Abstract
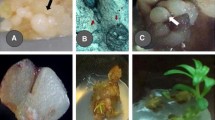
The effects of different factors on the embryogenesis and plant regeneration from mature embryos of Russian spring and winter genotypes were studied. Embryogenic callus induction was achieved on MS medium supplemented with different concentrations of 2,4-D (2,4-dichlorophenoxyacetic acid), 2,4,5-T (2,4,5-trichlorophenoxyacetic acid) or Dicamba (3,6-dichloro-o-anisic acid). Although all auxins were able to induce callus from explants with high frequency (98–100%), Dicamba was more effective for the induction of embryogenic callus (21.8–38.3%). Maximum embryogenic callus formation and high number of regenerated plants were observed at 12 mg l−1 of Dicamba. The time exposure to Dicamba (7, 14, 21 and 28 days) had a significant effect on efficiency of somatic embryogenesis. When contact of explants with callus induction medium was increased from 7 to 21 days the rate of somatic embryogenesis and number of regenerated plants per embryogenic callus gradually increased from 13.0 to 38.4% and 3.6 to 8.0%, respectively. Supplement of additional auxins (indoleacetic acid (IAA), indolebutyric acid (IBA), and naphthaleneacetic acid (NAA)) to callus induction medium with Dicamba had a positive effect on the rate of embryogenic callus formation, while the average number of regenerated shoots was not affected. The best rate of somatic embryogenesis was observed at the addition of 0.5 mg l−1 IAA with Dicamba (61.0%). The optimum combination of Dicamba and IAA increased the efficiency of somatic embryogenesis and plant regeneration from seven spring and winter wheat genotypes, thought overall morphogenic capacity was still genotype dependent.
Similar content being viewed by others
Abbreviations
- 2,4,5-T:
-
2,4,5-trichlorophenoxyacetic acid
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- IAA:
-
indoleacetic acid
- IBA:
-
indolebutyric acid
- NAA:
-
naphthaleneacetic acid
References
Ahmed KZ, Bartok T, Sagi F, (1992) A modified method for rapid callus induction by utilization of endosperm metabolites in mature and immature seeds of bread wheat (Triticum aestivum L.) and durum wheat (Triticum durum L.) Cereal. Res. Commun. 20: 81–86
Ahuja PS, Pental D, Cocking EC, (1982) Plant regeneration from leaf base callus and cell suspensions of Triticum aestivum Z. Pflanzenzücht. 89: 139–144
Barro F, Martin A, Lazzeri PA, Barceló P, (1999) Medium optimization for efficient somatic embryogenesis and plant regeneration from immature inflorescences and immature scutella of elite cultivars of wheat, barley and tritordeum Euphytica 108: 161–167
Bartok T, Sagi F, (1990) A new endosperm supported callus induction method for wheat (Triticum aestivum L.) Plant Cell Tiss. Org. Cult. 22: 37–41
Benkirane H, Sabounji K, Chlyah A, Chlyah H, (2000) Somatic embryogenesis and plant regeneration from fragments of immature inflorescences and coleoptiles of durum wheat Plant Cell Tiss. Org. Cult. 61: 107–113
De Klerk GJ, Ter Brugge JT, Marinova S, (1997) Effectiveness of indoleacetic acid, indolebutyric acid and naphthaleneacetic acid during adventitious root formation in vitro in Malus ‘Jork 9’.Plant Cell Tiss Org. Cult. 49: 39–44
Delporte F, Mostade O, Jacquemin JM, (2001) Plant regeneration through callus initiation from thin mature embryo fragments of wheat Plant Cell Tiss. Org. Cult. 67: 73–80
Fennel S, Bohoroba N, Crossa J, Hoisington D, Van-Ginkel M, (1996) Plant regeneration from immature embryos of 48 elite CIMMYT bread wheats Theor. Appl. Genet. 92: 163–169
He DG, Ouyang JW, (1984) Callus and plantlet formation from cultured wheat anthers at different developmental stages Plant Sci. Lett. 33: 71–79
Heyser JW, Nabors MW, MacKinnon C, Dykes TA, Demott KJ, Kautzman DC, Mujeeb-Kazi A, (1985) Long-term, high-frequency plant regeneration and the induction of somatic embryogenesis in callus cultures of wheat (Triticum aestivum L.) Z. Pflanzenzücht. 94: 218–233
Hunsinger H, Schauz K, (1987) The influence of Dicamba on somatic embryogenesis and frequency of plant regeneration from cultured immature embryos of wheat (Tritium aestivum L.). Plant Breed. 98: 119–123
Jelaska S, Rengel Z, Cesar V, (1984) Plant regeneration from mesocotyl callus of Hordeum vulgare L Plant Cell Rep. 3: 125–129
Jimenez VM, Bangerth F, (2001) Endogenous hormone concentrations and embryogenic callus development in wheat Plant Cell Tiss. Org. Cult. 67: 37–46
Kato K, Chowdhury SH, Harada S, (1991) Effect of culture condition on plant regeneration capacity of mature embryo derived callus in wheat (Triticum aestivum L.) Wheat Inf. Serv. 72: 95–97
Luhrs R, Lorz H, (1987) Plant regeneration in vitro from embryogenic cultures of spring- and winter-type barley (Hordeum vulgare L.) Theor. Appl. Genet. 82: 74–80
Machii H, Mizuno H, Hirabayashi T, Li H, Hagio T, (1998) Screening wheat genotypes for high callus induction and regeneration capability from anther and immature embryo cultures Plant Cell Tiss. Org. Cult. 53: 67–74
Maddock SE, Lancaster VA, Risiott R, Franklin J, (1983) Plant regeneration from cultured immature embryos and inflorescences of 25 cultivars of wheat (Triticum aestivum) J. Exp. Bot. 34: 915–926
McHughen A, (1983) Rapid regeneration of wheat in vitro Ann. Bot. 51: 851–853
Moore TC, (1989) Biochemistry and Physiology of Plant Hormones. Springer, Berlin Heidelberg New York, p 330
Murashige T, Skoog F, (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures Physiol. Plant 15: 473–497
Orshinsky BR, Sadasivaiah RS, (1997) Effect of plant growth conditions, plating density, and genotype on the anther culture response of soft white spring wheat hybrids Plant Cell Rep. 16: 758–762
Özgen M, Türet M, Altmok S, Sancak C, (1998) Efficient callus culture induction and plant regeneration from mature embryo culture of winter wheat (Triticum aestivum L) genotypes Plant Cell Rep. 18: 331–335
Özgen M, Türet M, Özcan S, Sancak C, (1996) Callus induction and plant regeneration from immature and mature embryos of winter durum wheat genotypes Plant Breed. 115: 455–458
Ozias-Akins P, Vasil IK, (1982) Plant regeneration from cultured embryos and inflorescence of Triticum aestivum (wheat): evidence for somatic embryogenesis Protoplasma 110: 95–105
Papenfus JM, Carman JG, (1987) Enhanced regeneration from wheat callus cultures using dicamba and kinetin Crop Sci. 27: 588–593
Patnaik D, Khurana P, (2001) Wheat biotechnology: a minireview Elect. J. Biotechnol. 4(2): 1–29
Šamaj J, Baluška F, Pretová A, Volkmann D, (2003) Auxin deprivation induces a developmental switch in maize somatic embryogenesis involving redistribution of microtubules and actin filaments from endoplasmic to cortical cytoskeletal arrays Plant Cell Rep. 21: 940–945
Redway FA, Vasil V, Lu D, Vasil IK, (1990) Identification of callus types for long-term maintenance and regeneration from commercial cultivars of wheat (Triticum aestivum L.) Theor. Appl. Genet. 79: 609–617
Vasil IK, Vasil V, (1986) Regeneration in cereals and other grass species In Vasil (ed) Cell Culture and Somatic Cell Genetics of Plant, vol 3 Plant Regeneration and Genetic Variability Academic Press, Orlando, pp 121–150
Zale JM, Borchardt-Wier H, Kidwell KK, Steber CM, (2004) Callus induction and plant regeneration from mature embryos of a diverse set of wheat genotypes Plant Cell Tiss. Org. Cult. 76: 277–281
Zamora AB, Scott KJ, (1983) Callus formation and plant regeneration from wheat leaves Plant Sci. Lett. 29: 183–189
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Filippov, M., Miroshnichenko, D., Vernikovskaya, D. et al. The effect of auxins, time exposure to auxin and genotypes on somatic embryogenesis from mature embryos of wheat. Plant Cell Tiss Organ Cult 84, 213–222 (2006). https://doi.org/10.1007/s11240-005-9026-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-005-9026-6