Abstract
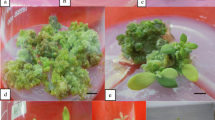
An efficient and reproducible procedure is described for direct shoot regeneration in Drymaria cordata Willd. using leaf explants cultured on Murashige and Skoog (MS) medium supplemented with α-naphthalene acetic acid (NAA) and 6-benzyladenine. The regeneration frequency varied with the plant growth regulator concentrations, orientation of the explants, and the carbon source and basal salts present in the regeneration medium. The highest mean number of shoots per explant (10.65 ± 1.03) was recorded on MS plates containing 3% sucrose and 0.8% agar supplemented with 0.1 mg/l NAA and 1.0 mg/l BAP. Shoot buds were induced in the basal parts of the leaf explants. Concentrations of NAA exceeding 1 mg/l suppressed shoot regeneration. Explants bearing the entire lamina and petiole were much more responsive than those having only the lamina. The plantlets that regenerated from the leaf explants were rooted successively on MS medium alone or in combination with indole butyric acid (IBA). The highest mean number of root organogenesis, with 25.67 ± 3.68 roots per leaf segment, was obtained in the presence of 1 mg/l IBA. Histological investigations of the regenerating shoots showed that the shoot buds had emerged from epidermal cells without callus formation. More than 90% of the in vitro-propagated plants survived when transferred to a greenhouse for acclimatization. Thus, this optimized regeneration system may be used for rapid shoot proliferation and genetic transformation.


Similar content being viewed by others
Abbreviations
- PGR:
-
Plant growth regulator
- MS:
-
Murashige and Skoog
- SH:
-
Schenk and Hildebrand
- B5:
-
Gambrog et al. (1968) medium
- IBA:
-
Indole-3-butyric acid
- BA:
-
6-Benzyladenine
- TDZ:
-
Thidiazuron
- NAA:
-
Naphthalene acetic acid
- 2,4-D:
-
Dicholorophenoxy acetic acid
References
Ahn YJ, Vang L, McKeon TA, Chen GQ (2007) High-frequency plant regeneration through adventitious shoot formation in castor (Ricinus communis L.). In Vitro Cell Dev Biol Plant 43:9–15
Bhagwat B, Lane WD (2004) In vitro shoot regeneration from leaves of sweet cherry (Prunus avium) ‘lapins’ and ‘sweetheart’. Plant Cell Tiss Org Cult 78:173–181
Dequan L, Gilbert MG (2001) Drymaria Willdenow ex Schultes in Roemer & Schultes. Flora China 6:5–6
Ding Z, Zhou J, Tan N (1999) A novel flavonoid glycoside from Drymaria diandra. Planta Med 65:578–579
Ding Z, Zhou J, Tan N, Teng R (2000) Two new cyclic peptides from Drymaria diandra. Planta Med 66:386–388
Espinosa AC, Pijut PM, Michler CH (2006) Adventitious shoot regeneration and rooting of Prunus serotina in vitro cultures. Hort Sci 41:193–201
Focho DA, Ndam WT, Fonge BA (2009) Medicinal plants of Aguambu Bamumbu in the Lebialem highlands, southwest province of Cameroon. Afr J Pharm Pharmacol 3:001–013
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp Cell Res 50:151–158
George EF, Sherrington PD (1984) Plant propagation by tissue culture. Exegetics Ltd, Westbury, pp 25–71
Guo DP, Zhu ZJ, Hu XX, Zheng SJ (2005) Effect of cytokinins on shoot regeneration from cotyledon and leaf segment of stem mustard (Brassica juncea var. tsatsai. Plant Cell Tiss Org Cult 83:123–127
Herve P, Jauneau A, Paques M, Marien J, Boudet AM, Teulieres C (2001) A procedure for shoot organogenesis in vitro from leaves and nodes of an elite Eucalyptus gunnii clone: comparative histology. Plant Sci 161:645–653
Hsieh PW, Chang FR, Lee KH, Hwang TL, Chang SM, Wu YC (2004) A new anti-HIV alkaloid, Drymaritin, and a new C-glycoside flavonoid, diandraflavone, from Drymaria diandra. J Nat Prod 67:1175–1177
Korban SS, O’Connor PA, Elobeidy A (1992) Effects of thidiazuron, naphthaleneacetic acid, dark incubation and genotype on shoot organogenesis from Malus leaves. J Hort Sci 67:341–349
Kukulczanka K, Suszyfiska G (1972) Regenerative properties of Saintpaulia ionantha Wendl. leaves cultured in vitro. Acta Soc Bot Poloniae 41:503–510
Kumar PP, Dimps Rao C, Goh CJ (1998) Influence of petiole and lamina on adventitious shoot initiation from leaf explants of Paulownia fortunei. Plant Cell Rep 17:886–890
Leblay C, Chevreau E, Raboin LM (1990) Adventitious shoot regeneration from in vitro leaves of several pear cultivars (Pyrus communis L.). Plant Cell Tiss Org Cult 25:99–105
Liu X, Pijut PM (2008) Plant regeneration from in vitro leaves of mature black cherry (Prunus serotina). Plant Cell Tiss Organ Cult 94:113–123
Mello MO, Dias CTS, Amaral AFC, Melo M (2001) Growth of Bauhinia forticata Link, Curcuma zedoaria Roscoe and Phaseolus vulgaris L. cell suspension cultures with carbon sources. Sci Agric 58:481–485
Miyazaki J, Sugimura Y, Kotani E, Furusawa T (2000) Shoot organogenesis from mulberry leaf tissue: origin and development of regenerated shoots. J Seic Sci Jpn 69(5):319–325
Mukherjee PK, Bhattacharya S, Saha K, Giri SN, Pal M, Saha BP (1996) Antibacterial evaluation of Drymaria cordata Willd. (Fam. Caryophyllaceae) extract. Phyto Res 11:249–250
Mukherjee PK, Saha K, Bhattacharya S, Giri SN, Pal M, Saha BP (1997) Studies on antitussive activity of Drymaria cordata Willd. J Ethnopharm 56:77–80
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Plant Phyiol 15:473–497
Neogi B, Prasad MNV, Roa RR (1989) Ethnobotany of some weeds of Khasi and Garo hills, Meghalaya, Northeastern India. Economic Bot 43:471–479
Purohit SD, Dava A, Kukda G (1994) Micropropagation of safed musli (Chlorophytum borivilianum), a rare Indian medicinal herb. Plant Cell Tiss Org Cult 39:93–96
Rao RR (1981) Ethnobotany of Meghalaya: medicinal plants used by Khasi and Garo tribes. Econ Bot 35:4–9
Sanyal I, Singh AK, Kaushik M, Amla DV (2005) Agrobacterium-mediated transformation of chickpea (Cicer arietinum L.) with Bacillus thuringiensis, cry1Ac gene for resistance against pod borer insect Helicoverpa armigera. Plant Sci 168:1135–1146
Schenk RU, Hildebrandt AC (1972) Medium and techniques for the induction and growth of monocotyledonous and dicotyledonous plant cell cultures. Can J Bot 50:199–204
Shende S, Rai M (2005) Multiple shoot formation and plant regeneration of a commercially-useful tropical plant, Buchanania lanzan (Spreng). Plant Biotech 22:59–61
Sikdar M, Dutta U (2008) Traditional phytotherapy among the Nath people of Assam. Ethno Med 2:39–45
Singh A, Malhotra S, Subban R (2008) Anti-inflammatory and analgesic agents from Indian medicinal plants. Inter J Integra Biol 3:57–72
Srivastava TN, Kaphi BK (1992) Medicinal and aromatic plant resources of Sikkim, Himalaya. J Econo Tax Bot 16:1–17
Tang H, Zhenglong R, Reustle G, Krczal G (2002) Plant regeneration from leaves of sweet and sour cherry cultivars. Sci Hort 93:235–244
Tawfik AA, Noga G (2001) Adventitious shoot proliferation from hypocotyls and internodal stem explants of cumin. Plant Cell Tiss Org Cult 66:141–147
Vargas D, Dominguez XA, Acuna Askar AK, Gutierrez M, Hostettmann K (1988) Norditerpenes and norditerpene glycosides from Drymaria arenarioides. Phytochem 27:1532–1534
Veitch NC, Grayer RJ (2008) Flavonoids and their glycosides, including anthocyanins. Nat Prod Rep 25:555–611
Verma DC, Dougall DK (1977) Influence of carbohydrates on quantitative aspects of growth and embryo formation in wild carrot suspension cultures. Plant Physiol 59:81–85
Yancheva SD, Golubowicz S, Fisher E, Yadun SL, Flaishman MA (2003) Auxin type and timing of application determine the activation of the developmental program during in vitro organogenesis in apple. Plant Sci 165:299–309
Acknowledgments
This work was supported by Konkuk University BrainPool for Dr B. K. Ghimire.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghimire, B.K., Seong, E.S., Goh, E.J. et al. High-frequency direct shoot regeneration from Drymaria cordata Willd. leaves. Plant Cell Tiss Organ Cult 100, 209–217 (2010). https://doi.org/10.1007/s11240-009-9627-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-009-9627-6