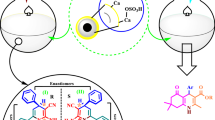
Abstract
An eco-friendly and efficient green protocol is developed for the synthesis of sixteen pyrazolo-phthalazine derivatives (5a–p) by using inexpensive biodegradable eggshell powder (ESP) as a heterogeneous catalyst. The four-component one-pot condensation reaction proceeded through Knoevenagel–Michael reaction of different chosen active methylene compounds, phthalic anhydride, and hydrazine hydride with various substituted aromatic aldehydes in the water at 60 °C, and furnished the high yields of products (93–98%) in rapid reaction time of 28 to 45 min. The material was characterized by different analytical techniques (SEM, TEM, XRD, BET, and FT-IR), and was composed of the high percentage of calcium oxides and carbonates, and less percentage of Na and Mg elements (based on EDX analysis). The ESP material displayed recyclability (4 times) without any notable loss of catalytic efficacy. Besides, this procedure offers 98% of the atom economy and 100% of carbon efficiency together with significant fiscal and enviro-friendly benefits.
Graphic abstract








Similar content being viewed by others
References
S.S. Chavan, M.S. Degani, Green Chem. 14, 296 (2012)
A. Kumar, M.K. Gupta, M. Kumar, Green Chem. 14, 290 (2012)
Q. Zhang, H. Su, J. Luo, Y.Y. Wei, Green Chem. 14, 201 (2012)
E. Kolvari, S. Zolfagharinia, RSC Adv. 6, 93963 (2016)
H. Faridi, A. Arabhosseini, Res. Agric. Eng. 64, 104 (2018)
A.K. Jagadeesan, J.A. Duvuru, J.S. Anuradha, S.K. Ponnusamy, V.A. Kabali, N. Gopakumaran, K.R.N. Selvaraj, K. Thangavelu, S. Sunny, P.P. Somasundaram, Y. Devarajan, J. Clean. Prod. 237, 117691 (2019)
J.A. Kumar, D.J. Amarnath, P.S. Kumar, C.S. Kaushik, M.E. Varghese, A. Saravanan, Desalin. Water Treat. 106, 238 (2018)
Y. Guo, Y. Sun, D.P. Yang, J. Dai, Z. Liu, Y. Chen, J. Huang, Q. Li, A.C.S. Appl, Mater. Interfaces 12, 2469 (2020)
Q. Ding, Z. Kang, L. Cao, M. Lin, H. Lin, D.P. Yang, Appl. Surf. Sci. 510, 145526 (2020)
X. Zhang, J. Huang, Z. Kang, D.P. Yang, R. Lupue, Mol. Catal. 484, 110786 (2020)
X. Zhang, M. Liu, Z. Kang, B. Wang, B. Wang, F. Jiang, X. Wang, D.P. Yang, R. Luque, Chem. Eng. J. 388, 124304 (2020)
A. Laca, A. Laca, M. Diaz, J. Environ. Manag. 197, 351 (2017)
M. Balaz, Acta Biomater. 10, 3827 (2014)
M. Nasrollahzadeh, S.M. Sajadi, A. Hatamifard, Appl. Catal. B Environ. 191, 209 (2016)
F.D. Kopinke, A. Georgi, K.U. Goss, Water Res. 129, 520 (2018)
H.N. Tran, S.J. You, A.H. Bandegharaei, H.P. Chao, Water Res. 120, 88 (2017)
S.B. Chavan, R.R. Kumbhar, D. Madhu, B. Singh, Y.C. Sharma, RSC Adv. 5, 63596 (2015)
S. Niju, K.M. Meera, S. Begum, N. Anantharaman, J. Saudi Chem. Soc. 18, 702 (2014)
A. Montilla, M.D. del Castillo, M.L. Sanz, A. Olano, Food Chem. 90, 883 (2005)
S.M. Fan, X.Z. Yuan, L. Zhao, L.H. Xu, T.J. Kang, H.T. Kim, Fuel 165, 397 (2016)
J. Rovensky, M. Stancikova, P. Masaryk, K. Svik, R. Istok, Int. J. Clin. Pharmacol. Res. 23, 83 (2003)
Y. Gao, C. Xu, Catal. Today 190, 107 (2012)
S. Patil, S. Jadhav, M.B. Deshmukh, J. Chem. Sci. 125, 851 (2013)
E. Mosaddegh, A. Hassankhani, Chinese. J. Catal. 35, 351 (2014)
E. Mosaddegh, Ultrason. Sonochem. 20, 1436 (2013)
E. Mosaddegh, A. Hassankhani, Catal. Commun. 33, 70 (2013)
A.V. Borhade, B.K. Uphade, A.G. Gadhave, Res. Chem. Intermed. 42, 6301 (2016)
M. Asif, Curr. Med. Chem. 19, 2984 (2012)
X. Deng-Qi, Z. Xu-Yao, W. Chao-Jie, M. Li-Ying, Z. Nan, H. Peng, S. Kun-Peng, C. Peng-Ju, G. Yi-Fei, Z. Xiao-Song, W. Cai-Feng, J. Cong-Hui, Z. Qiu-Rong, L. Hong-Min, Eur. J. Med. Chem. 85, 235 (2014)
A.G.A. El-Helby, R.R. Ayyad, H.M. Sakr, A.S. Abdelrahim, K. El-Adl, F.S. Sherbiny, I.H. Eissa, M.M. Khalifa, J. Mol. Struct. 1130, 333 (2017)
Z. Qiu-Rong, X. Deng-Qi, H. Peng, S. Kun-Peng, C. Peng-Ju, G. Yi-Fei, R. Jing-Li, S. Li-Hong, L. Hong-Min, Bioorg. Med. Chem. Lett. 24, 1236 (2014)
B. Maleki, S.B.N. Chalaki, S.S. Ashrafi, E.R. Seresht, F. Moeinpour, A. Khojastehnezh, R. Tayebe, Appl. Organomet. Chem. 29, 290 (2015)
R.G. Vaghei, J. Mahmoodi, Y. Maghbooli, Appl. Organomet. Chem. 31, e3717 (2017)
P. Arora, J.K. Rajput, Appl. Organomet. Chem. 32, e4001 (2017)
E. Mosaddegh, A. Hassankhani, Catal. Commun. 71, 65 (2015)
J.S. Ghomi, H.S. Alavi, A. Ziarati, R. Teymuri, M.R. Saberi, Chem. Lett. 25, 401 (2014)
F. Mohamadpour, M.T. Maghsoodlou, R. Heydari, M. Lashkari, Res. Chem. Intermed. 42, 7841 (2016)
R.G. Vaghei, S. Noori, Z.T. Semiromi, Z. Salimi, RSC Adv. 4, 47925 (2014)
C.S. Maheswari, C. Shanmugapriya, K. Revathy, A. Lalitha, J. Nanostruct. Chem. 7, 283 (2017)
S. Patil, A. Mane, S.D. Desai, J. Iran. Chem. Soc. 16, 1665 (2019)
R. Tayebee, B. Maleki, M. Sabeti, J. Iran. Chem. Soc. 14, 1176 (2017)
M.V. Reddy, Y.T. Jeong, Tetrahedron Lett. 54, 3546 (2013)
N. Kerru, S.V.H.S. Bhaskaruni, L. Gummidi, S.N. Maddila, S. Rana, P. Singh, S.B. Jonnalagadda, Appl. Organomet. Chem. 33, e4722 (2019)
N. Kerru, S.V.H.S. Bhaskaruni, L. Gummidi, S.N. Maddila, P. Singh, S.B. Jonnalagadda, Mol. Divers. (2019)
T. Liu, Y. Zhu, X. Zhang, T. Zhang, T. Zhang, X. Li, Mater. Lett. 64, 2575 (2010)
A. Antonakos, E. Liarokapis, T. Leventouri, Biomaterials 28, 3043 (2007)
Y. Yanagisawa, S. Kashima, Surf. Sci. 454, 379 (2000)
M.J. Blesa, J.L. Miranda, R. Moliner, Vib. Spectrosc. 33, 31 (2003)
M.V. Reddy, P.C.R. Kumar, G.C.S. Reddy, C.S. Reddy, C. R. Chimie 17, 1250 (2014)
Acknowledgements
The authors are thanks to the University of KwaZulu-Natal, Durban, South Africa, for financial support and research facilities.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kerru, N., Gummidi, L., Bhaskaruni, S.V.H.S. et al. One-pot green synthesis of novel 5,10-dihydro-1H-pyrazolo[1,2-b]phthalazine derivatives with eco-friendly biodegradable eggshell powder as efficacious catalyst. Res Chem Intermed 46, 3067–3083 (2020). https://doi.org/10.1007/s11164-020-04135-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-020-04135-6