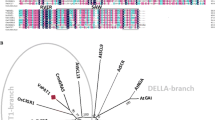
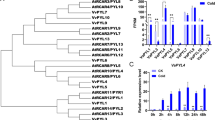
Abstract
Environmental stress has a great impact on fruit yield and quality in grapes. Understanding mechanisms underlying stress tolerance in plants is useful for grape breeding. Here, a CBF gene, designated VaCBF4, was identified in V. amurensis. The expression of VaCBF4 was induced by several abiotic stresses, including cold, exogenous abscisic acid (ABA), drought, salinity, and cold-drought conditions. A yeast one-hybrid assay demonstrated that VaCBF4 protein could bind to a conserved DRE cis-element, which contains the core sequence ACCGAC and regulates cold- and dehydration-responsive. Transgenic Arabidopsis seedlings overexpressing VaCBF4 showed enhanced tolerance to cold, drought, and salinity when compared with wild-type controls. LT50, a chilling temperature required to cause 50 % electrolyte leakage in leaves, was 4 °C lower in transgenic Arabidopsis lines than that in non-cold-acclimated wild-type seedlings. Moreover, two stress-responsive genes, AtRD29A and AtCOR47, also showed higher levels of expression in the transgenic lines than in wild-type seedlings under normal growth condition. Taken together, all these results clearly indicate that VaCBF4 is involved in the response to abiotic stresses, and it may be a good candidate gene for genetic improvement to develop stress-tolerant varieties in grapes.







Similar content being viewed by others
References
Allen MD, Yamasaki K, Ohme-Takagi M, Tateno M, Suzuki M (1998) A novel mode of DNA recognition by a β-sheet revealed by the solution structure of the GCC-box binding domain in complex with DNA. EMBO J 17:5484–5496
Benedict C, Skinner JS, Meng R, Chang Y, Bhalerao R, Huner NPA, Finn CE, Chen THH, Hurry V (2006) The CBF1-dependent low temperature signalling pathway, regulon and increase in freeze tolerance are conserved in Populus spp. Plant Cell Environ 29:1259–1272
Canella D, Gilmour SJ, Kuhn LA, Thomashow MF (2010) DNA binding by the Arabidopsis CBF1 transcription factor requires the PKKP/RAGRxKFxETRHP signature sequence. Biochim Biophys Acta 1799:454–462
Cao ZF, Li J, Chen F, Li YQ, Zhou HM, Liu Q (2001) Effect of two conserved amino acid residues on DREB1A function. Biochem Mosc 66:623–627
Champ KI, Febres VJ, Moore GA (2007) The role of CBF transcriptional activators in two Citrus species (Poncirus and Citrus) with contrasting levels of freezing tolerance. Physiol Plant 129:529–541
Choi DW, Rodriguez EM, Close TJ (2002) Barley Cbf3 gene identification, expression pattern, and map location. Plant Physiol 129:1781–1787
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Desikan R, AH-Mackerness S, Hancock JT, Neill SJ (2001) Regulation of the Arabidopsis transcriptome by oxidative stress. Plant Physiol 127:159–172
El Kayal W, Navarro M, Marque G, Keller G, Marque C, Teulieres C (2006) Expression profile of CBF-like transcriptional factor genes from Eucalyptus in response to cold. J Exp Bot 57:2455–2469
Fennell A (2004) Freezing tolerance and injury in grapevines. J Crop Improv 10:201–235
Gamboa MC, Rasmussen-Poblete S, Valenzuela PD, Krauskopf E (2007) Isolation and characterization of a cDNA encoding a CBF transcription factor from E. globulus. Plant Physiol Biochem 45:1–5
Gao MJ, Allard G, Byass L, Flanagan AM, Singh J (2002) Regulation and characterization of four CBF transcription factors from Brassica napus. Plant Mol Biol 49:459–471
Gilmour SJ, Zarka DG, Stockinger EJ, Salazar MP, Houghton JM, Thomashow MF (1998) Low temperature regulation of the Arabidopsis CBF family of AP2 transcriptional activators as an early step in cold-induced COR gene expression. Plant J 16:433–442
Gilmour SJ, Sebolt AM, Salazar MP, Everard JD, Thomashow MF (2000) Overexpression of the Arabidopsis CBF3 transcriptional activator mimics multiple biochemical changes associated with cold acclimation. Plant Physiol 124:1854–1865
Gupta N, Rathore M, Goyary D, Khare N, Anandhan S, Pande V, Ahmed Z (2012) Marker-free transgenic cucumber expressing Arabidopsis cbf1 gene confers chilling stress tolerance. Biol Plant 56:57–63
Haake V, Cook D, Riechmann JL, Pineda O, Thomashow MF, Zhang JZ (2002) Transcription factor CBF4 is a regulator of drought adaptation in Arabidopsis. Plant Physiol 130:639–648
Jia GX, Zhu ZQ, Chang FQ, Li YX (2002) Transformation of tomato with the BADH gene from Atriplex improves salt tolerance. Plant Cell Rep 21:141–146
Kasuga M, Liu Q, Miura S, Yamaguchi-Shinozaki K, Shinozaki K (1999) Improving plant drought, salt, and freezing tolerance by gene transfer of a single stress-inducible transcription factor. Nat Biotechnol 17:287–291
Kasuga M, Miura S, Shinozaki K, Yamaguchi-Shinozaki K (2004) A combination of the Arabidopsis DREB1A gene and stress-inducible rd29A promoter improved drought- and low-temperature stress tolerance in tobacco by gene transfer. Plant Cell Physiol 45:346–350
Kitashiba H, Ishizaka T, Isuzugawa K, Nishimura K, Suzuki T (2004) Expression of a sweet cherry DREB1/CBF ortholog in Arabidopsis confers salt and freezing tolerance. J Plant Physiol 161:1171–1176
Kobayashi H, Takase H, Suzuki Y, Tanzawa F, Takata R, Fujita K, Kohno M, Mochizuki M, Suzuki S, Konno T (2011) Environmental stress enhances biosynthesis of flavor precursors, S-3-(hexan-1-ol)-glutathione and S-3-(hexan-1-ol)-L-cysteine, in grapevine through glutathione S-transferase activation. J Exp Bot 62:1325–1336
Li D, Zhang Y, Hu X, Shen X, Ma L, Su Z, Wang T, Dong J (2011) Transcriptional profiling of Medicago truncatula under salt stress identified a novel CBF transcription factor MtCBF4 that plays an important role in abiotic stress responses. BMC Plant Biol 11:109–127
Liu Q, Kasuga M, Sakuma Y, Abe H, Miura S, Yamaguchi-Shinozaki K, Shinozaki K (1998) Two transcription factors, DREB1 and DREB2, with an EREBP/AP2 DNA binding domain separate two cellular signal transduction pathways in drought- and low-temperature-responsive gene expression, respectively, in Arabidopsis. Plant Cell 10:1391–1406
Medina J, Bargues M, Terol J, Perez-Alonso M, Salinas J (1999) The Arabidopsis CBF gene family is composed of three genes encoding AP2 domain-containing proteins whose expression is regulated by low temperature but not by abscisic acid or dehydration. Plant Physiol 119:463–470
Mittler R, Blumwald E (2010) Genetic engineering for modern agriculture: challenges and perspectives. Annu Rev Plant Biol 61:443–462
Navarro M, Marque G, Ayax C, Keller G, Borges JP, Marque C, Teulieres C (2009) Complementary regulation of four Eucalyptus CBF genes under various cold conditions. J Exp Bot 60:2713–2724
Qin F, Sakuma Y, Li J, Liu Q, Li YQ, Shinozaki K, Yamaguchi-Shinozaki K (2004) Cloning and functional analysis of a novel DREB1/CBF transcription factor involved in cold-responsive gene expression in Zea mays L. Plant Cell Physiol 45:1042–1052
Roy R, Purty RS, Agrawal V, Gupta SC (2006) Transformation of tomato cultivar ‘Pusa Ruby’ with bspA gene from Populus tremula for drought tolerance. Plant Cell Tissue Organ Cult 84:56–68
Sakuma Y, Liu Q, Dubouzet JG, Abe H, Shinozaki K, Yamaguchi-Shinozaki K (2002) DNA-binding specificity of the ERF/AP2 domain of Arabidopsis DREBs, transcription factors involved in dehydration- and cold-inducible gene expression. Biochem Biophys Res Commun 290:998–1009
Schmidt F, Marnef A, Cheung MK, Wilson I, Hancock J, Staiger D, Ladomery M (2010) A proteomic analysis of oligo(dT)-bound mRNP containing oxidative stress-induced Arabidopsis thaliana RNA-binding proteins ATGRP7 and ATGRP8. Mol Biol Rep 37:839–845
Shinozaki K, Yamaguchi-Shinozaki K, Seki M (2003) Regulatory network of gene expression in the drought and cold stress responses. Curr Opin Plant Biol 6:410–417
Siddiqua M, Nassuth A (2011) Vitis CBF1 and Vitis CBF4 differ in their effect on Arabidopsis abiotic stress tolerance, development and gene expression. Plant Cell Environ 34:1345–1359
Stockinger EJ, Gilmour SJ, Thomashow MF (1997) Arabidopsis thaliana CBF1 encodes an AP2 domain-containing transcriptional activator that binds to the C-repeat/DRE, a cis-acting DNA regulatory element that stimulates transcription in response to low temperature and water deficit. Proc Natl Acad Sci USA 94:1035–1040
Suzuki N, Rizhsky L, Liang H, Shuman J, Shulaev V, Mittler R (2005) Enhanced tolerance to environmental stress in transgenic plants expressing the transcriptional coactivator multiprotein bridging factor 1c. Plant Physiol 139:1313–1322
Terrier N, Glissant D, Grimplet J, Barrieu F, Abbal P, Couture C, Ageorges A, Atanassova R, Leon C, Renaudin JP, Dedaldechamp F, Romieu C, Delrot S, Hamdi S (2005) Isogene specific oligo arrays reveal multifaceted changes in gene expression during grape berry (Vitis vinifera L.) development. Planta 222:832–847
Tillett RL, Wheatley MD, Tattersall EA, Schlauch KA, Cramer GR, Cushman JC (2012) The Vitis vinifera C-repeat binding protein 4 (VvCBF4) transcriptional factor enhances freezing tolerance in wine grape. Plant Biotechnol J 10:105–124
Uemura M, Joseph RA, Steponkus PL (1995) Cold acclimation of Arabidopsis thaliana (Effect on plasma membrane lipid composition and freeze-induced lesions). Plant Physiol 109:15–30
Walworth AE, Rowland LJ, Polashock JJ, Hancock JF, Song GQ (2012) Overexpression of a blueberry-derived CBF gene enhances cold tolerance in a southern highbush blueberry cultivar. Mol Breeding 30:1313–1323
Welling A, Palva ET (2008) Involvement of CBF transcription factors in winter hardiness in birch. Plant Physiol 147:1199–1211
Wisniewski M, Norelli J, Bassett C, Artlip T, Macarisin D (2011) Ectopic expression of a novel peach (Prunus persica) CBF transcription factor in apple (Malus × domestica) results in short-day induced dormancy and increased cold hardiness. Planta 233:971–983
Xiao H, Siddiqua M, Braybrook S, Nassuth A (2006) Three grape CBF/DREB1 genes respond to low temperature, drought and abscisic acid. Plant Cell Environ 29:1410–1421
Xiao H, Tattersall EA, Siddiqua MK, Cramer GR, Nassuth A (2008) CBF4 is a unique member of the CBF transcription factor family of Vitis vinifera and Vitis riparia. Plant Cell Environ 31:1–10
Xin H, Zhu W, Wang L, Xiang Y, Fang L, Li J, Sun X, Wang N, Londo JP, Li S (2013) Genome wide transcriptional profile analysis of Vitis amurensis and Vitis vinifera in response to cold stress. PLoS ONE 8:e58740. doi:10.1371/journal.pone.0058740
Yang C, Wang Y, Liang Z, Fan P, Wu B, Yang L, Wang Y, Li S (2009) Volatiles of grape berries evaluated at the germplasm level by headspace-SPME with GC–MS. Food Chem 114:1106–1114
Yang S, Tang XF, Ma NN, Wang LY, Meng QW (2011) Heterology expression of the sweet pepper CBF3 gene confers elevated tolerance to chilling stress in transgenic tobacco. J Plant Physiol 168:1804–1812
Zhang X, Fowler SG, Cheng H, Lou Y, Rhee SY, Stockinger EJ, Thomashow MF (2004) Freezing-sensitive tomato has a functional CBF cold response pathway, but a CBF regulon that differs from that of freezing-tolerant Arabidopsis. Plant J 39:905–919
Zhao TJ, Sun S, Liu Y, Liu JM, Liu Q, Yan YB, Zhou HM (2006) Regulating the drought-responsive element (DRE)-mediated signaling pathway by synergic functions of trans-active and trans-inactive DRE binding factors in Brassica napus. J Biol Chem 281:10752–10759
Zhuang J, Yao QH, Xiong AS, Zhang J (2010) Isolation, phylogeny and expression patterns of AP2-like genes in apple (Malus × domestica Borkh). Plant Mol Biol Rep 29:209–216
Acknowledgments
This work was supported by the National Natural Science Foundation of China (NSFC Accession No.: 31130047, 31000902 and 31171931) and a CAS special grant for postgraduate research, innovation and practice. The authors thank Professor Yuepeng Han (Key Laboratory of Plant Germplasm Enhancement and Specialty Agriculture, Wuhan Botanical Garden, The Chinese Academy of Sciences, Wuhan, China) for critical review of this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
The nucleotide and deduced amino acid sequence of VaCBF4 (GenBank Accession No. KC524504). The N-terminal region (PKKR/PRAGRxKFxETHRP) is framed. The AP2 domain is underlined. The 14th V and 19th E amino acid are labeled using stars. The C-flanking motif (DSAWR) is shaded. The C-terminus LWSY motif is double-underlined. (JPEG 186 kb)
Supplementary Fig. 2
Testing the bait strain for AbA r expression. The minimal concentration of AbA needed to suppress the basal expression of both Y1HGold (pDRE-AbAi) and Y1HGold (pmDRE-AbAi) was 300 ng.mL−1, whereas for the positive control group [Y1HGold (p53-AbAi)], it was 200 ng.mL−1. (a) schematic illustration of the positions of three yeast strains. The yeast strain [Y1HGold (p53-AbAi)], Y1HGold (pDRE-AbAi) and Y1HGold (pmDRE-AbAi) were grown on SD/-Ura medium without AbA (b), with 200 ng.mL−1 AbA (c) and 300 ng.mL−1 AbA (d). (JPEG 119 kb)
Rights and permissions
About this article
Cite this article
Li, J., Wang, N., Xin, H. et al. Overexpression of VaCBF4, a Transcription Factor from Vitis amurensis, Improves Cold Tolerance Accompanying Increased Resistance to Drought and Salinity in Arabidopsis . Plant Mol Biol Rep 31, 1518–1528 (2013). https://doi.org/10.1007/s11105-013-0627-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-013-0627-7