Abstract
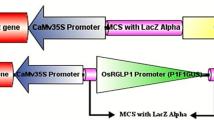
In this study, the promoter activity for three types of Euonymus-related lectins (EUL) from rice, further referred to as OrysaEULS2, OrysaEULS3, and OrysaEULD1A was analyzed. In silico promoter analyses showed that the EUL promoters from rice contain next to the typical promoter elements some motifs that are considered to be stress-responsive elements. Furthermore, Arabidopsis thaliana plants were transformed with a promoter::β-glucuronidase (GUS) construct for each of the proteins under study. Subsequently, one-insertion homozygous lines were selected and analyzed for GUS activity. Experiments were performed under normal growth conditions or after application of different stress conditions, in particular treatments with 150 mM NaCl, 100 mM mannitol, and 100 μM abscisic acid (ABA) for 24 h. GUS activity was detected with the OrysaEULS3 and OrysaEULD1A promoters especially in the cotyledons and the young true leaves, respectively, but not with the OrysaEULS2 promoter. The activity of OrysaEULS3 and OrysaEULD1A promoters was increased after ABA and mannitol treatments but decreased after NaCl treatment. We hypothesize that the Euonymus-related rice proteins have a role in sensing and responding to external stresses as well as in the growth of the plant.




Similar content being viewed by others
Abbreviations
- EUL:
-
Euonymus lectin
- GUS:
-
β-Glucuronidase
- OSR40:
-
Oryza sativa repeats 40 kDa
- TSS:
-
Transcription start site
References
Al Atalah B, Fouquaert E, Vanderschaeghe D, Proost P, Balzarini J, Smith DF, Rougé P, Lasanajak Y, Callewaert N, Van Damme EJM (2011) Expression analysis of the nucleocytoplasmic lectin ‘Orysata’ from rice in Pichia pastoris. FEBS J 278:2064–2079
Al Atalah B, Rougé P, Smith DF, Proost P, Lasanajak Y, Van Damme EJM (2012) Expression analysis of a type S2 EUL-related lectin from rice in Pichia pastoris. Glycoconj J 29:467–479
Boyer JS (1982) Plant productivity and environment. Science 218:443–448
Bray EA, Bailey-Serres J, Weretilnyk E (2000) Responses to abiotic stresses. In: Gruissem W, Buchannan B, Jones R (eds) Biochemistry and molecular biology of plants. American Society of Plant Biologists, Rockville, pp 1158–1249
Chen Y, Peumans WJ, Hause B, Bras J, Kumar M, Proost P, Barre A, Rougé P, Van Damme EJM (2002) Jasmonic acid methyl ester induces the synthesis of a cytoplasmic/nuclear chitooligosaccharide binding lectin in tobacco leaves. FASEB J 16:905–907
Clough DJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Cutler SR, Rodriguez PL, Finkelstein RR, Abrams SR (2010) Abscisic acid: emergence of a core signaling network. Annu Rev Plant Biol 61:651–679
Delporte A, Lannoo N, Vandenborre G, Ongenaert M, Van Damme EJM (2011) Jasmonate response of the Nicotiana tabacum agglutinin promoter in Arabidopsis thaliana. Plant Physiol Biochem 49:843–851
Dooki AD, Mayer-Posner FJ, Askari H, Zaiee AA, Salekdeh GH (2006) Proteomic responses of rice young panicles to salinity. Proteomics 6:6498–6507
Farooq M, Wahid A, Basra SMA, Din ID (2009) Improving water relations and gas exchange with brassinosteroids in rice under drought stress. J Agron Crop Sci 195:262–269
Fouquaert E, Peumans WJ, Smith DF, Proost P, Savvides SN, Van Damme EJM (2008) The old Euonymus europaeus agglutinin represents a novel family of ubiquitous plant proteins. Plant Physiol 147:1316–1324
Fouquaert E, Peumans WJ, Vandekerckhove TTM, Ongenaert M, Van Damme EJM (2009) Proteins with an Euonymus lectin-like domain are ubiquitous in Embryophyta. BMC Plant Biol 9:136
Fouquaert E, Van Damme EJM (2012) Promiscuity of the Euonymus carbohydrate-binding domain. Biomolecules 2:415–434
Giri J, Vij S, Dansana PK, Tyagi AK (2011) Rice A20⁄AN1 zinc-finger containing stress-associated proteins (SAP1⁄11) and a receptor-like cytoplasmic kinase (OsRLCK253) interact via A20 zinc-finger and confer abiotic stress tolerance in transgenic Arabidopsis plants. New Phytol 191:721–732
Godfray HC, Beddington JR, Crute IR, Haddad L, Lawrence D, Muir JF, Pretty J, Robinson S, Thomas SM, Toulmin C (2010) Food security: the challenge of feeding 9 billion people. Science 327:812–818
Harrison SJ, Mott EK, Parsley K, Aspinall S, Gray JC, Cottage A (2006) A rapid and robust method of identifying transformed Arabidopsis thaliana seedlings following floral dip transformation. Plant Methods 2:19
Higo K, Ugawa Y, Iwamoto M, Korenaga T (1999) Plant cis-acting regulatory DNA elements (PLACE) database. Nucleic Acids Res 27:297–300
Hirayama T, Shinozaki K (2010) Research on plant abiotic stress responses in the post-genome era: past, present and future. Plant J 61:1041–1052
Imanishi S, Kito-Nakamura K, Matsuoka K, Morikami A, Nakamura K (1997) A major jasmonate-inducible protein of sweet potato, ipomoelin, is an ABA-independent wound-inducible protein. Plant Cell Physiol 38:643–652
Jain M, Tyagi AK, Khurana JP (2006) Overexpression of putative topoisomerase 6 genes from rice confers stress tolerance in transgenic Arabidopsis plants. FEBS J 273:5245–5260
Jefferson RA (1987) Assaying chimeric genes in plants: the GUS gene fusion system. Plant Mol Biol Rep 5:387–405
Karimi M, Inzé D, Depicker A (2002) GATEWAY™ vectors for Agrobacterium mediated plant transformation. Trends Plant Sci 7:193–195
Kawasaki S, Borchert C, Deyholos M, Wang H, Brazille S, Kawai K, Galbraith D, Bohnert HJ (2001) Gene expression profiles during the initial phase of salt stress in rice. Plant Cell 13:889–905
Khurana R, Kapoor S, Tyagi AK (2013) Spatial and temporal activity of upstream regulatory regions of rice anther-specific genes in transgenic rice and Arabidopsis. Transgenic Res 22:31–46
Kuriakose B, Arun V, Gnanamanickam SS, Thomas G (2009) Tissue-specific expression in transgenic rice and Arabidopsis thaliana plants of GUS gene driven by the 50 regulatory sequences of an anther specific rice gene YY2. Plant Sci 177:390–397
Lannoo N, Peumans WJ, Van Damme EJM (2006) The presence of jasmonate-inducible lectin genes in some but not all Nicotiana species explains a marked intragenus difference in plant responses to hormone treatment. J Exp Bot 57:3145–3155
Lannoo N, Vandenborre G, Miersch O, Smagghe G, Wasternack C, Peumans WJ, Van Damme EJM (2007) The jasmonate-induced expression of the Nicotiana tabacum leaf lectin. Plant Cell Physiol 48:1207–1218
Lannoo N, Van Damme EJM (2010) Nucleocytoplasmic plant lectins. Biochim Biophys Acta 1800:190–201
Mittler R (2006) Abiotic stress, the field environment and stress combination. Trends Plant Sci 11:15–19
Moons A, Gielen J, Vandekerckhove J, Van der Straeten D, Gheysen G, Montagu MV (1997) An abscisic-acid- and salt-stress-responsive rice cDNA from a novel plant gene family. Planta 202:443–454
Morris RT, O’Connor TR, Wyrick JJ (2008) Osiris: an integrated promoter database for Oryza sativa L. Bioinformatics 24:2915–2917
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio-assays with tobacco tissue cultures. Physiol Plant 15:473–497
Nakagawa R, Yasokawa D, Okumura Y, Nagashima K (2000) Cloning and sequence analysis of cDNA coding for a lectin from Helianthus tuberosus callus and its jasmonate-induced expression. Biosci Biotechnol Biochem 64:1247–1254
Nambara E, Marion-Poll A (2005) Abscisic acid biosynthesis and catabolism. Annu Rev Plant Biol 56:165–185
Ouyang S, Zhu W, Hamilton J, Lin H, Campbell M, Childs K, Thibaud-Nissen F, Malek RL, Lee Y, Zheng L, Orvis J, Haas B, Wortman J, Buell CR (2007) The TIGR rice genome annotation resource: improvements and new features. Nucleic Acids Res 35:846–851
Peumans WJ, Van Damme EJM (1995) Lectins as plant defense proteins. Plant Physiol 109:347–352
Serraj R, Kumar A, McNally KL, Slamet-Loedin I, Bruskiewich R, Mauleon R, Cairns J, Hijmans RJ (2009) Improvement of drought resistance in rice. Adv Agron 103:41–99
Shahmuradov IA, Gammerman AJ, Hancock JM, Bramley PM, Solovyev VV (2003) PlantProm: a database of plant promoter sequences. Nucleic Acids Res 31:114–117
Shiyou L, Hongya G, Yuan X, Wang X, Ai-Min W, Lijia Q, Liu J-Y (2007) The GUS reporter-aided analysis of the promoter activities of a rice metallothionein gene reveals different regulatory regions responsible for tissue-specific and inducible expression in transgenic Arabidopsis. Transgenic Res 16:177–191
Tanaka T, Antonio BA, Kikuchi S, Matsumoto T, Nagamura Y, Numa H, Sakai H, Wu J, Itoh T, Sasaki T, Aono R, Fujii Y, Habara T, Harada E, Kanno M, Kawahara Y, Kawashima H, Kubooka H, Matsuya A, Nakaoka H, Saichi N, Sanbonmatsu R, Sato Y, Shinso Y, Suzuki M, Takeda J, Tanino M, Todokoro F, Yamaguchi K, Yamamoto N, Yamasaki C, Imanishi T, Okido T, Tada M, Ikeo K, Tateno Y, Gojobori T, Lin YC, Wei FJ, Hsing YI, Zhao Q, Han B, Kramer MR, McCombie RW, Lonsdale D, O'Donovan CC, Whitfield EJ, Apweiler R, Koyanagi KO, Khurana JP, Raghuvanshi S, Singh NK, Tyagi AK, Haberer G, Fujisawa M, Hosokawa S, Ito Y, Ikawa H, Shibata M, Yamamoto M, Bruskiewich RM, Hoen DR, Bureau TE, Namiki N, Ohyanagi H, Sakai Y, Nobushima S, Sakata K, Barrero RA, Sato Y, Souvorov A, Smith-White B, Tatusova T, An S, An G, OOta S, Fuks G, Fuks G, Messing J, Christie KR, Lieberherr D, Kim H, Zuccolo A, Wing RA, Nobuta K, Green PJ, Lu C, Meyers BC, Chaparro C, Piegu B, Panaud O, Echeverria M (2008) The rice annotation project database (RAP-DB): 2008 update. Nucleic Acids Res 36:1028–1033
Tester M, Langridge P (2010) Breeding technologies to increase crop production in a changing world. Science 327:818–822
Van Damme EJM, Peumans WJ, Barre A, Rougé P (1998) Plant lectins: a composite of several distinct families of structurally and evolutionary related proteins with diverse biological roles. Crit Rev Plant Sci 17:575–692
Van Damme EJM, Lannoo N, Fouquaert E, Peumans WJ (2004a) The identification of inducible cytoplasmic/nuclear carbohydrate binding proteins urges to develop novel concepts about the role of plant lectins. Glycoconj J 20:449–460
Van Damme EJM, Barre A, Rougé P, Peumans WJ (2004b) Cytoplasmic/nuclear plant lectins: a new story. Trends Plant Sci 9:484–489
Van Damme EJM, Lannoo N, Peumans WJ (2008) Plant lectins. Adv Bot Res 48:107–209
Van Damme EJM, Fouquaert E, Lannoo N, Vandenborre G, Schouppe D, Peumans WJ (2011) Novel concepts about the role of lectins in the plant cell. In: Wu AM (ed) The molecular immunology of complex carbohydrates-3, vol 705. Springer, New York
Vandenborre G, Miersch O, Hause B, Smagghe G, Wasternack C, Van Damme EJM (2009) Spodoptera littoralis-induced lectin expression in tobacco. Plant Cell Physiol 50:1142–1155
Wang YQ, Zhu SY, Wang Y, Zhang MY (2007) Tissue and inducible expression of a rice glutathione transporter gene promoter in transgenic Arabidopsis. Bot Stud 48:35–41
Yang Z, Lu Q, Wen X, Chen F, Lu (2012) Functional analysis of the rice rubisco activase promoter in transgenic Arabidopsis. Biochem Biophys Res Commun 418:565–570
Yong WD, Xu YY, Xu WZ, Wang X, Li N, Wu JS, Liang TB, Chong K, Xu ZH, Tan KH, Zhu ZQ (2003) Vernalization-induced flowering in wheat is mediated by a lectin-like gene VER2. Planta 217:261–270
Zeng L, Shannon MC, Grieve CM (2002) Evaluation of salt tolerance in rice genotypes by multiple agronomic parameters. Euphytica 127:235–245
Zhang W, Peumans WJ, Barre A, Astoul CH, Rovira P, Rougé P, Proost P, Truffa-Bachi P, Jalali AA, Van Damme EJM (2000) Isolation and characterization of a jacalin-related mannose-binding lectin from salt-stressed rice (Oryza sativa) plants. Planta 210:970–978
Zhu JK (2002) Salt and drought stress signal transduction in plants. Annu Rev Plant Biol 53:247–273
Acknowledgments
This work was funded primarily by the Fund for Scientific Research—Flanders (FWO grant G.0022.08), the Research Council of Ghent University (project BOF10/GOA/003), and the Hercules Foundation. Bassam Al Atalah is a recipient of a doctoral grant from the Special Research Council of Ghent University.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary file 1
(DOCX 21 kb)
Supplementary file 2
(DOCX 19 kb)
Supplementary Fig. 1
Histochemical analysis of GUS activity in Arabidopsis seedlings harboring a 35S promoter::GUS construct. Images were taken using a Nikon eclipse TE2000-e epi-fluorescence Microscope (Nikon Benelux, Brussels, Belgium) and a Leica microscope (Leica, Nussloch, Germany) (PPTX 264 kb)
Supplementary Fig. 2
Histochemical analysis of GUS activity in Arabidopsis seedlings. No GUS staining was detected for Arabidopsis plants harbouring the OrysaEULS2 promoter. Non-treated and stress-treated plant tissues did not show any GUS activity. Some representative pictures are shown for each plant stage (a). GUS staining was absent in the leaf, stem and root samples analyzed for developmental stages 4 and 5 of Arabidopsis plants expressing each of the EUL promoter constructs.. Some representative pictures are shown for Arabidopsis plants harboring the OrysaEULD1A promoter at developmental stage 5 (b). Images were taken using a Nikon eclipse TE2000-e epi-fluorescence Microscope (Nikon Benelux, Brussels, Belgium) and a Leica microscope (Leica, Nussloch, Germany) (PPTX 493 kb)
Supplementary Fig. 3
Comparative analysis of GUS activity under OrysaEULD1A promoter. GUS staining was analyzed in seedlings of plant stage 1 grown on MS (a), or after mannitol (b) and ABA (c) treatments (PPTX 1.50 MB)
Supplementary Fig. 4
Comparative analysis of GUS activity under OrysaEULS3 promoter. GUS staining was analyzed in seedlings of plant stage 2 grown on MS (a), or after mannitol (b) and ABA (c) treatments (PPTX 188 KB)
Rights and permissions
About this article
Cite this article
Al Atalah, B., Fouquaert, E. & Van Damme, E.J.M. Promoter Analysis for Three Types of EUL-Related Rice Lectins in Transgenic Arabidopsis . Plant Mol Biol Rep 31, 1315–1324 (2013). https://doi.org/10.1007/s11105-013-0611-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-013-0611-2