Abstract
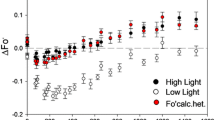
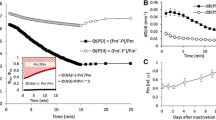
The functionality of photosystem II (PS II) following high-light pre-treatment of leaf segments at a chilling temperature was monitored as F v /F m, the ratio of variable to maximum chlorophyll fluorescence in the dark-adapted state and a measure of the optimal photochemical efficiency in PS II. Recovery of PS II functionality in low light (LL) and at a favourable temperature was retarded by (1) water stress and (2) growth in LL, in both spinach and Alocasia macrorrhiza L. In spinach leaf segments, water stress per se affected neither F v /F m nor the ability of the adenosine triphosphate (ATP) synthase to be activated by far-red light for ATP synthesis, but it induced chloroplast shrinkage as observed in frozen and fractured samples by scanning electron microscopy. A common feature of water stress and growth of plants in LL is the enhanced anchoring of PS II complexes, either across the shrunken lumen in water-stress conditions or across the partition gap in larger grana due to growth in LL. We suggest that such enhanced anchoring restricts the mobility of PS II complexes in the thylakoid membrane system, and hence hinders the lateral migration of photoinactivated PS II reaction centres to the stroma-located ribosomes for repair.






Similar content being viewed by others
Abbreviations
- ATP:
-
Adenosine triphosphate
- Chl:
-
Chlorophyll
- D1 protein:
-
psbA gene product
- EC:
-
Electrochromic signal
- Fo, Fm:
-
Chl fluorescence corresponding to open and closed PS II traps, respectively
- F v :
-
Variable Chl fluorescence
- LL, HL:
-
Low light and high light, respectively
- PS II:
-
Photosystem II
References
Albertsson P-Å (1982) Interaction between the lumenal sides of the thylakoid membrane. FEBS Lett 140:186–190. doi:10.1016/0014-5793(82)81098-3
Anderson JM, Aro E-M (1994) Granal stacking and protection of photosystem II in thylakoid membranes of higher plant leaves under sustained high irradiance: an hypothesis. Photosynth Res 41:315–326. doi:10.1007/BF00019409
Anderson JM, Park Y-I, Chow WS (1997) Photoinactivation and photoprotection of photosystem II in nature. Physiol Plant 100:214–223. doi:10.1111/j.1399-3054.1997.tb04777.x
Andersson B, Anderson JM (1980) Lateral heterogeneity in the distribution of chlorophyll-protein complexes of the thylakoid membranes of spinach chloroplasts. Biochim Biophys Acta 593:427–440. doi:10.1016/0005-2728(80)90078-X
Aro EM, Virgin I, Andersson B (1993) Photoinhibition of photosystem II. Inactivation, protein damage and turnover. Biochim Biophys Acta 1143:113–134. doi:10.1016/0005-2728(93)90134-2
Aro EM, Suorsa M, Rokka A et al (2005) Dynamics of photosystem II—a proteomic approach to thylakoid protein complexes. J Exp Bot 56:347–356. doi:10.1093/jxb/eri041
Barbato R, Friso G, Rigoni F et al (1992) Structural changes and lateral redistribution of photosystem II during donor side photoinhibition of thylakoids. J Cell Biol 119:325–335. doi:10.1083/jcb.119.2.325
Barber J (1980) Membrane surface charges and potentials in relation to photosynthesis. Biochim Biophys Acta 594:253–308
Barber J, Andersson B (1992) Too much of a good thing: light can be bad for photosynthesis. Trends Biochem Sci 17:61–66. doi:10.1016/0968-0004(92)90503-2
Bhaya D, Jagendorf AT (1984) Optimal conditions for translation by thylakoid-bound polymers from pea chloroplasts. Plant Physiol 75:832–838
Boekema EJ, van Roon H, Calkoen F et al (1999) Multiple types of association of photosystem II and its light-harvesting antenna in partially solubilized photosystem II membranes. Biochemistry 38:2233–2239. doi:10.1021/bi9827161
Bowyer JR, Packer JC, McCormack BA et al (1992) Carboxyl-terminal processing of the D1 protein and photoactivation of water-splitting in photosystem II. Partial purification and characterization of the processing enzyme from Scenedesmus obliquus and Pisum sativum. J Biol Chem 267:5424–5433
Chow WS, Aro E-M (2005) Photoinactivation and mechanisms of repair. In: Wydrzynski T, Satoh K (eds) Advances in photosynthesis and respiration, vol 22, Photosystem II: the light-driven water: plastoquinone oxidoreductase. Springer, Dordrecht, The Netherlands, pp 627–648
Chow WS, Hope AB (2004) Kinetics of reactions around the cytochrome bf complex studied in intact leaf disks. Photosynth Res 81:153–163. doi:10.1023/B:PRES.0000035027.02655.8c
Chow WS, Qian L, Goodchild DJ et al (1988) Photosynthetic acclimation of Alocasia macrorrhiza (L.) G. Don to growth irradiance: structure, function and composition of chloroplasts. Aust J Plant Physiol 15:107–122
Chow WS, Lee H-Y, He J et al (2005a) Photoinactivation of photosystem II in leaves. Photosynth Res 84:35–41. doi:10.1007/s11120-005-0410-1
Chow WS, Kim E-H, Horton P et al (2005b) Granal stacking of thylakoid membranes in higher plant chloroplasts: the physicochemical forces at work and the functional consequences that ensue. Photochem Photobiol Sci 4:1081–1090. doi:10.1039/b507310n
De Las Rivas J, Heredia P, Roman A (2007) Oxygen-evolving extrinsic proteins (PsbO, P, Q, R): bioinformatics and functional analysis. Biochim Biophys Acta 1767:575–582. doi:10.1016/j.bbabio.2007.01.018
Dekker JP, Boekema EJ (2005) Supramolecular organization of the thylakoid membrane proteins in green plants. Biochim Biophys Acta 1706:12–39. doi:10.1016/j.bbabio.2004.09.009
Demmig-Adams B, Adams WWIII (1992) Photoprotection and other responses of plants to high light stress. Annu Rev Plant Physiol Plant Mol Biol 43:599–626. doi:10.1146/annurev.pp.43.060192.003123
Ewart AJ (1896) On assimilatory inhibition in plants. J Linn Soc 31:364–461
Galmés J, Abadía A, Cifre J et al (2007) Photoprotection processes under water stress and recovery in Mediterranean plants with different growth forms and leaf habits. Physiol Plant 130:495–510. doi:10.1111/j.1399-3054.2007.00919.x
Grossman A, Bartlett S, Chua N-H (1980) Energy-dependent uptake of cytoplasmically synthesized polypeptides by chloroplasts. Nature 285:625–628. doi:10.1038/285625a0
Hankamer B, Nield J, Zhelva D et al (1997) Isolation and biochemical characterization of monomeric and dimeric photosystem II complexes from spinach and their relevance to the organization of photosystem II in vivo. Eur J Biochem 243:422–429. doi:10.1111/j.1432-1033.1997.0422a.x
He J, Chow WS (2003) The rate coefficient or repair of photosystem II after photoinactivation. Physiol Plant 118:297–304. doi:10.1034/j.1399-3054.2003.00107.x
Horton P, Ruban AV, Walter RG (1996) Regulation of light harvesting in green plants. Annu Rev Plant Physiol Plant Mol Biol 47:655–684. doi:10.1146/annurev.arplant.47.1.655
Huang CX, Canny MJ, Oates K et al (1994) Planing frozen hydrated plant specimens for SEM observation and EDX microanalysis. Microsc Res Tech 28:67–74. doi:10.1002/jemt.1070280108
Israelachvili JN, Pashley RM (1983) Molecular layering of water at surfaces and origin of repulsive hydration forces. Nature 306:249–250. doi:10.1038/306249a0
Kuroda H, Kobashi K, Kaseyama H et al (1996) Possible involvement of a low redox potential component(s) downstream of photosystem I in the translational regulation of the D1 subunit of the photosystem II reaction center in isolated pea chloroplasts. Plant Cell Physiol 37:754–761
Kyle DJ, Ohad I, Arntzen CJ (1984) Membrane protein damage and repair; selective loss of a quinone protein function in chloroplast membranes. Proc Natl Acad Sci USA 81:4070–4074. doi:10.1073/pnas.81.13.4070
Mattoo AK, Hoffman-Falk H, Marder JB et al (1984) Regulation of protein metabolism: coupling of photosynthetic electron transport to in vivo degradation of the rapidly metabolized 32-kilodalton protein of the chloroplast membranes. Proc Natl Acad Sci USA 81:1380–1384. doi:10.1073/pnas.81.5.1380
Melis A (1999) Photosystem-II damage and repair cycle in chloroplasts: what modulates the rate of photodamage in vivo? Trends Plant Sci 4:130–135. doi:10.1016/S1360-1385(99)01387-4
Murata N, Takahashi S, Nishiyama Y et al (2007) Photoinhibition of photosystem II under environmental stress. Biochim Biophys Acta 1767:414–421. doi:10.1016/j.bbabio.2006.11.019
Nir IM, Pease DC (1973) Chloroplast organization and the ultrastructural location of photosystem I and II. J Ultrastruct Res 42:534–550. doi:10.1016/S0022-5320(73)80025-5
Nishiyama Y, Yamamoto H, Allakhverdiev SI et al (2001) Oxidative stress inhibits the repair of photodamage to the photosynthetic machinery. EMBO J 20:5587–5594. doi:10.1093/emboj/20.20.5587
Nishiyama Y, Allakhverdiev SI, Yamamoto H et al (2004) Singlet oxygen inhibits the repair of photosystem II by suppressing translation elongation of the D1 protein in Synechocystis sp. PCC 6803. Biochemistry 43:11321–11330. doi:10.1021/bi036178q
Niyogi KK (2000) Safety valves for photosynthesis. Curr Opin Plant Biol 3:455–460. doi:10.1016/S1369-5266(00)00113-8
Öquist O, Huner NPA (2003) Photosynthesis of overwintering evergreen plants. Annu Rev Plant Biol 54:329–355. doi:10.1146/annurev.arplant.54.072402.115741
Prásil O, Adir N, Ohad I (1992) Dynamics of photosystem II: mechanisms of photoinhibition and recovery process. In: Barber J (ed) The photosystems: structure, function and molecular biology, vol 11. Elsevier, Amsterdam, pp 295–348
Standfuss J, van Scheltinga ACT, Lamborghini M (2005) Mechanisms of photoprotection and nonphotochemical quenching in pea light-harvesting complex at 2.5 Å resolution. EMBO J 24:919–928. doi:10.1038/sj.emboj.7600585
Takahashi S, Murata N (2005) Interruption of the Calvin cycle inhibits the repair of photosystem II from photodamage. Biochim Biophys Acta 1708:352–361. doi:10.1016/j.bbabio.2005.04.003
Takahashi S, Murata N (2008) How do environmental stresses accelerate photoinhibition? Trends Plant Sci 13:178–182. doi:10.1016/j.tplants.2008.01.005
Taniguchi M, Kuroda H, Satoh K (1993) ATP-dependent protein synthesis in isolated pea chloroplasts. FEBS Lett 317:57–61. doi:10.1016/0014-5793(93)81491-H
Trebitsh T, Danon A (2001) Translation of chloroplast psbA mRNA is regulated by signals initiated by both photosystems II and I. Proc Natl Acad Sci USA 98:12289–12294. doi:10.1073/pnas.211440698
Tyystjärvi E (2008) Photoinhibition of photosystem II and photodamage of the oxygen evolving manganese cluster. Coord Chem Rev 252(3–4):361–376
Tyystjärvi E, Ali-Yrkko K, Kettunen R et al (1992) Slow degradation of the D1 protein is related to the susceptibility of low-light-grown pumpkin plants to photoinhibition. Plant Physiol 100:1310–1317
van Gorkom HJ, Schelvis JPM (1993) Kok’s oxygen clock: what makes it tick? The structure of P680 and consequences of its oxidising power. Photosynth Res 38:297–301. doi:10.1007/BF00046753
van Wijk KJ, Eichacker L (1996) Light is required for efficient translation elongation and subsequent integration of the D1-protein into photosystem II. FEBS Lett 388:89–93. doi:10.1016/0014-5793(96)00540-6
Zhang L, Paakkarinen V, van Wijk KJ et al (2000) Biogenesis of the chloroplast-encoded D1 protein: regulation of translational elongation, insertion, and assembly into photosystem II. Plant Cell 12:1769–1781
Acknowledgements
This work was supported by an Australian Research Council (Grant DP0664719) to W.S.C. and J.B., and a JSPS Research Fellowship for Young Scientists awarded to R.O. We are grateful to Dr. Cheng X. Huang for his kind help with cryo-scanning electron microscopy, and Prof. Jan Anderson for constructive comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s11120-008-9385-z
Rights and permissions
About this article
Cite this article
Oguchi, R., Jia, H., Barber, J. et al. Recovery of photoinactivated photosystem II in leaves: retardation due to restricted mobility of photosystem II in the thylakoid membrane. Photosynth Res 98, 621–629 (2008). https://doi.org/10.1007/s11120-008-9363-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-008-9363-5