Abstract
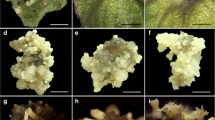
An efficient in vitro plant regeneration method through somatic embryogenesis has been established in Haworthia retusa. Somatic embryos were induced from leaf explants on Murashige and Skoog (MS) medium supplemented with different concentrations of 2,4-dichlorophenoxyacetic acid (2,4-D), indole-3-acetic acid (IAA), indole-3-butyric acid (IBA), and α-naphthaleneacetic acid (NAA) either alone or in combination with 4 μM thidiazuron (TDZ). Of the four auxins studied, IBA was found to be the most promising in terms of somatic embryo induction, followed in decreasing frequency by 2,4-D, IAA, and NAA. The highest somatic embryo induction (60.7%), with a mean of 20.7 embryos per leaf explant, was observed on MS medium amended with 20 μM IBA. The inclusion of 4 μM TDZ to the auxin-containing medium significantly (p < 0.0001) increased the somatic embryo induction frequency as well as the number of somatic embryos. The best combination for somatic embryogenesis was IBA + TDZ. The highest incidence of somatic embryo induction (100%), with a mean of 55.8 somatic embryos, was obtained on a culture medium containing 16 μM IBA + 4 μM TDZ. Somatic embryos germinated best on MS medium supplemented with 2 μM gibberellic acid. Morphological variations were observed among the regenerated plantlets. Well-developed plantlets obtained from germination media were acclimatized in the greenhouse.






Similar content being viewed by others
References
Bairu MW, Aremu AO, Van Staden J (2011) Somaclonal variation in plants: causes and detection methods. Plant Growth Regul 63:147–173. https://doi.org/10.1007/s10725-010-9554-x
Beyl CA, Sharma GC (1983) Picloram induced somatic embryogenesis in Gasteria and Haworthia. Plant Cell Tissue Organ Cult 2:23–132. https://doi.org/10.1007/BF00043357
Bradaï F, Pliego-Alfaro F, Sánchez-Romero C (2016) Somaclonal variation in olive (Olea europaea L.) plants regenerated via somatic embryogenesis: influence of genotype and culture age on phenotypic stability. Sci Hortic 213:208–215. https://doi.org/10.1016/j.scienta.2016.10.031
Devi K, Sharma M, Ahuja PS (2014) Direct somatic embryogenesis with high frequency plantlet regeneration and successive cormlet production in saffron (Crocus sativus L.). S Afr J Bot 93:207–216. https://doi.org/10.1016/j.sajb.2014.04.006
Hatzilazarou S, Kostas S, Economou A, Scaltsoyiannes A (2017) Efficient propagation of Nerium oleander L. through tissue culture. Propag Ornam Plants 17:64–74
Kaul K, Sabharwal PS (1972) Morphogenetic studies on Haworthia: establishment of tissue culture and control of differentiation. Am J Bot 59:377–385. https://doi.org/10.2307/2441548
Kaul K, Sabharwal PS (1975) Morphogenetic studies on Haworthia: effects of inositol on growth and differentiation. Am J Bot 62:655–659. https://doi.org/10.1002/j.1537-2197.1975.tb14098.x
Kim DH, Kang KW, Sivanesan I (2017a) In vitro propagation of Cymbidium hybrid. Propag Ornam Plants 17:48–54
Kim DH, Kang KW, Sivanesan I (2017b) Micropropagation of Haworthia retusa Duval. Propag Ornam Plants 17:77–82
Kumar V, Moyo M, Van Staden J (2016) Enhancing plant regeneration of Lachenalia viridiflora, a critically endangered ornamental geophyte with high floricultural potential. Sci Hortic 211:263–268. https://doi.org/10.1016/j.scienta.2016.08.024
Lincy KA, Remashree AB, Sasikumar B (2009) Indirect and direct somatic embryogenesis from aerial stem explants of ginger (Zingiber officinale Rosc.). Acta Bot Croat 68:93–103
Liu B, Fang H, Meng C, Chen M, Chai Q, Zhang K, Liu S (2017) Establishment of a rapid and efficient micropropagation system for succulent plant Haworthia turgida haw. HortScience 52:1278–1282. https://doi.org/10.21273/HORTSCI12056-17
Mahendran G, Narmatha Bai V (2016) Direct somatic embryogenesis of Malaxis densiflora (A. Rich.) Kuntze. J Genet Eng Biotechnol 14:77–81. https://doi.org/10.1016/j.jgeb.2015.11.003
Moradi Z, Farahani F, Sheidai M, Satari TN (2017) Somaclonal variation in banana (Musa acuminate colla cv. Valery) regenerated plantlets from somatic embryogenesis: histological and cytogenetic approaches. Caryologia 70:1–6. https://doi.org/10.1080/00087114.2016.1198665
Mujib A, Ali M, Tonk D, Zafar N (2017) Nuclear 2C DNA and genome size analysis in somatic embryo regenerated gladiolus plants using flow cytometry. Adv Hortic Sci 31:165–174. https://doi.org/10.13128/ahs-21956
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Mycock DJ, Watt MP, Hannweg KF, Naicker K, Makwarela M, Berjak P (1997) Somatic embryogenesis of two indigenous south African Haworthia spp. (H. limifolia and H. koelmaniorum). South Afr J Bot 63:345–350. https://doi.org/10.1016/S0254-6299(15)30784-5
Ogihara Y (1981) Tissue culture in Haworthia. Theor Appl Genet 60:353–363. https://doi.org/10.1007/BF00264330
Ogihara Y, Tsunewaki K (1978) Tissue culture in Haworthia I. effects of auxins and kinetin on callus growth. Bot Mag Tokyo 91:83–91. https://doi.org/10.1007/BF02489105
Ouyang Y, Chen Y, Lü J, Teixeira da Silva JA, Zhang X, Ma G (2016) Somatic embryogenesis and enhanced shoot organogenesis in Metabriggsia ovalifolia W. T. Wang. Sci Rep 19:6–24. https://doi.org/10.1038/srep24662
Pandey KN, Sabharwal PS, Calkins J (1979) Effects of ionizing radiation (60Co gamma rays) on growth and morphogenesis of Haworthia mirabilis, haw. callus tissues. In Vitro 15:246–251. https://doi.org/10.1007/BF02618947
Pikulthong V, Teerakathiti T, Thamchaipenet A, Peyachoknagul S (2016) Development of somatic embryos for genetic transformation in Curcuma longa L. and Curcuma manga Valeton & Zijp. Agric Nat Resour 50:276–285. https://doi.org/10.1016/j.anres.2015.08.004
Pinheiro MVM, Martins FB, da Cruz ACF, de Carvalho ACPP, Ventrella MC, Otoni WC (2014) Somatic embryogenesis in anthurium (Anthurium andraeanum cv. Eidibel) as affected by different explants. Acta Sci Agron 36:87–98. https://doi.org/10.4025/actasciagron.v36i1.16557
Raju CS, Aslam A, Shajahan A (2015) High-efficiency direct somatic embryogenesis and plant regeneration from leaf base explants of turmeric (Curcuma longa L.). Plant Cell Tissue Organ Cult 122:79–87. https://doi.org/10.1007/s11240-015-0751-1
Richwine AM, Tipton JL, Thompson GA (1995) Establishment of Aloe, Gasteria and Haworthia shoot cultures from inflorescence explants. HortScience 30:1443–1444.
Rogers SMD (1993a) Optimization of plant regeneration and rooting from leaf explants of five rare Haworthia. Sci Hortic 56:157–161. https://doi.org/10.1016/0304-4238(93)90016-J
Rogers SMD (1993b) Culture phenotype affects on regeneration capacity in the monocot Haworthia comptoniana. In Vitro Cell Dev Biol Plant 26:9–12. https://doi.org/10.1007/BF02632232
Sherif NA, Benjamin JHF, Kumar TS, Rao MV (2018) Somatic embryogenesis, acclimatization and genetic homogeneity assessment of regenerated plantlets of Anoectochilus elatus Lindl., an endangered terrestrial jewel orchid. Plant Cell Tissue Organ Cult 132:303–316. https://doi.org/10.1007/s11240-017-1330-4
Sivanesan I, Jeong BR (2012) Identification of somaclonal variants in proliferating shoot cultures of Senecio cruentus cv. Tokyo Daruma. Plant Cell Tissue Organ Cult 111:247–253. https://doi.org/10.1007/s11240-012-0186-x
Sivanesan I, Son MS, Jana S, Jeong BR (2012) Secondary somatic embryogenesis in Crocus vernus (L.) hill. Propag Ornam Plants 12:163–170
Standifer LC, O’Rourke EN, Porche-Sorbet R (1984) Propagation of Haworthia from floral scapes. Plant Prop 30:4–6
Stanišić M, Raspor M, Ninković S, Milošević S, Ćalić D, Bohanec B, Trifunović M, Petrić M, Subotić A, Jevremović S (2015) Clonal fidelity of Iris sibirica plants regenerated by somatic embryogenesis and organogenesis in leaf-base culture-RAPD and flow cytometer analyses. South Afr J Bot 96:42–52. https://doi.org/10.1016/j.sajb.2014.10.014
Sun Y, Heil BM, Kahl G, Kohlenbach HW (1987) Plant regeneration from protoplasts of the monocotyledonous Haworthia magnifica v. Poelln. Plant Cell Tissue Organ Cult 8:91–100. https://doi.org/10.1007/BF00040736
Takamori LM, Neto NBM, Vieira LGE, Ribas AF (2015) Optimization of somatic embryogenesis and in vitro plant regeneration of Urochloa species using picloram. In Vitro Cell Dev Biol Plant 51:554–563. https://doi.org/10.1007/s11627-015-9701-1
Verma VM (2017) Direct somatic embryogenesis and organogenesis from axillary meristem in taro (Colocasia esculenta var. esculenta). Am J BioSci 5:114–122. https://doi.org/10.11648/j.ajbio.20170506.13
Wang X, Li Y, Zeng H, Cai N, Qiao Z, Wang X (2017) Micropropagation of Weigela florida ‘tango’ through in vitro shoot culture. HortScience 52:274–277. https://doi.org/10.21273/HORTSCI11413-16
Wessels DC, Groenewald EG, Koeleman A (1976) Callus formation and subsequent shoot and root development from leaf tissue of Haworthia planifolia cf. var. setulifera v. Poelln. Z Pflanzenphysiol 78:141–145. https://doi.org/10.1016/S0044-328X(78)80185-8
Zhong L, Liu E, Yang C, Jin S, Diao Y, Hu Z (2017) High embryogenic ability and regeneration from floral axis of Amorphophallus konjac (Araceae). Open Life Sci 12:34–41. https://doi.org/10.1515/biol-2017-0004
Acknowledgments
This article was supported by the KU Research Professor Program of Konkuk University.
Author information
Authors and Affiliations
Contributions
DHK, KWK, and IS conceived and designed the experiments and wrote the paper. IS performed the somatic embryogenesis studies, and KWK performed the greenhouse experiments. All authors have seen and agreed to the submitted manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that there are no conflicts of interest.
Rights and permissions
About this article
Cite this article
Kim, D.H., Kang, K.W. & Sivanesan, I. Influence of auxins on somatic embryogenesis in Haworthia retusa Duval. Biologia 74, 25–33 (2019). https://doi.org/10.2478/s11756-018-0151-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-018-0151-1