Abstract
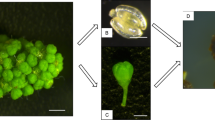
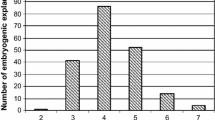
A study of four parameters (induction medium, floral explant, developmental stage and year) was carried out to determine the best combination for the embryogenesis induction of eight grapevine (Vitis vinifera L.) cultivars. Anthers and ovaries were extracted from flower buds at three developmental phases and incubated in two induction media over two consecutive years. As average, the percentage of embryogenesis on Nitsch and Nitsch-derived medium (9.1%) was higher than in Murashige and Skoog-derived medium (5.9%) and embryogenesis from ovaries (10.1%) was 2-fold higher than from anthers (4.9%). Earlier flower developmental stages (II–III) favored embryogenic induction from anthers, while later stages (III–V) did it from ovaries. Induction of embryogenic cultures was genotype dependent. Two years after the establishment of the embryogenic lines, an average of 48.0% of the pro-embryogenic masses were viable and suitable to initiate cell suspensions. Embryogenic cultures of four genotypes showed a high percentage of conversion from embryos to plants: Albariño (61.8%), Garnacha (48.8%), Tempanillo (71.0%) and Sultanina (69.0%). Moreover, cell suspensions were competent for transient transformation based on β-glucuronidase assay, as up to 6,387 blue spots per Petri plate after Biolistic bombardment were obtained. Here, we present the advantage of ovaries over anthers for the embryogenesis induction of several grapevine cultivars. This is the first report of embryogenesis from the cultivars Albariño, Verdejo and Muscat Hamburg as well as transient transformation of Albariño and Tempranillo.

Similar content being viewed by others
Abbreviations
- 2, 4-D:
-
2,4-Dichlorophenoxyacetic acid
- 4-CPPU:
-
N-(2-chloro-4-pyridyl)-N′-phenylurea
- BAP:
-
6-Benzylaminopurine
- NOA:
-
β-Naphthoxyacetic acid
- MS:
-
Murashige and Skoog medium
- NN:
-
Nitsch and Nitsch medium
- PEM:
-
Pro-embryogenic masses
References
Agresti A (1996) An introduction to categorical data analysis. Wiley, London
Ben Amar A, Cobanov P, Boonrod K, Krczal G, Bouzid S, Ghorbel A, Reustle GM (2007) Efficient procedure for grapevine embryogenic suspension establishment and plant regeneration: role of conditioned medium for cell proliferation. Plant Cell Rep 26:1439–1447
Bornhoff BA, Harst M (2000) Establishment of embryo suspension cultures of grapevines (Vitis L.). Vitis 39:27–29
Coutos-Thevenot P, Goebeel I, Mauro MC, Jouanneau JP, Boulay M, Deloire A, Guern J (1992) Somatic embryogenesis from grapevine cells. Improvement of embryo development by changes in culture conditions. Plant Cell Tissue Organ Cult 29:125–133. doi:10.1007/BF00033617
Franks T, He FG, Thomas M (1998) Regeneration of transgenic Vitis vinifera L. Sultana plants: genotypic and phenotypic analysis. Mol Breed 4:321–333. doi:10.1023/A:1009673619456
Franks T, Botta R, Thomas MR (2002) Chimerism in grapevines: implications for cultivar identity, ancestry and genetic improvement. Theor Appl Genet 104:192–199. doi:10.1007/s001220100683
Gambino G, Ruffa P, Vallania R, Gribaudo I (2007) Somatic embryogenesis from whole flowers, anthers and ovaries of grapevine (Vitis spp). Plant Cell Tissue Organ Cult 90:79–83. doi:10.1007/s11240-007-9256-x
Gray DJ (1995) Somatic embryogenesis in grape. In: Jain S, Gupta P, Newton R (eds) Somatic embryogenesis in woody plants, vol 2. Kluwer Academic Publishers, Dordrecht, pp 191–217
Gribaudo I, Gambino G, Vallania R (2004) Somatic embryogenesis from grapevine anthers: the optimal developmental stage for collecting explants. Am J Enol Vitic 55:427–430
Hébert D, Kikkert JR, Smith FD, Reisch BI (1993) Optimization of biolistic transformation of embryogenic grapes cell suspension. Plant Cell Rep 12:585–589. doi:10.1007/BF00233066
Ihaka R, Gentleman R (1996) R: a language for data analysis and graphics. J Comput Graph Stat 5:299–314. doi:10.2307/1390807
Iocco P, Franks T, Thomas MR (2001) Genetic transformation of major wine grape cultivars of Vitis vinifera L. Transgenic Res 10:105–112. doi:10.1023/A:1008989610340
Jaillon O, Aury JM, Noel B, Policriti A, Clepet C, Casagrande A et al (2007) The grapevine genome sequence suggest ancestral hexaploidization in major angiosperm phyla. Nature 449:463–465. doi:10.1038/nature06148
Jayasankar S, Gray DJ, Litz RE (1999) High-efficiency somatic embryogenesis and plant regeneration from suspension cultures of grapevine. Plant Cell Rep 18:533–537. doi:10.1007/s002990050617
Kikkert JR, Hébert-Soulé D, Wallace PG, Striem MJ, Reisch BI (1996) Transgenic plantlets of ‘Chancellor’ grapevine (Vitis sp.) from biolistic transformation of embryogenic cell suspensions. Plant Cell Rep 15:311–316
Kikkert JR, Vidal JR, Reisch BI (2004) Stable plant transformation via particle bombardment/biolistics. In: Peña L (ed) Methods in molecular biology, volume 286, transgenic plants: methods and protocols. Humana Press, Totowa, pp 61–78
Kikkert JR, Striem MJ, Vidal JR, Wallace PG, Barnard J, Reisch BI (2005) Long-term study of somatic embryogenesis from anthers and ovaries of 12 grapevine (Vitis sp.) genotypes. In Vitro Cell Dev Biol Plant 41:232–239. doi:10.1079/IVP2004609
López-Pérez AJ, Carreño J, Martínez-Cutillas A, Dabauza M (2005) High embryogenesis ability and plant regeneration of table grapevine cultivars (Vitis vinifera L.) induced by activated charcoal. Vitis 44:79–85
Martinelli L, Gribaudo I (2001) Somatic embryogenesis in grapevine. In: Roubelakis-Angelakis KA (ed) Molecular biology and biotechnology of the grapevine. Kluwer Academic Publishers, Dordrecht, pp 393–410
Martinelli L, Mandolino G (1994) Genetic transformation and regeneration of transgenic plants in grapevine (Vitis rupestris S.). Theor Appl Genet 88:621–628. doi:10.1007/BF01253963
Martinelli L, Bragagna P, Poletti V, Scienza A (1993) Somatic embryogenesis from leaf- and petiole-derived callus of Vitis rupestris. Plant Cell Rep 12:207–210. doi:10.1007/BF00237055
Martinelli L, Gribaudo I, Bertoldi D, Candioli E, Poletti V (2001) High efficiency somatic embryogenesis and plant germination in grapevine cultivars Chardonnay and Brachetto a grappolo lungo. Vitis 40:111–115
Mauro MC, Nef C, Fallot J (1986) Stimulation of somatic embryogenesis and plant regeneration from anther culture of Vitis vinifera cv. Cabernet-Sauvignon. Plant Cell Rep 5:377–380. doi:10.1007/BF00268606
Mauro MC, Toutain S, Walter B, Pinck L, Otten L, Coutos-Thevenot P, Deloire A, Barbier P (1995) High efficiency regeneration of grapevine plants transformed with the GFLV coat protein gene. Plant Sci 112:97–106. doi:10.1016/0168-9452(95)04246-Q
Mullins MG, Bouquet A, Williams LE (1992) Biology of the grapevine. Cambridge University Press, Cambridge
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497. doi:10.1111/j.1399-3054.1962.tb08052.x
Nakano M, Sakakibara T, Watanabe Y, Mii M (1997) Establishment of embryogenic cultures in several cultivars of Vitis vinifera and V. x labruscana. Vitis 36:141–145
Nitsch JP, Nitsch C (1969) Haploid plants from pollen grains. Science 163:85–87. doi:10.1126/science.163.3862.85
Perl A, Lotan O, Abuabied M, Holland D (1996) Establishment of an Agrobacterium-mediated transformation system for Vitis vinifera L.: the role of antioxidants during grape-agrobacterium interactions. Nat Biotechnol 14:624–628. doi:10.1038/nbt0596-624
Perrin M, Martin D, Joly D, Demangeat G, This P, Masson JE (2001) Medium-dependent response of grapevine somatic embryogenic cells. Plant Sci 161:107–116. doi:10.1016/S0168-9452(01)00385-5
Perrin M, Gertz C, Masson JE (2004) High efficiency initiation of regenerable embryogenesis callus from anther filaments of 19-grapevine genotypes grown worldwide. Plant Sci 167:1343–1349. doi:10.1016/j.plantsci.2004.07.009
Scorza R, Cordts JM, Gray DJ, Gonsalves D, Emershad RI, Ramming DW (1996) Producing transgenic thompson seedless grape (Vitis vinifera) plants. J Am Soc Hortic Sci 121:616–619
Stamp JA, Meredith CP (1988) Somatic embryogenesis from leaves and anthers of grapevine. Sci Hortic (Amsterdam) 35:235–250. doi:10.1016/0304-4238(88)90117-3
Steponkus PL, Lanphear FO (1967) Refinement of the triphenyltetrazolium chloride method of determining cold injury. Plant Physiol 42:1423–1426
Torregrosa L (1998) A simple an efficient method to obtain stable embryogenic cultures from anthers of Vitis vinifera. Vitis 37:91–92
Vidal JR, Kikkert JR, Wallace PG, Reisch BI (2003) High-efficiency biolistic co-transformation and regeneration of Chardonnay (Vitis vinifera L.) containing npt-II and antimicrobial peptide genes. Plant Cell Rep 22:252–260. doi:10.1007/s00299-003-0682-x
Vidal JR, Kikkert JR, Malnoy MA, Wallace PG, Barnard J, Reisch BI (2006a) Evaluation of transgenic Chardonnay (Vitis vinifera) containing magainin genes for resistance to crown gall and powdery mildew. Transgenic Res 15:69–82. doi:10.1007/s11248-005-4423-5
Vidal JR, Kikkert JR, Donzelli BD, Wallace PG, Reisch BI (2006b) Biolistic transformation of grapevine using minimal gene cassette technology. Plant Cell Rep 25:807–814. doi:10.1007/s00299-006-0132-7
Yamamoto T, Iketani H, Leki H, Nishizawa Y, Notsuka K, Hibi T, Hayashi T, Matsuta N (2000) Transgenic grapevine plants expressing a rice chitinase with enhanced resistance to fungal pathogens. Plant Cell Rep 19:639–646. doi:10.1007/s002999900174
Acknowledgments
We gratefully acknowledge financial support from a Marie Curie International Re-integration Grant (MIRG-2005-28341) of the European Commission and an I + D + i Grant (AGL-2006-5856) of the Spanish Ministry of Education and Science (MEC). JR Vidal was fully supported by a Ramón y Cajal contract from MEC. Thanks are also extended to F. Cabello for allowing us to collect flower buds from vineyards at IMIDRA (Comunidad de Madrid) and to MJ Carmona for the use of her laboratory facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vidal, J.R., Rama, J., Taboada, L. et al. Improved somatic embryogenesis of grapevine (Vitis vinifera) with focus on induction parameters and efficient plant regeneration. Plant Cell Tiss Organ Cult 96, 85–94 (2009). https://doi.org/10.1007/s11240-008-9464-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-008-9464-z