Abstract
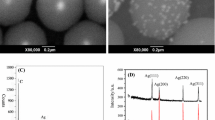
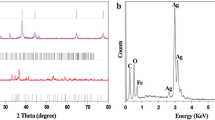
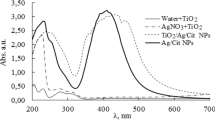
Ag nanoparticles encapsulated by TiO2 shells have the ability to catalyze redox reactions on their surface. By continually monitoring by use of UV–visible spectroscopy it was found that the surface charge of both TiO2-coated and uncoated colloidal silver particles changed after chemical electron injection. The charging and discharging process of Ag@TiO2 vary, depending on the different Ag content of the core–shell nanoparticles. In order to enhance the stability of Ag@TiO2 colloids, Fe3+ was doped into the lattice of the TiO2 shells. The experimental results showed that the Fe3+ ions have the capacity to store and transfer electrons. Furthermore, the charging and discharging rate can be controlled by changing the thickness of the TiO2 shells, because they are limited by the diffusion distance of electrons through the TiO2 shells.






Similar content being viewed by others
References
U. Kreibig, Optical Properties of Metal Clusters (Springer, Berlin Heidelberg New York, 1995)
G.C. Papavassiliou, Prog. Solid State Chem. 12, 185 (1980)
J.A. Perenboom, P. Wyder, P. Meier, Phys. Rep. 78, 173 (1981)
A.E. Hughes, S.C. Jain, Adv. Phys. 28, 717 (1979)
M. Kerker, The Scattering of Light and Other Electromagnetic Radiation (Academic Press, New York, 1969)
C.F. Bohren, Absorption and Scattering of Light by Small Particles (Wiley, New York, 1983)
J.A. Creighton, D.G. Eadon, J. Chem. Soc. Faraday Trans. 87, 3881 (1991)
P. Mulvaney, Langmuir 12, 788 (1996)
G. Schmid, Clusters and Colloids: From Theory to Application (VCH, Weinheim, 1994)
K. Foger, in Catalysis Science and Technology, vol. 6, ed. by J.R. Anderson, M. Bouchart (Springer, Berlin Heidelberg New York, 1984), p. 227
J.S. Bradley, in Clusters and Colloids. From Theory to Applications, ed by G. Schmid (VCH, Weinheim, 1994)
J. Kiwi, M. Grätzel, J. Am. Chem. Soc. 101, 7214 (1979)
T. Hirakawa, P.V. Kamat, J. Am. Chem. Soc. 127, 3928 (2005)
L. Xiao, J.L. Zhang, Y. Cong, B.Z. Tian, F. Chen, M. Anpo, Catal. Lett. 111, 207 (2006)
R.T. Tom, A.S. Nair, N. Singh, M. Aslam, C.L. Nagendra, R. Philip, K. Vijayamohanan, T. Pradeep, Langmuir 19, 3439 (2003)
J.H. Sinfelt, G.H. Via, J. Catal. 56, 1 (1979)
I. Manninger, Z. Paál, B. Tesche, U. Klengler, J. Halász, I. Kiricsi, J. Mol. Catal. 64, 361 (1991)
B.J. Tan, K.J. Klabunde, M.A. Sherwood, J. Am. Chem. Soc. 113, 855 (1991)
T. Ung, L.M. Liz-Marzán, P. Mulvaney, J. Phys. Chem. B 103, 6770 (1999)
Acknowledgments
This work was supported by the Shanghai Nanotechnology Promotion Centre (0652nm045, 0752nm001), the National Nature Science Foundation of China (20577009, 20773039), the National Basic Research Program of China (973 Program, 2007CB613306), and the Ministry of Science and Technology of China (2006AA06Z379, 2006DFA52710).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, W., Zhang, J., Chen, F. et al. Catalysis of redox reactions by Ag@TiO2 and Fe3+-doped Ag@TiO2 core–shell type nanoparticles. Res Chem Intermed 36, 163–172 (2010). https://doi.org/10.1007/s11164-010-0130-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-010-0130-9