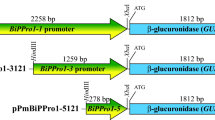
Abstract
Tissue-specific promoters for efficient expression of transgenes at specific times or in specific plant tissues can be applied to develop new transgenic plants. To exploit a promoter capable of driving strong expression in potato tubers, we isolated the promoter region of the laccase gene from potato (Solanum tuberosum L. cv. Desiree) and characterized its activity in transgenic Solanaceae plants, such as potato, tobacco and tomato. The ability of the laccase promoter to induce the β-glucuronidase (GUS) reporter was evaluated in independent transgenic potato lines and compared with that of the constitutive CaMV35S promoter. To determine the tissue specificity of expression in transgenic potato, GUS levels in shoot tips, leaves, stems, roots and tubers were measured by histochemical analysis. The laccase promoter conferred tuber-specific expression in transgenic potato regardless of the developmental stage, and there was no GUS reporter expression in leaves, stems or roots. Serial 5′ deletion analysis of the laccase promoter revealed that the tuber-specific regulatory elements might be scattered throughout the promoter. The laccase promoter responded weakly to salt stress, mannitol stress, and mechanical wounding but not to cold stress in the leaves and stems of transgenic potato. In transgenic tobacco, weak GUS expression driven by the laccase promoter was detected throughout the entire plant, whereas in transgenic tomato, GUS expression was detected only in the roots and seeds. Our data show that the laccase promoter represents a feasible candidate to drive high and preferential expression of genes in potato tubers.






Similar content being viewed by others
References
Balazs E, Bouzoubaa S, Guilley H, Jonard G, Paszkowski J, Richards K (1985) Chimeric vector construction for higher-plant transformation. Gene 40:343–348
Bansal A, Kumari V, Taneja D, Sayal R, Das N (2012) Molecular cloning and characterization of granule-bound starch synthase I (GBSSI) alleles from potato and sequence analysis for detection of cis-regulatory motifs. Plant Cell, Tissue Organ Cult 109:247–261. doi:10.1007/s11240-011-0090-9
Bernards MA, Fleming WD, Llewellyn DB, Priefer R, Yang X, Sabatino A, Plourde GL (1999) Biochemical characterization of the suberization-associated anionic peroxidase of potato. Plant Physiol 121:135–146. doi:10.1104/pp.121.1.135
Bhat DS, Anjanasree KN, Lenka SK, Bansal KC (2010) Isolation and characterization of fruit-specific promoters ACS4 and EXP1 from tomato (Solanum lycopersicum L). J Plant Biochem Biotechnol 19:51–57. doi:10.1007/BF03323435
Caparrós-Ruiz D, Fornalé S, Civardi L, Puigdomènech P, Rigau J (2006) Isolation and characterisation of a family of laccases in maize. Plant Sci 171:217–225. doi:10.1016/j.plantsci.2006.03.007
Chatterjee M, Banerjee AK, Hannapel DJ (2007) A BELL1-like gene of potato is light activated and wound inducible. Plant Physiol 145(4):1435–1443. doi:10.1104/pp.107.105924
Covey SN, Lomonossoff GP, Hull R (1981) Characterisation of cauliflower mosaic virus DNA sequences which encode major polyadenylated transcripts. Nucl Acid Res 9:6735–6748. doi:10.1093/nar/9.24.6735
de Marco A, Roubelakis-Angelakis KA (1997) Laccase activity could contribute to cell-wall reconstitution in regenerating protoplasts. Phytochemistry 46:421–425. doi:10.1016/S0031-9422(97)00301-4
Deikman J, Kline R, Fischer RL (1992) Organization of ripening and ethylene regulatory regions in a fruit-specific promoter from tomato (Lycopersicon esculentum). Plant Physiol 100:2013–2017. doi:10.1104/pp.100.4.2013
Diaz-De-Leon F, Klotz KL, Lagrimini LM (1993) Nucleotide sequence of the tobacco (Nicotiana tabacum) anionic peroxidase gene. Plant Physiol 101:1117–1118. doi:10.1104/pp.101.3.1117
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh tissue. Phytochem Bull 19:11–15
Driouich A, Lainé AC, Vian B, Faye L (1992) Characterization and localization of laccase forms in stem and cell cultures of sycamore. Plant J 2:13–24. doi:10.1111/j.1365-313X.1992.00013.x
Engel ML, Holmes-Davis R, McCormick S (2005) Green sperm. Identification of male gamete promoters in Arabidopsis. Plant Physiol 138(4):2124–2133. doi:10.1104/pp.104.054213
Fu H, Kim SY, Park WD (1995) High-level tuber expression and sucrose inducibility of a potato Sus4 sucrose synthase gene require 5′ and 3′ flanking sequences and the leader intron. Plant Cell 7:1387–1394. doi:10.1105/tpc.7.9.1387
Gallusci P, Salamini F, Thompson R (1994) Differences in cell type-specific expression of the gene Opaque 2 in maize and transgenic tobacco. Mol Gen Genet 244:391–400. doi:10.1007/BF00286691
Gavnholt B, Larsen K (2002) Molecular biology of plant laccases in relation to lignin formation. Physiol Plant 116:273–280. doi:10.1034/j.1399-3054.2002.1160301
Gavnholt B, Larsen K, Rasmussen SK (2002) Isolation and characterisation of laccase cDNAs from meristematic and stem tissues of ryegrass (Lolium perenne). Plant Sci 162:873–885
Gittins JR, Pellny TK, Hiles ER, Rosa C, Biricolti S, James DJ (2000) Transgene expression driven by heterologous ribulose-1, 5-bisphosphate carboxylase/oxygenase small-subunit gene promoters in the vegetative tissues of apple (Malus pumila Mill.). Planta 210:232–240. doi:10.1007/PL00008130
Gowik U, Burscheidt J, Akyildiz M, Schlue U, Koczor M, Streubel M, Westhoff P (2004) Cis-regulatory elements for mesophyll-specific gene expression in the C4 plant Flaveria trinervia, the promoter of the C4 phosphoenolpyruvate carboxylase gene. Plant Cell 16:1077–1090. doi:10.1105/tpc.060510
Grierson C, Du JS, Zabala MT, Beggs K, Goldsworth M, Bevan M (1994) Separate cis sequences and trans factors direct metabolic and developmental regulation of a potato tuber storage protein gene. Plant J 5:815–826
Guillet C, Aboul-Soud MA, Le Menn A, Viron N, Pribat A, Germain V, Just D, Baldet P, Rousselle P, Lemaire-Chamley M, Rothan C (2012) Regulation of the fruit-specific PEP carboxylase SIPPC2 promoter at early stages of tomato fruit development. PLoS ONE 7:e36795. doi:10.1371/journal.pone.0036795
Higo K, Ugawa Y, Iwamoto M, Korenaga T (1999) Plant cis-acting regulatory DNA elements (PLACE) database:1999. Nucleic Acids Res 27(1):297–300. doi:10.1093/nar/27.1.297
Hoegger PJ, Kilaru S, James TY, Thacker JR, Kües U (2006) Phylogenetic comparison and classification of laccase and related multicopper oxidase protein sequences. FEBS J 273:2308–2326. doi:10.1111/j.1742-4658.2006.05247.x
Horsch RB, Fry JE, Hoffmann NL, Eichholtz D, Rogers SG, Fraley RT (1985) A simple and general method for transferring genes into plants. Science 227:1229–1231. doi:10.1126/science.227.4691.1229
Jefferson JA (1987) Assaying chimeric genes in plants: the GUS gene function system. Plant Mol Biol Rep 5:387–405
Jefferson R, Goldsbrough A, Bevan M (1990) Transcriptional regulation of a patatin-1 gene in potato. Plant Mol Biol 14:995–1006
Kiefer-Meyer MC, Gomord V, O’Connell A, Halpin C, Faye L (1996) Cloning and sequence analysis of laccase-encoding cDNA clones from tobacco. Gene 178:205–207
Kim SY, May GD, Park WD (1994) Nuclear protein factors binding to a class I patatin promoter region are tuber-specific and sucrose-inducible. Plant Mol Biol 26:603–615
Kim HS, Euym JW, Kim MS, Lee BC, Mook-Jung I, Jeon JH, Joung H (2003) Expression of human amyloid-beta peptide in transgenic potato. Plant Sci 165:1445–1451. doi:10.1016/j.plantsci.2003.07.007
Kim HS, Youm JW, Moon KB, Ha JH, Kim YH, Joung H, Jeon JH (2012) Expression analysis of human β-secretase in transgenic tomato fruits. Protein Exp Purif 82:125–131. doi:10.1016/j.pep.2011.11.012
Kobayashi H, Oikawa Y, Koiwa H, Yamamura S (1998) Flower-specific gene expression directed by the promoter of a chalcone synthase gene from Gentiana triflora in Petunia hybrida. Plant Sci 131:173–180. doi:10.1016/S0168-9452(97)00236-7
Korth KL, Stermer BA, Bhattacharyya MK, Dixon RA (1997) HMG-CoA reductase gene families that differentially accumulate transcripts in potato tubers are developmentally expressed in floral tissues. Plant Mol Biol 33:545–551. doi:10.1023/A:1005743011651
LaFayette PR, Eriksson KEL, Dean JFD (1995) Nucleotide sequence of a cDNA clone encoding an acidic laccase from sycamore maple (Acer pseudoplatanus L.). Plant Physiol 107:667–668. doi:10.1104/pp.107.2.667
LaFayette PR, Eriksson KEL, Dean JFD (1999) Characterization and heterologous expression of laccase cDNAs from xylem tissues of yellow-poplar (Liriodendron tulipifera). Plant Mol Biol 40:23–35. doi:10.1023/A:1026437406859
Lee BC (2004) A study on cloning and expression pattern of genes related in suberin biosynthesis of potato (Solanum tuberosum L.). Dissertation, Kongju National University
Liang M, Haroldsen V, Cai X, Wu Y (2006) Expression of a putative laccase gene, ZmLAC1, in maize primary roots under stress. Plant, Cell Environ 29:746–753. doi:10.1111/j.1365-3040.2005.01435.x
Liu X, Prat S, Willmitzer L, Frommer W (1990) Cis regulatory elements directing tuber-specific and sucrose-inducible expression of a chimeric class I patatin promoter/GUS-gene fusion. Mol Gen Genet 223:401–406. doi:10.1007/BF00264446
Liu Y, Lou Q, Xu W, Xin Y, Bassett C, Wang Y (2011) Characterization of a chalcone synthase (CHS) flower-specific promoter from Lilium orential ‘Sorbonne’. Plant Cell Rep 30:2187–2194. doi:10.1007/s00299-011-1124-9
Manjunath S, Sachs MM (1997) Molecular characterization and promoter analysis of the maize cytosolic glyceraldehyde 3-phosphate dehydrogenase gene family and its expression during anoxia. Plant Mol Biol 33:97–112. doi:10.1023/A:1005729112038
Molnár A, Lovas A, Bánfalvi Z, Lakatos L, Polgár Z, Horváth S (2001) Tissue-specific signal(s) activate the promoter of a metallocarboxypeptidase inhibitor gene family in potato tuber and berry. Plant Mol Biol 46:301–311. doi:10.1023/A:1010649503229
Müller M, Knudsen S (1993) The nitrogen response of a barley C-hordein promoter is controlled by positive and negative regulation of the GCN4 and endosperm box. Plant J 4:343–355. doi:10.1046/j.1365-313X.1993.04020343.x
Noh SA, Lee H-S, Huh GH, Oh M-J, Paek K-H, Shin JS, Bae JM (2012) A sweetpotato SRD1 promoter confers strong root-, taproot-, and tuber-specific expression in Arabidopsis, carrot, and potato. Transgenic Res 21:265–278. doi:10.1007/s11248-011-9528-4
Ow DW, de Wet JR, Helinski DR, Howell SH, Wood KV, Deluca M (1986) Transient and stable expression of the firefly luciferase gene in plant cells and transgenic plants. Science 234:856–859. doi:10.1126/science.234.4778.856
Pourcel L, Routaboul JM, Kerhoas L, Caboche M, Lepiniec L, Debeaujon I (2005) TRANSPARENT TESTA10 encodes a laccase-like enzyme involved in oxidative polymerization of flavonoids in Arabidopsis seed coat. Plant Cell 17:2966–2980. doi:10.1105/tpc.105.035154
Ranocha P, McDougall G, Hawkins S, Sterjiades R, Borderies G, Stewart D, Cabanes-Macheteau M, Boudet AM, Goffner D (1999) Biochemical characterization, molecular cloning and expression of laccases-a divergent gene family-in poplar. Eur J Biochem 259:485–495. doi:10.1046/j.1432-1327.1999.00061.x
Rombauts S, Déhais P, Van Montagu M, Rouze P (1999) PlantCARE, a plant cis-acting regulatory element database. Nucleic Acids Res 27(1):295–296. doi:10.1093/nar/27.1.295
Sharma N, Russell SD, Bhalla PL, Singh MB (2011) Putative cis-regulatory elements in genes highly expressed in rice sperm cells. BMC 4:319. doi:10.1186/1756-0500-4-319
Shelton AM, Zhao JZ, Roush RT (2002) Economic, ecological, food safety, and social consequences of the deployment of Bt transgenic plants. Ann Rev Entomol 47:845–881. doi:10.1146/annurev.ento.47.091201.145309
Tu CH, Liu WP, Huang LL, Mo YQ, Yang DZ (2009) Cloning and transcriptional activity of a novel ovarian-specific promoter from rat retrovirus-like elements. Arch Biochem Biophys 485:24–29. doi:10.1016/j.abb.2009.02.004
Venter M, Botha FC (2004) Promoter analysis and transcription profiling: integration of genetic data enhances understanding of gene expression. Physiol Planta 120:74–83. doi:10.1111/j.0031-9317.2004.0209.x
Villain P, Mache R, Zhou DX (1996) The mechanism of GT element-mediated cell type-specific transcriptional control. J Biol Chem 271:32593–32598. doi:10.1074/jbc.271.51.32593
Washida H, Wu C-Y, Suzuki A, Yamanouchi U, Akihama T, Harada K, Takaiwa F (1999) Identification of cis-regulatory elements required for endosperm expression of the rice storage protein glutelin gene GluB-1. Plant Mol Biol 40:1–12. doi:10.1023/A:1026459229671
Wei JZ, Tirajoh A, Effendy J, Plant AL (2000) Characterization of salt-induced changes in gene expression in tomato (Lycopersicon esculentum) roots and the role played by abscisic acid. Plant Sci 159:135–148. doi:10.1016/S0168-9452(00)00344-7
Wu CY, Washida H, Onodera Y, Harada K, Takaiwa F (2000) Quantitative nature of the prolamin-box, ACGT and AACA motifs in a rice glutelin gene promoter: minimal cis-element requirements for endosperm-specific gene expression. Plant J 23:415–421. doi:10.1046/j.1365-313x.2000.00797.x
Xie C, Zhang B, Wang D, Kou F, Zhao X, Yang X (2011) Molecular cloning and characterization of an achene-seed-specific promoter from motherwort (Leonurus japonicus Houtt). Biotechnol Lett 33(1):167–172. doi:10.1007/s10529-010-0392-8
Yin T, Wu H, Zhang S, Liu J, Lu H, Zhang L, Xu Y, Chen D (2009) Two negative cis-regulatory regions involved in friut-specific promoter activity from watermelon (Citrullus vulgaris S.). J Exp Bot 60:169–185. doi:10.1093/jxb/ern273
Zavallo D, Bilbao ML, Hopp HE, Heinz R (2010) Isolation and functional characterization of two novel seed-specific promoters from sunflower (Helianthus annuus L.). Plant Cell Rep 29:239–248. doi:10.1007/s00299-010-0816-x
Acknowledgments
This work was supported by a Grant from the KRIBB Research Initiative Program and the National Coordinating Center for Global Cosmetics R&D (HN13C0071) of the Republic of Korea.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ha, JH., Moon, KB., Kim, MS. et al. The laccase promoter of potato confers strong tuber-specific expression in transgenic plants. Plant Cell Tiss Organ Cult 120, 57–68 (2015). https://doi.org/10.1007/s11240-014-0578-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-014-0578-1