Abstract
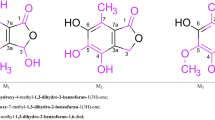
New sterically hindered catecholaldimines derived from hydrazine were synthesized in high yields. Two procedures for the synthesis of sterically hindered catecholaldimines were developed: by the reaction of 4,6-di-tert-butyl-2,3-dihydroxybenzaldehyde with hydrazines (hydrazine hydrate, phenyl-, 1,1-diphenyl-, and (diphenylmethylene)hydrazines, acetyl and benzoyl hydrazides) or by the reaction of 3,5-di-tert-butyl-6-(hydrazonomethyl)catechol with a carbonyl compound (the reaction with 4-N,N-dimethylaminobenzaldehyde). The X-ray diffraction analysis and IR, UV, and NMR spectroscopic studies showed that these compounds exist in the catecholic form both in solution and the crystalline state.
Similar content being viewed by others
References
V. Lyaskovskyy, B. de Bruin, ACS Catalysis, 2012, 270.
O. R. Luca, R. H. Crabtree, Chem. Soc. Rev., 2013, 42, 1440.
W. Kaim, B. Schwederski, Coord. Chem. Rev., 2010, 254, 1580.
I. S. Belostotskaya, E. V. Dzhuaryan, V. V. Ershov, Izv. Akad. Nauk SSSR, Ser. Khim., 1973, 2808 [Bull. Acad. Sci. USSR, Div. Chem. Sci. (Engl. Transl.), 1973, 22].
T. I. Prokof’eva, V. B. Vol’eva, A. I. Prokof’ev, I. S. Belostotskaya, N. L. Komissarov, V. V. Ershov, Chem. Heterocycl. Compd. (Engl. Transl.), 2000, 36, 923 [Khim. Geterotsikl. Soedin., 2000, 1057].
A. I. Prokof’ev, V. B. Vol’eva, I. A. Novikova, I. S. Belostotskaya, V. V. Ershov, M. I. Kabachnik, Bull. Acad. Sci. USSR, Div. Chem. Sci. (Engl. Transl.), 1980, 29, 1883 [Izv. Akad. Nauk SSSR, Ser. Khim., 1980, 2707].
M. P. Shurygina, N. O. Druzhkov, M. V. Arsen’ev, M. P. Bubnov, G. K. Fukin, S. A. Chesnokov, V. K. Cherkasov, Russ. J. Org. Chem., 2011, 47, 486 [Zh. Org. Khim., 2011, 47, 490].
G. A. Abakumov, V. K. Cherkasov, T. N. Kocherova, N. O. Druzhkov, Yu. A. Kurskii, L. G. Abakumova, Russ. Chem. Bull. (Int. Ed.), 2006, 55, 1195 [Izv. Akad. Nauk, Ser. Khim., 2006, 1151].
G. A. Abakumov, V. K. Cherkasov, T. N. Kocherova, N. O. Druzhkov, Yu. A. Kurskii, M. P. Bubnov, G. K. Fukin, L. G. Abakumova, Russ. Chem. Bull. (Int. Ed.), 2007, 56, 1849 [Izv. Akad. Nauk, Ser. Khim., 2007, 1786].
G. Poneti, M. Mannini, B. Cortigiani, L. Poggini, L. Sorace, E. Otero, P. Sainctavit, R. Sessoli, A. Dei, Inorg. Chem., 2013, 11798.
G. A. Abakumov, V. I. Nevodchikov, N. V. Zaitova, N. O. Druzhkov, L. G. Abakumova, Y. A. Kurskii, V. K. Cherkasov, Russ. Chem. Bull. (Engl. Transl.), 1997, 46, 337 [Izv. Akad. Nauk, Ser. Khim., 1997, 351].
G. A. Abakumov, V. K. Cherkasov, L. G. Abakumova, V. I. Nevodchikov, N. O. Druzhkov, N. P. Makarenko, Y. A. Kursky, J. Organomet. Chem., 1995, 491, 127.
M. Arsenyev, E. Baranov, S. Chesnokov, G. Abakumov, Acta Crystallogr., Sec. E, 2013, E69, o1565.
M. V. Arsenyev, E. V. Baranov, S. A. Chesnokov, V. K. Cherkasov, G. A. Abakumov, Russ. Chem. Bull. (Int. Ed.), 2013, 62, 2394 [Izv. Akad. Nauk, Ser. Khim., 2013, 2394].
R. M. Haak, A. Decortes, E. C. Escudero-Adan, M. M. Belmonte, E. Martin, J. Benet-Buchholz, A. W. Kleij, Inorg. Chem., 2011, 50, 7934.
L. Salmon, P. Thuery, E. Riviere, M. Ephritikhine, Inorg. Chem., 2006, 45, 83
M. Albrecht, Y. Shang, T. Rhyssen, J. Stubenrauch, H. D. F. Winkler, C. A. Schalley, Eur. J. Org. Chem., 2012, 2422.
M. Albrecht, Y. Shang, K. Hasui, V. Gossen, G. Raabe, K. Tahara, Y. Tobe, Dalton Trans., 2012, 41, 9316.
M. Albrecht, Y. Liu, S. S. Zhu, C. A. Schalley, R. Frohlich, Chem. Commun., 2009, 48, 1195.
M. Furutachi, Z. Chen, S. Matsunaga, M. Shibasaki, Molecules, 2010, 15, 532.
M. V. Arsenyev, E. V. Baranov, A. Yu. Fedorov, S. A. Chesnokov, G. A. Abakumov, Mendeleev Commun., 2015, 25, 312.
A. J. Gordon, R. A. Ford, The Chemist’s Companion, A. Handbook of Practical Data, Techniques, and References, Wiley-Interscience, New York, 1972.
Bruker (2003) SAINTPlus Data Reduction and Correction Program, v. 6.45a, Bruker AXS, Madison, Wisconsin, USA.
Data Collection, Reduction and Correction Program, CrysAlis Pro–Software Package, Agilent Technologies, 2012.
G. M. Sheldrick, SHELXTL v. 6.14, Structure Determination Software Suite, Bruker AXS, Madison, Wisconsin, USA, 2003.
G. M. Sheldrick, SADABS v. 2.01, Bruker/Siemens Area Detector Absorption Correction Program, Bruker AXS, Madison, Wisconsin, USA, 1998a.
SCALE3 ABSPACK: Empirical Absorption Correction, CrysAlis Pro–Software Package, Agilent Technologies, 2012.
Author information
Authors and Affiliations
Corresponding author
Additional information
Based on the materials of the International Conference “Organometallic and Coordination Chemistry: Achievements and Challenges” (VI Razuvaev Lectures; September 18–23, 2015, Nizhny Novgorod).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 7, pp. 1805–1813, July, 2016.
Rights and permissions
About this article
Cite this article
Arsenyev, M.V., Khamaletdinova, N.M., Baranov, E.V. et al. Synthesis, structures, and properties of new sterically hindered hydrazine-based catecholaldimines. Russ Chem Bull 65, 1805–1813 (2016). https://doi.org/10.1007/s11172-016-1514-9
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-016-1514-9