Abstract
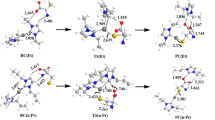
Complexes Au(DMDT)Br2 (DMDT = N,N-dimethyldithiocarbamate), Au(ESDT)Br2 (ESDT = ethylsarcosinedithiocarbamate) and Au(ESDT)Cl2 are likely to be considered as important candidates for antitumor agents. In the reactions, there is H-bond reciprocity between attacking group and H2O so that it will enhance the stability of the whole structure. On the basis of the optimized gas-phase geometry, the data of monofunctional reactions indicate that the S site of cysteine is superior to other active sites, and by analyzing the order of the activation barriers of two gold compounds with different halogen ligand in the bromoaqua (chloroaqua) substitution reaction, we discover that different halogen ligand has a slight effect on substitution reaction in the aqueous solution. Meanwhile, the cysteine as a drug target is better than purine bases. Afterward, we performed geometry optimizations in two different environments (gas phase and aqueous solution). Follow on to the bifunctional substitution reactions based on the optimized gas-phase geometry, except for the reaction when diaqua adduct [Au(ESDT)Cys(S, N)(H2O)]2+ acts as reactant, all the energy barriers of cysteine’s S as attack site are the lowest in the aqueous solution. Nevertheless, on the basis of the optimized aqueous solution geometry, our computations show that cysteine’s O site is superior to other targets in the bifunctional substitution reaction, when monofunctional adduct [Au(ESDT)Cys(S, N)(H2O)]2+ acts as reactant in the aqueous solution. Therefore, this conclusion by our calculations obtained is found to be in line with some laboratory experimental results. Besides, in the whole substitution reactions, the environmental influences should not be ignored.









Similar content being viewed by others
References
Cutillas N, Yellol GS, De Haro C, Vicente C, Rodríguez V, Ruiz J (2013) Coord Chem Rev 257:2784–2797
Wheate NJ, Walker S, Craig GE, Oun R (2010) Dalton Trans 39:8113–8127
Van Rijt SH, Sadler PJ (2009) Drug Discov Today 14:1089–1097
Koch R (1890) Dtsch Med Wochenstr 16:756
Berners-Price SJ, Mirabelli CK, Johnson RK, Mattern MR, McCabe FL, Faucette LF, Sung CM, Mong SM, Sadler PJ, Crooke ST (1986) Cancer Res 46:5486–5493
Berners-Price SJ, Girard GR, Hill DT, Sutton BM, Jarrett PS, Faucette LF, Johnson RK, Mirabelli CK, Sadler PJ (1990) J Med Chem 33:1386–1392
Rackham O, Nichols SJ, Leedman PJ, Berners-Price SJ, Filipovska A (2007) Biochem Pharmacol 74:992–1002
Tiekink ERT (2008) Inflammopharmacology 16:138–142
Ott I (2009) Coord Chem Rev 253:1670–1681
Bird HA (1990) Ann Rheum Dis 49:331–336
Saccoccia F, Angelucci F, Boumis G, Brunori M, Miele AE, Williams DL, Bellelli A (2012) J Inorg Biochem 108:105–111
McCubbin QJ, Stoddart FJ, Welton T, White AJP, Williams DJ (1998) Inorg Chem 37:3753–3758
Fox OD, Drew MG, Beer PD (2000) Angew Chem Int Ed Engl 39:136–140
Berry NG, Pratt MD, Fox OD, Beer PD (2001) Supramol Chem 13:677–682
Beer PD, Berry N, Drew MGB, Fox OD, Padilla-Tosta ME, Patell S (2001) Chem Commun 2:199–200
Beer PD, Berry NG, Cowley AR, Hayes EJ, Oates EC, Wong WW (2003) Chem Commun 19:2408–2409
Zhao Y, Pérez-Segarra W, Shi Q, Wei A (2005) J Am Chem Soc 127:7328–7329
Ronconi L, Giovagnini L, Marzano C, Bettìo F, Graziani R, Pilloni G, Fregona D (2005) Inorg Chem 44:1867–1881
Cattaruzza L, Fregona D, Mongiat M, Ronconi L, Fassina A, Colombatti A, Aldinucci D (2011) Int J Cancer 128:206–215
Casini A, Hartinger C, Gabbiani C, Mini E, Dyson PJ, Keppler BK, Messori L (2008) J Inorg Biochem 102:564–575
Ronconi L, Marzano C, Zanello P, Corsini M, Miolo G, Maccà C, Trevisan A, Fregona D (2006) J Med Chem 49:1648–1657
Aldinucci D, Lorenzon D, Stefani L, Giovagnini L, Colombatti A, Fregona D (2007) Anticancer Drugs 18:323–332
Casini A, Kelter G, Gabbiani C, Cinellu MA, Minghetti G, Fregona D, Fiebig HH, Messori L (2009) J Biol Inorg Chem 14:1139–1149
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Jr, Montgomery JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Keith T, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2010) Gaussian 09 Revision C.01 ed. Gaussian, Inc., Wallingford
Becke AD (1988) Phys Rev A 38:3098–3100
Mielich B, Savin A, Stoll H, Peuss H (1989) Chem Phys Lett 157:200–206
Becke AD (1993) J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Wadt WR, Hay PJ (1985) J Chem Phys 82:284–298
Hay PJ, Wadt WR (1985) J Chem Phys 82:270–283
Hay PJ, Wadt WR (1985) J Chem Phys 82:299–310
Mennucci B, Tomasi J (1997) J Chem Phys 106:5151–5158
Mennucci B, Cances E, Tomasi J (1997) J Phys Chem B 101:10506–10517
Tomasi J, Mennucci B, Cancès E (1999) J Mol Struct: Theochem 464:211–226
Gonzalez C, Schlegel HB (1990) J Phys Chem 94:5523–5527
Zhao HL, Zhou LX (2012) Comput Theor Chem 979:22–32
Liao JZ, Zhao HL, Zhou LX (2014) Comput Theor Chem 1048:84–94
Saggioro D, Rigobello MP, Paloschi L, Folda A, Moggach SA, Parsons S, Ronconi L, Fregona D, Bindoli A (2007) Chem Biol 14:1128–1139
Deepa P, Kolandaivel P, Senthilkumar K (2013) Struct Chem 24:583–595
Li T, Gao Y, Li J, Zhou LX (2013) Struct Chem 24:2137–2148
Acknowledgments
We would like to acknowledge financial support from the National Natural Science Foundation of China (Grant No. 21271088).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liao, J., Zhou, L. Insights into the mechanism of binding of the gold(III) dithiocarbamate derivatives to cysteine or DNA purine bases. Struct Chem 27, 651–662 (2016). https://doi.org/10.1007/s11224-015-0600-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-015-0600-1