Abstract
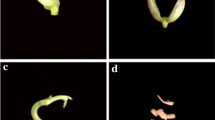
A protocol for in vitro regeneration via indirect organogenesis for Phaseolus vulgaris cv. Negro Jamapa was established. The explants used were apical meristems and cotyledonary nodes dissected from the embryonic axes of germinating seeds. Several auxin/cytokinin combinations were tested for callus induction. The best callus production was obtained with medium containing 1.5 μM 2,4-dichlorophenoxyacetic acid. After 2 weeks of growth calli were transferred to shooting medium containing 22.2 μM 6-benzylaminopurine. Shoots regenerated with a frequency of approximately 0.5 shoots per callus, and upon transfer to rooting medium these shoots produced roots with 100% efficiency. Histological analyses of the regeneration process confirmed the indirect organogenesis pattern. Greenhouse grown regenerated plants showed normal development and were fertile. The protocol was reproducible for other nine P. vulgaris cultivars tested, suggesting a genotype independent procedure.



Similar content being viewed by others
Abbreviations
- AM:
-
Apical meristem
- BAP:
-
6-Benzylaminopurine
- CIM:
-
Callus induction medium
- CN:
-
Cotyledonary node
- 2,4-D:
-
2,4-Dichlorophenoxyacetic acid
- IAA:
-
Indole-3-acetic acid
- MES:
-
2-(N-Morpholino) ethanesulfonic acid
- MS:
-
Murashige and Skoog medium
- NAA:
-
α-naphthaleneacetic acid
- PVP-360:
-
Polyvinylpyrrolidone 360,000 molecular weight
- SM:
-
Shooting medium
- RM:
-
Rooting medium
References
Broughton WJ, Hernández G, Blair M, Beebe S, Gepts P, Vanderleyden J (2003) Beans (Phaseolus spp.)—model food legumes. Plant Soil 252:55–128. doi:10.1023/A:1024146710611
Chu CC, Wang CC, Sun SC, Hsu C, Yin KC, Chu CY, Bi FY (1975) Establishment of an efficient medium for anther culture of rice through comparative experiments on the nitrogen sources. Sci Sin 18:659–688
Cruz de Carvalho MH, Van Le B, Zuily-Fodil Y, Pham Thi AT, Tran Thanh Van K (2000) Efficient whole plant regeneration of common bean (Phaseolus vulgaris L.) using thin-cell-layer culture and silver nitrate. Plant Sci 159:223–232. doi:10.1016/S0168-9452(00)00346-0
Delgado-Sánchez P, Saucedo-Ruiz M, Guzmán-Maldonado SH, Villordo-Pineda E, González Chavira M, Fraire-Velázquez S, Acosta-Gallegos JA, Mora-Avilés A (2006) An organogenic plant regeneration system for common bean (Phaseolus vulgaris L). Plant Sci 170:822–827. doi:10.1016/j.plantsci.2005.11.015
Dillen W, De Clercq J, Goossens A, Van Montagu M, Angenon G (1997) Agrobacterium mediated transformation of Phaseolus acutifolius A. gray. Theor Appl Genet 94:151–158. doi:10.1007/s001220050394
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp Cell Res 50:151–158. doi:10.1016/0014-4827(68)90403-5
Malik KA, Saxena PK (1992) Somatic embryogenesis and shoot regeneration from intact seedlings of Phaseolus acutifolius A., P. aureus (L.) Wilczek, P.coccineus L., and P. wrightii L. Plant Cell Rep 11:163–168. doi:10.1007/BF00232172
Mohamed MF, Coyne DP, Read PE (1993) Shoot organogenesis in callus induced from pedicel explants of common bean (Phaseolus vulgaris L.). J Am Soc Hortic Sci 118:158–162
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio-assays with tobacco tissue cultures. Physiol Plant 15:473–497. doi:10.1111/j.1399-3054.1962.tb08052.x
Nagl W, Ignacimuthu S, Becker J (1997) Genetic engineering and regeneration of Phaseolus and Vigna. State of the art and new attempts. J Plant Physiol 150:625–644
Pigeaire A, Abernethy D, Smith PM, Simpson K, Fletcher N, Lu CY, Atkins CA, Cornish E (1997) Transformation of a grain legume (Lupinus angustifolius L) via Agrobacterium tumefaciens-mediated gene transfer to shoot apices. Mol Breed 3:341–349. doi:10.1023/A:1009642620907
Rech EL, Giovanni R, Vianna GR, Aragao FJL (2008) High-efficiency transformation by biolistics of soybean, common bean and cotton transgenic plants. Nat Protoc 3:410–418. doi:10.1038/nprot.2008.9
Santalla M, Power JB, Davey MR (1998) Efficient in vitro shoot regeneration responses of Phaseolus vulgaris and P coccineus. Euphytica 102:195–202. doi:10.1023/A:1018317327302
Trepp GB, Van de Mortel M, Yoshioka H, Miller SS, Samac DA, Gant JS, Vance CP (1999) NADH-glutamate synthase in alfalfa root nodules. Genetic regulation and cellular expression. Plant Physiol 119:817–828. doi:10.1104/pp.119.3.817
Zambre M, Goossens A, Cardona C, Van Montagu M, Terryn N, Angenon G (2005) A reproducible genetic transformation system for cultivated Phaseolus acutifolius (tepary bean) and its use to assess the role of arcelins in resistance to the Mexican bean weevil. Theor Appl Genet 110:914–924. doi:10.1007/s00122-004-1910-7
Zambre MA, De Clercq J, Vranová E, Van Montagu M, Angenon G, Dillen W (1998) Plant regeneration from embryo-derived callus in Phaseolus vulgaris L (common bean) and P acutifolius A. Gray (tepary bean). Plant Cell Rep 17:626–630. doi:10.1007/s002990050455
Zambre MA, Geerts P, Maquet A, Van Montagu M, Dillen W, Angenon G (2001) Regeneration of fertile plants from callus in Phaseolus polyanthus Greenman (Year bean). Ann Bot (Lond) 88:371–377. doi:10.1006/anbo.2001.1468
Acknowledgments
We thank Drs. M. Blair, S. Beebe and D. Debouck from CIAT for kindly supplying seeds, PhD Pallavolu Maheswara Reddy from CCG-UNAM for critical review, Victor Bustos from CCG-UNAM, Elizabeth González from CEIB-UAEM and to Ana Isabel Bieler Antolín from Microcine FC-UNAM, for technical assistance on plant maintenance, histological analysis and microphotography, respectively.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arellano, J., Fuentes, S.I., Castillo-España, P. et al. Regeneration of different cultivars of common bean (Phaseolus vulgaris L.) via indirect organogenesis. Plant Cell Tiss Organ Cult 96, 11–18 (2009). https://doi.org/10.1007/s11240-008-9454-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-008-9454-1