effects of Arbuscular mycorrhizal fungi on photosynthesis, carbon content, and calorific value of black locust seedlings. Photosynthetica 52:247–252
Acknowledgments
Financial support was provided by the National Natural Science Foundation of China (Grant No. 31070443), a project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions, Promoting Project for Industrialization of Jiangsu Higher Education Institutions (Grant No. JHB2012-16), and Nanjing Produce-Learn-Research Project (Grant No. 201306019).
Authors’ contribution
J.Y. designed and carried out most of the experiments. J.Y.Z. helped for metabolite determination and GC–MS analysis. X.L. helped for net photosynthesis rate measurement. J.Y. analyzed data and wrote the manuscript. C.C.D. supervised the work and interpreted data with J.Y. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest and the publication of the work has been approved by all co-authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yuan, J., Zhou, JY., Li, X. et al. The primary mechanism of endophytic fungus Gilmaniella sp. AL12 promotion of plant growth and sesquiterpenoid accumulation in Atractylodes lancea . Plant Cell Tiss Organ Cult 125, 571–584 (2016). https://doi.org/10.1007/s11240-016-0971-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-016-0971-z