Abstract
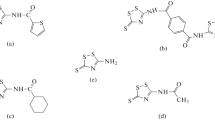
Density functional theory at the B3LYP/6-31G(d,p) basis set level was performed on three thiadiazolines, namely 4-chloro-N-(5-phenyl-1,3,4-thiadiazol-2(3H)-ylidene)aniline (TD01), 4-chloro-N-(5-(4-methoxyphenyl)-1,3,4-thiadiazol-2(3H)-ylidene)aniline (TD02), and 2-(5-(4-chlorophenylimino)-4,5-dihydro-1,3,4-thiadiazol-2-yl) phenol (TD03), and the inhibitive effect of these thiadiazolines against the corrosion of mild steel in acidic medium is elucidated. The calculated quantum chemical parameters correlated to the inhibition efficiency are EHOMO (highest occupied molecular orbital energy), ELUMO (lowest unoccupied molecular orbital energy), the energy gap (ΔE) hardness (η), softness (S), dipole moment (μ), electron affinity (EA) ionization potential (IE), the absolute electro negativity (χ), and the fraction of electron transferred (ΔN). The decreasing order of %IE of the thiadiazolines studied was found to be in agreement with experimental corrosion inhibition efficiencies. The local reactivity has been analyzed through the condensed Fukui function and local softness indices using population analysis.


Similar content being viewed by others
References
N.O. Eddy, S.A. Odoemelam, Adv. Nat. Appl. Sci. 2(1), 35 (2008)
S.A. Umoren, I.B. Obot, E.E. Ebenso, N.O. Obi-Egbedi, Int. J. Electrochem. Sci. 3, 1029 (2008)
S.S. Abd El-Rehim, M.A.M. Ibrahimand, F.F.J. Khaled, Appl. Electrochem. 29, 593 (1999)
H.L. Wang, R.B. Liu, J. Xin, Corros. Sci. 46, 2455 (2004)
S. Rengamati, S. Muralidharan, M. Anbu Kulamdainathan, S. Venkatakrishna Iyer, J. Appl. Electrochem. 24, 355 (1994)
E.E. Ebenso, Bull. Electrochem. 19, 209 (2003)
N. Khalil, Electrochim. Acta 48, 2635 (2003)
S.L. Granese, Corrosion 44, 322 (1988)
D. Wang, S. Li, Y. Ying, M. Wang, H. Xiao, Z. Chen, Corros. Sci. 41, 1911 (1999)
J. Vosta, J. Eliasek, Corros. Sci. 11, 223 (1971)
P. Choa, Q. Liang, Y. Li, Appl. Surf. Sci. 252, 1596 (2005)
Y. Xiao-Ci, Z. Hong, L. Ming-Dao, R. Hong-Xuang, Y. Lu-An, Corros. Sci. 42, 645 (2000)
F. Bentiss, M. Lebrini, M. Lagrenee, Corros. Sci. 47, 2915 (2005)
R.M. Issa, M.K. Awad, F.M. Atlam, Appl. Surf. Sci. 255, 2433 (2008)
P. Hohenberg, W. Kohn, Phys. Rev. 136B, 864 (1964)
R.G. Parr, W. Yang, Density Functional Theory of Atoms and Molecules (Oxford University Press, New York, 1989)
M.H. Cohen, in Topics in Current Chemistry, ed. by R.F. Nalewajski (Springer, Heidelberg, 1996), p. 143
R.T. Sanderson, J. Am. Chem. Soc. 74, 272 (1952)
R.G. Parr, R.A. Donnelly, M. Levy, W.E. Palke, J. Chem. Phys. 68, 801 (1978)
R.G. Parr, W. Yang, J. Am. Chem. Soc. 106, 4049 (1984)
R.G.J. Pearson, Am. Chem. Soc. 85, 3533 (1963)
J. Fang, J. Li, J. Mol. Struct. Theochem. 593, 179 (2002)
T. Arslan, F. Kandemirli, E.E. Ebenso, I. Love, H. Alemu, Corros. Sci. 51, 35 (2009)
L.M. Rodriguez-Valdez, A. Martinez-Villafane, D. Glossman-Mitnik, J. Mol. Struct. 713, 65 (2005)
M. Lebrini, M. Lagrenee, H. Vezin, L. Gengembre, F. Bentiss, Corros. Sci. 47(2), 485 (2005)
H. El Sayed, El Ashry, Ahmed El Nemr, S.A. Esawy, S. Ragab, Electrochim. Acta. 51, 3957 (2006)
F. Bentiss, M. Lebrini, M. Lagrenee, M. Traisnel, A. Elfarouk, H. Vezin, Electrochim. Acta 52(24), 6865 (2007)
A.S. Fouda, F.E. Heakal, M.S. Radwan, J. Appl. Electrochem. 39, 391 (2009)
M. Lebrini, M. Lagrenee, M. Traisnel, I. Gengembre, H. Vezin, F. Bentiss, Appl. Surf. Sci. 253, 9267 (2007)
A. Shamitha Begum, J. Mallika, P. Gayathri, E J. Chem. 7(1), 185 (2010)
M.J. Frisch, G.W. Trucks, H.B. Schlegel et al., Gaussian 03, (Gaussian, Inc., Pittsburgh, 2003)
A.D. Becke, J. Chem. Phys. 98, 5648 (1993)
C. Lee, W. Yang, R.G. Parr, Phys. Rev. B41, 785 (1988)
L. Rodriguez-Valdez, A. Martinez-Villafane, D. Glossman-Mitnik, J. Mol. Struct. (THEOCHEM). 716, 61 (2005)
M. Lashkari, M.R. Arshadi, Chem. Phys. 299, 131 (2004)
R.G. Parr, R.A. Donnelly, M. Levy, W.E. Palke, J. Chem. Phys. 68, 3801 (1978)
R.G. Parr, R.G. Pearson, J. Am. Chem. Soc. 105, 7512 (1983)
R.G. Pearson, Inorg. Chem. 27, 734 (1988)
R.G. Pearson, J. Am. Chem. Soc. 85, 3533 (1963)
H. Chermette, J. Comput. Chem. 20, 129 (1999)
N.O. Eddy, S.R. Stoyanov, E.E. Ebenso, J. Electro Chem. Sci. 5, 1127 (2010)
H. Wang, X. Wang, H. Wang, L. Wang, A. Liu, J. Mol. Model. 13, 147 (2007)
I.B. Obot, N.O. Obi-Egbedi, S.A. Umoren, Int. J. Electrochem. Sci. 4, 863 (2009)
A. Dwivedi, N. Mishra, Der. Pharm. Chem. 2, 58 (2010)
X. Li, S. Deng, H. Fu, T. Li, Electrochim. Acta 54, 4089 (2009)
P. Geerlings, F. De Proft, Int. J. Mol. Sci. 3, 276 (2002)
S.R. Stoyanov, S. Gusarov, M.S. Kuznicki, A. Kovalenko, J. Phys. Chem. 112, 6794 (2008)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Udhayakala, P., Jayanthi, A., Rajendiran, T.V. et al. Quantum chemical studies on some thiadiazolines as corrosion inhibitors for mild steel in acidic medium. Res Chem Intermed 39, 895–906 (2013). https://doi.org/10.1007/s11164-012-0603-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-012-0603-0