Abstract
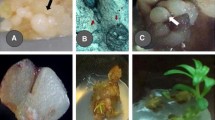
An efficient protocol for somatic embryogenesis was developed for the edible oilseed crop Guizotia abyssinica Cass. (cv. IGP 76). Induction and proliferation of callus was achieved from cotyledonary explants on MS medium amended with 1.5 mg l−1 6-benzyladenine (BA) and 0.5 mg l−1 naphthalene acetic acid (NAA). Several auxin/cytokinin combinations, physical conditions, osmotic stresses and supplementary components, viz., amino acids and casein hydrolysate were tested for induction of embryogenic callus. Higher frequency of embryogenic calli (73.7 %) and somatic embryos per culture (22) were observed on MS + 1.5 mg l−1 BA + 0.5 mg l−1 NAA + 0.5 % NaCl. Histological analyses showed typical globular and other stages of embryo development. The MS medium lacking growth regulators was best for the maximum germination of embryos (53.4 %) and formation of plantlets (65.4 %). Regenerated plantlets were acclimatized and transferred to the field with 62 % survival rate.





Similar content being viewed by others
References
Adda, S., Reddy, T. P., & Kavi Kishore, P. B. (1994). Somatic embryogenesis and organogenesis in Guizotia abyssinica. In Vitro Cellular and Developmental Biology—Plant, 30(2), 104–107.
Agarwal, S., Kanwar, K., Sharma, D. R., et al. (2004). Factors affecting secondary somatic embryogenesis and embryo maturation in Morus alba L. Scientia Horticulture, 102, 359–368.
Akula, A., Becker, D., Bateson, M., et al. (2000). High-yielding repetitive somatic embryogenesis and plant recovery in a selected tea clone, ‘TRI-2025’, by temporary immersion. Plant Cell Reports, 19, 1140–1145.
Alkhateeb, A. A. (2006). Somatic embryogenesis in date palm (Phoenix dactylifera L.) cv. Sukary in response to sucrose and polyethylene glycol. Biotechnology, 5(4), 466–470.
Andrýskova, L., Reinöhl, V., Klems, M., Prochâzka, S., et al. (2009). Long-term suspension cultures of cucumber (Cucumis sativus L.) with high embryogenic potential. Acta Physiologiae Plantarum, 31, 675–681.
Arnold, S. V., Izabela, S., Peter, B., Dyachok, J., Filonova, L., et al. (2002). Development pathway of somatic embryogenesis. Plant Cell, Tissue and Organ Culture, 69, 233–249.
Ashok Kumar, H. G., Murthy, H. N., Jadimath, V. G., Sheelavantmath, S. S., Pyati, A. N., Ravishankar, B. V., et al. (2000). Direct somatic embryogenesis and plantlet regeneration from leaf explants of Niger, Guizotia abyssinica (L. f.) Cass. Indian Journal of Experimental Biology, 38, 1073–1075.
Ashok Kumar, H. G., Murthy, H. N., et al. (2004). Effect of sugars and amino acids on androgenesis of Cucumis sativus. Plant Cell, Tissue and Organ Culture, 78, 201–208.
Awal, A., Taha, R. M., Hasbullah, et al. (2008). Induction of somatic embryogenesis and plant regeneration in Begonia x hiemalis Fotsch. in vitro. Journal of Biological Sciences, 8(5), 920–924.
Bhat, J. G., & Murthy, H. N. (2007). Factors affecting in vitro gynogenic haploid production in Niger (Guizotia abyssinica (L. f.) Cass.). Plant Growth Regulation, 52, 241–248.
Bozhkov, P. V., Filonova, L. H., von Arnold, S., et al. (2002). A key developmental switch during Norway spruce somatic embryogenesis is induced by withdrawal of growth regulators and is associated with cell death and extracellular acidification. Biotechnology Bioengineering, 77(6), 658–667.
Ceasar, S. A., & Ignacimuthu, S. (2008). Efficient somatic embryogenesis and plant regeneration from shoot apex explants of different Indian genotypes of finger millet (Eleusine coracana (L.) Gaertn.). In Vitro Cellular and Development Biology—Plant, 44, 427–435.
Ceasar, S. A., & Ignacimuthu, S. (2010). Effects of cytokinins, carbohydrates and amino acids on induction and maturation of somatic embryos in kodo millet (Paspalum scorbiculatum Linn.). Plant Cell, Tissue and Organ Culture, 102, 153–162.
Chaudhary, A., & Qu, R. (2000). Somatic embryogenesis and plant regeneration turf-type bermudagrass: Effect of 6-benzyladenine on callus induction medium. Plant Cell, Tissue and Organ Culture, 60, 113–120.
Chen, J. T., & Chang, W. C. (2000). Efficient plant regeneration through somatic embryogenesis from callus cultures of Oncidium (Orchidaceae). Plant Science, 160, 87–93.
Choi, Y. E., Yang, D. C., Choi, K. T., et al. (1998). Induction of somatic embryos by macrosalt stress from mature zygotic embryos of Panax ginseng. Plant Cell, Tissue and Organ Culture, 52, 177–181.
Chung, H. H., Chen, J. T., Chang, W. C., et al. (2005). Cytokinins induce direct somatic embryogenesis of Dendrobium Chiengmai Pink and subsequent plant regeneration. In Vitro Cellular and Developmental Biology—Plant, 41, 765–769.
Correa-Aragunde, N., Graziano, M., Chevalier, C., Lamattina, L., et al. (2006). Nitric oxide modulates the expression of cell cycle regulatory genes during lateral root formation in tomato. Journal of Experimental Botany, 57(3), 581–588.
D’onofrio, C., & Morini, S. (2002). Increasing NaCl and CaCl2 concentrations in the growth medium of quince leaves:I. Effects on somatic embryo and root regeneration. In Vitro Cellular and Development Biology—Plant, 38, 366–372.
Dixon, R. A., & Gonzales, R. A. (1994). Plant cell culture: A practical approach. New York: Oxford University Press.
Dudits, D., Gyorgyey, J., Bogre, L., & Bako, L. (1995). Molecular biology of somatic embryogenesis. In T. A. Thorpe (Ed.), In vitro embryogenesis in plants. Dordrecht: Kluwer Academic Publishers.
Ganesan, M., & Jayabala, N. (2004). Evaluation of haemoglobin (erythrogen): for improved somatic embryogenesis and plant regeneration in cotton (Gossypium hirsutum L. cv. SVPR2). Plant Cell Reports, 23, 181–187.
George, E.F. (1993). Plant propagation by tissue culture: components of culture media (pp. 313–336). London: Exegetics Limited.
Getinet, A. & Sharma, S. M. (1996). Niger [Guizotia abyssinica (L. f.) Cass]. Promoting the conservation and use of underutilized and neglected crops. Institute Plant Genetic Crop Plant Research, Gatersleben/International Plant Genetic Resource Institute, Rome.
Groll, J., Mycock, D. J., Gray, V. M., et al. (2002). Effect of medium salt concentration on differentiation and maturation of somatic embryos of Cassava (Manihot esculenyta Crantz). Annals of Botany, 89, 645–648.
Guzman, M., & Zapata, F. J. (2000). Increasing anther culture efficiency in rice (Oryza sativa) using anthers from ratooned plants. Plant Science, 151, 107–114.
Hema, B. P., & Murthy, H. N. (2008). Improvement of in vitro androgenesis in Niger using amino acids and polyamines. Biologia Plantarum, 52(1), 121–125.
Hofmann, N., Nelson, R. L., Korban, S. S., et al. (2004). Influence of media components and pH on somatic embryo induction in three genotypes of soybean. Plant Cell, Tissue and Organ Culture, 77, 157–163.
Husaini, A. M., & Abdin, M. Z. (2007). Interactive effect of light, temperature and TDZ on the regeneration potential of leaf discs of Fragaria x ananassa. In Vitro Cellular and Development Biology—Plant, 43, 576–584.
Ikeda-Iwai, M., Umehara, M., Satoh, S., Kamada, H., et al. (2003). Stress-induced somatic embryogenesis in vegetative tissues of Arabidopsis thaliana. The Plant Journal, 34, 107–114.
Jeannin, G., Bronner, R., Hahne, G., et al. (1995). Somatic embryogenesis and organogenesis induced on the immature zygotic embryo of sunflower (Helianthus annuus L.) cultivated in vitro: role of the sugar. Plant Cell Reports, 15, 200–204.
Junaid, A., Mujib, A., Sharma, M. P., Tang, W., et al. (2007). Growth regulators affect primary and secondary somatic embryogenesis in Madagaskar periwinkle (Catharanthus roseus (L.) G. Don) at morphological and biochemical levels. Plant Growth Regulation, 51, 271–281.
Kamada, H., Ishikawa, K., Saga, H., Harada, H., et al. (1993). Induction of somatic embryogenesis in carrot by osmotic stress. Plant Tissue Culture Letter, 10, 38–44.
Kandel, H., & Porter, P. (2002). Niger (Guizotia abyssinica) (L. f.) Cass. Production in northwest Minnesota. Minnesota: University of Minnesota Extension Service.
Khaleda, L., & Al-Forkan, M. (2006). Stimulatory effects of casein hydrolysate in in vitro callus induction and plant regeneration from five deepwater rice (Oryza sativa L.). Biotechnology, 5(3), 379–384.
Komatsuda, T., Lee, W., Oka, S., et al. (1992). Maturation and germination of somatic embryos as affected by sucrose and plant growth regulators in soybeans Glycine gracilis Skvortz and Glycine max (L.) Merr. Plant Cell, Tissue and Organ Culture, 28, 103–113.
Konan, N. K., Sangwan, R. S., Sangwan, B. S., et al. (1994). Somatic embryogenesis from cultured mature cotyledons of cassava (Manihot esculenta Crantz). Identification of parameters influencing the frequency of embryogenesis. Plant Cell, Tissue and Organ Culture, 37, 91–102.
Li, J., & Grabau, E. A. (1996). Comparison of somatic embryogenesis and embryo conversion in commercial soybean cultivars. Plant Cell, Tissue and Organ Culture, 44, 87–89.
Liao, Y. K., & Amerson, H. V. (1995). Slash pine (Pinus elliottii Engelm.) somatic embryogenesis II. Maturation of somatic embryos and plant regeneration. New Forests, 10, 165–182.
Lou, H., & Kako, S. (1995). Role of high sugar concentrations in inducing somatic embryogenesis from cucumber cotyledons. Scientia Horticulture, 64, 11–20.
Mandal, A. K. A., Gupta, S. D., Chatterji, A. K., et al. (2001). Factors affecting somatic embryogenesis from cotyledonary explants of safflower. Biologia Plantarum, 44(4), 503–507.
Meijer, E. G. M., & Brown, C. W. B. (1987). Role of exogenous reduced nitrogen and sucrose in rapid high frequency somatic embryogenesis in Medicago sativa. Plant Cell, Tissue and Organ Culture, 10(1), 11–19.
Meijer, E. A., de Vries, S. C., Mordhorst, A. P., et al. (1999). Co-culture with Daucus carota somatic embryos reveals high 2,4-D uptake and release rates of Arabidopsis thaliana cultured cells. Plant Cell Reports, 18(7–8), 656–663.
Muralidharan, E. M., Gupta, P. K., Mascarenhas, A. E., et al. (1989). Plantlet production through high frequency somatic embryogenesis in long term cultures of Eucalyptus citriodora. Plant Cell Reports, 8, 41–43.
Murashige, T., & Skoog, F. (1962). A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiologia Plantarum, 15, 473–497.
Naik, P. M., & Murthy, H. N. (2010). Somatic embryogenesis and plant regeneration from cell suspension culture of Niger (Guizotia abyssinica Cass.). Acta Physiologia Plantarum, 32, 75–79.
Nomura, K. (2003). Long-term conservation of embryogenic competence by induction and disorganization of somatic embryos in carrot. Plant Breeding, 122(4), 343–346.
Ramadan, M. F., & Morsel, J. T. (2002). Proximate neutral lipid composition of Niger. Czech Journal of Food Sciences, 20, 98–104.
Santarem, E. R., Pelissier, B., Finer, J. J., et al. (1997). Effect of explant orientation, pH, solidifying agent and wounding on initiation of soybean somatic embryos. In Vitro Cellular and Development Biology—Plant, 33, 13–19.
Sarreb, D. A., Ładyżyński, M., Malepszy, S., et al. (2002). Comparison of triploid and diploid cucumber in long-term liquid cultures. Plant Cell, Tissue and Organ Culture, 71, 231–235.
Sarvesh, A., Reddy, T. P., Kavikishor, P. B., et al. (1993). Embryogenesis and organogenesis in cultured anthers of an oil yielding crop Niger (Guizotia abyssinica. Cass). Plant Cell, Tissue and Organ Culture, 35, 75–80.
Sen, J., Kalia, S., Mukherjee, S. G., et al. (2002). Level of endogenous free amino acids during various stages of culture of Vigna mungo (L.) Hepper—Somatic embryogenesis, organogenesis and plant regeneration. Current Science, 82(4), 429–433.
Shahsavari, E. (2010). Evaluation and optimizations of media on the tissue culture system of upland rice. International Journal of Agriculture Biology, 12, 537–540.
Steiner, N. F., Vieira, F. N., Maldonado, S., Guerra, M. P., et al. (2005). Effect of carbon source on morphology and histodifferentiation of Araucaria angustifolia embryogenic cultures. Brazilian Archives of Biology and Technology, 43, 895–903.
Te-chato, S., & Hilae, A. (2007). High-frequency plant regeneration through secondary somatic embryogenesis in oil palm (Elaeis guineensis Jacq. var. tenera). Journal of Agriculture Technology, 3(2), 345–357.
van der Valk, P., Ruis, F., Tettelaar-Schrier, A. M., van de Velde, C. M., et al. (1995). Optimizing plant regeneration from seed-derived callus cultures of Kentucky bluegrass. The effect of benzyladenine. Plant Cell, Tissue and Organ Culture, 40, 101–103.
Van Der Vyver, C. (2010). Genetic transformation of the euploid Saccharum officinarum via direct and indirect embryogenesis. Sugar Technology, 12(1), 21–25.
Vengadesan, G., Ganapathi, A., Ramesh Anbazhagan, V., Prem Anand, R., et al. (2002). Somatic embryogenesis in cell suspension cultures of Acacia sinuata (Lour.) Merr. In Vitro Cellular Development Biology—Plant, 38, 52–57.
Vila, S. K., Rey, H. Y., Mroginski, L. A., et al. (2007). Factors affecting somatic embryogenesis induction and conversion in ‘‘Paradise Tree’’ (Melia azedarach L.). Journal Plant Growth Regulation, 26, 268–277.
Yang, J. L., Seong, E. S., Kim, M. J., Ghimire, B. K., Kang, W. H., Yu, C. Y., et al. (2010). Direct somatic embryogenesis from pericycle cells of broccoli (Brassica oleracea L. var. italica) root explants. Plant Cell, Tissue and Organ Culture, 100, 49–58.
Zavattieri, M. A., Frederico, A. M., Lima, M., Sabino, R., Arnholdt-Schmitt, B., et al. (2010). Induction of somatic embryogenesis as an example of stress-related plant reactions. European Journal of Biotechnology,. doi:10.2225/vol13-issue1-fulltext-4.
Zhang, C. L., Chen, D. F., Kubalakova, M., Zhang, J., Scott, N. W., Elliott, M. C., et al. (2008). Efficient somatic embryogenesis in sugar beet (Beta vulgaris L.) breeding lines. Plant Cell, Tissue and Organ Culture, 93, 209–221.
Acknowledgments
S.G. Ghane is thankful to University Grant Commission (UGC), New Delhi for financial assistance through Rajiv Gandhi National Fellowship at Botany Department, University of Pune. UGC-ASIST, DST-PURSE and UGC-DRS-SAP programs are duly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghane, S.G., Nikam, T.D. Influence of osmotic stress, physicochemical factors and nitrogen supplements on embryogenesis and plantlet formation in Guizotia abyssinica Cass. (Niger). Ind J Plant Physiol. 19, 263–272 (2014). https://doi.org/10.1007/s40502-014-0108-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40502-014-0108-5