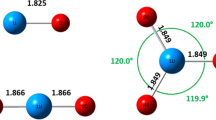
Within the framework of multi-configurational self-consistent field (MCSCF) approximation, the ground and excited states of the N4(D2H) cluster of N2 molecules are modeled from different initial states. The potential barriers to the cluster formation and dissociation are calculated. Investigations of the N4(D2H) electronic spectrum demonstrate that the most promising method of forming nitrogen nanostructures is compression of the excited triplet N2 molecules.
Similar content being viewed by others
References
Chemical Encyclopedia, Vol. 1 [in Russian], Sovetskaya Entsiklopediya, Moscow (1988).
V. F. Elesin, N. N. Degtyarenko, and L. A. Openov, Inzh. Fiz., 3, 2 (2002).
A. K. MacMahan and R. LeSar, Phys. Rev. Lett., 54, 1929 (1985).
M. I. Eremets et al., Nature Mater., 3, 558 (2004).
E. Gregoryanz, A. F. Goncharov, R. J. Hemley, and H.-K. Mao, Phys. Rev., B64, 52103 (2001).
F. Cacace, G. de Petris, and A. Troiani, Science, 295, 480 (2002).
J. P. Zheng, J. Waluk, J. Spanget-Larsen, et al., Chem. Phys. Lett., 328, 227 (2000).
M. B. Gimarc and Ming Zhao, Inorg. Chem., 35, 3289 (1996).
A. A. Korkin et al., J. Phys. Chem., 100, 5702 (1996).
M. Bitterova, H. Ostmark, and N. Brinck, J. Chem. Phys. Lett., 347, 220 (2001).
T. J. Lee and J. E. Rice, J. Chem. Phys., 94, 1215 (1991).
O. N. Temkin, Sorosovskii Obraz. Zh., 10, 98 (1997).
B. M. Smirnov and A. S. Yatsenko, Usp. Fiz. Nauk, 166, 225 (1996).
A. V. Nemukhin, B. L. Grigorenko, and A. A. Granovsky, Moscow University Chemistry Bulletin, 45, 75 (2004).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Izvestiya Vysshikh Uchebnykh Zavedenii, Fizika, No. 11, pp. 103‒112, November, 2009.
Rights and permissions
About this article
Cite this article
Elesin, V.F., Degtyarenko, N.N., Pazhitnykh, K.S. et al. Modeling of synthesis and dissociation of the N4 nitrogen cluster of D2H symmetry. Russ Phys J 52, 1224–1234 (2009). https://doi.org/10.1007/s11182-010-9362-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11182-010-9362-9