Abstract
Background and aims
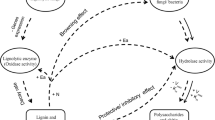
Litter decomposition is a critical process in terrestrial ecosystems and, since in natural conditions plant litter occurs in mixtures, understanding the interactive effects of mixed litter is of great ecological relevance. In this context, we test the hypothesis that N transfer between high quality litter to N-poor substrates are at the base of synergistic interactions, positively affecting litter decay rate, temperature sensitivity, and changes of organic C quality.
Methods
We carried out a manipulative experiment using four organic substrates, encompassing a wide range of biochemical quality (Hedera helix and Quercus ilex leaf litter, cellulose strips and woody sticks), each decomposing either separately or in matched pair mixtures for 360 days. Organic substrates were characterized for mass loss, C and N content and by 13C CPMAS NMR to assess biochemical quality changes.
Results
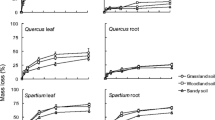
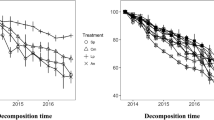
Litter response to mixing was related to the biochemical quality of the components in the mixture: additive when substrates with similarly high (H. helix and Q. ilex) or low (cellulose and wood) N content were paired, but synergistic when substrates with contrasting N content were associated (either of the two leaf litters with either cellulose or wood). Overall, no antagonist effects were observed in this experiment. Interestingly, decomposition of cellulose and wood showed an higher temperature sensitivity, compared to monospecific substrates, when paired with N rich materials. Significant N transfer was found from N rich litter to N poor substrates and 13C CPMAS NMR showed rapid changes of C quality of cellulose and wood sticks only when paired with N rich litter.
Conclusions
Our findings support the hypothesis that mixing litters of different quality, with quality expressed in terms of C/N ratio and N content, increases decomposition rate and temperature sensitivity of the lower quality substrates.





Similar content being viewed by others
References
Aerts R (1997) Climate, leaf litter chemistry and leaf litter decomposition in terrestrial ecosystems: a triangular relationship. Oikos 79:439–449
Ågren GI, Bosatta E, Magill AH (2001) Combining theory and experiment to understand effects of inorganic nitrogen on litter decomposition. Oecologia 128:94–98
Almendros G, Dorado J, González-Vila FJ, Blanco MJ, Lankes U (2000) 13C NMR assessment of decomposition patterns during composting of forest shrub biomass. Soil Biol Biochem 32:793–804
Berg B, Matzner E (1997) The effect of N deposition on the mineralization of C from plant litter and humus. Environ Rev 5:1–25
Berg B, McClaugherty C (2008) Plant litter: decomposition, humus formation and carbon sequestration, 2nd edn. Springer, Berlin
Berglund SL, Ågren GI (2012) When will litter mixtures decompose faster or slower than individual litters? A model for two litters. Oikos 121:1112–1120
Bonanomi G, Incerti G, Antignani V, Capodilupo M, Mazzoleni S (2010) Decomposition and nutrient dynamics in mixed litter of mediterranean species. Plant Soil 331:481–496
Bonanomi G, Incerti G, Barile E, Capodilupo M, Antignani V, Mingo A, Lanzotti V, Mazzoleni S (2011) Phytotoxicity, not nitrogen immobilization, explains plant litter inhibitory effects: evidence from solid-state 13C NMR spectroscopy. New Phytol 191:1018–1030
Bonanomi G, Incerti G, Giannino F, Mingo A, Lanzotti V, Mazzoleni S (2013) Litter quality assessed by solid state 13C NMR spectroscopy predicts decay rate better than C/N and Lignin/N ratios. Soil Biol Biochem 56:40–48
Carreiro MM, Sinsabaugh RL, Repert DA, Parkhurst DF (2000) Microbial enzyme shifts explain litter decay responses to simulated nitrogen deposition. Ecology 81:2359–2365
Castro-Díez P, Godoy O, Alonso A, Gallardo A, Saldaña A (2014) What explains variation in the impacts of exotic plant invasions on the nitrogen cycle? A meta-analysis. Ecol Lett 17:1–12
Chapman SK, Koch GW (2007) What type of diversity yields synergy during mixed litter decomposition in a natural forest ecosystem? Plant Soil 299:153–162
Chapman K, Whittaker JB, Heal OW (1988) Metabolic and faunal activity in litters of tree mixtures compared with pure stands. Agric Ecosyst Environ 24:33–40
Cornwell WK, Cornelissen JHC, Amatangelo K, Dorrepaal E, Eviner VT, Godoy O, Hobbie SE, Hoorens B, Kurokawa H, Pérez-Harguindeguy N, Quested HM, Santiago LS, Wardle DA, Wright IJ, Aerts R, Allison SD, van Bodegom P, Brovkin V, Chatain A, Callaghan TV, Díaz S, Garnier E, Gurvich DE, Kazakou E, Klein JA, Read J, Reich PB, Soudzilovskaia NA, Vaieretti MV, Westoby M (2008) Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecol Lett 11:1065–1071
Davidson EA, Janssens IA (2006) Temperature sensitivity of soil carbon decomposition and feedbacks to climatic change. Nature 440:165–173
Gartner TB, Cardon ZG (2004) Decomposition dynamics in mixed-species leaf litter. Oikos 104:230–246
Gessner MO (2005) Proximate lignin and cellulose. In: Graca MAS, Bärlocher F, Gessner MO (eds) Methods to study litter decomposition. A Practical Guide. Springer Verlag, The Netherlands, pp 115–120
Giardina CP, Ryan M (2000) Evidence that decomposition rates of organic carbon in mineral soil do not vary with temperature. Nature 404:858–861
Hartley IP, Ineson P (2008) Substrate quality and the temperature sensitivity of soil organic matter decomposition. Soil Biol Biochem 40:1567–1574
Hatakka A (2001) Biodegradation of lignin. In: Hofman M, Stein A (eds) Biopolymers, vol 1, Lignin, humic substances and coal. Wiley, Weinheim, pp 129–180
Hättenschwiler S, Tiunov AV, Scheu S (2005) Biodiversity and litter decomposition in terrestrial ecosystems. Ann Rev Ecol Syst 36:191–218
Hobbie SE (1996) Temperature and plant species control over litter decomposition in Alaskan tundra. Ecol Monogr 66:503–522
Hobbie SE, Eddy WC, Buyarski CR, Adair EC, Ogdahl ML, Weisenhorn P (2012) Response of decomposing litter and its microbial community to multiple forms of nitrogen enrichment. Ecol Monogr 82:389–405
Hoorens B, Aerts R, Stroetenga M (2003) Does initial litter chemistry explain litter mixture effects on decomposition? Oecologia 137:578–586
Knorr M, Frey SD, Curtis PS (2005) Nitrogen additions and litter decomposition: a meta-analysis. Ecology 86:3252–3257
Kögel-Knabner I (2002) The macromolecular organic composition of plant and microbial residues as inputs to soil organic matter. Soil Biol Biochem 34:139–162
Lecerf A, Risnoveanu G, Popescu C, Gessner MO, Chauvet E (2007) Decomposition of diverse litter mixtures in streams. Ecology 88:219–227
Lecerf A, Marie G, Kominoski JS, LeRoy CJ, Bernadet C, Swan CM (2011) Incubation time, functional litter diversity, and habitat characteristics predict litter-mixing effects on decomposition. Ecology 92:160–169
Lummer D, Scheu S, Butenschoen O (2012) Connecting litter quality, microbial community and nitrogen transfer mechanisms in decomposing litter mixtures. Oikos 121:1649–1655
Meentemeyer V (1978) Macroclimate and lignin control of litter decomposition rates. Ecology 59:465–472
Preston CM, Nault JR, Trofymow JA (2009) Chemical changes during 6 years of decomposition of 11 litters in some Canadian forest sites. Part 2. 13C abundance, solid-state 13C NMR spectroscopy and the meaning of “lignin”. Ecosystems 12:1078–1102
Schimel JP, Hättenschwiler S (2007) Nitrogen transfer between decomposing leaves of different N status. Soil Biol Biochem 39:1428–1436
Schwendener CM, Lehmann J, de Camargo PB, Luizão RC, Fernandes E (2005) Nitrogen transfer between high-and low-quality leaves on a nutrient-poor Oxisol determined by 15 N enrichment. Soil Biol Biochem 37:787–794
Tan Y, Chen J, Yan L, Huang J, Wang L, Chen S (2013) Mass loss and nutrient dynamics during litter decomposition under three mixing treatments in a typical steppe in inner Mongolia. Plant Soil 366:107–118
Taylor BR, Parkinson D, Parsons WFJ (1989) Nitrogen and lignin content as predictors of litter decay rates: a microcosm test. Ecology 70:97–104
Wardle DA, Bonner KI, Nicholson KS (1997) Biodiversity and plant litter: experimental evidence which does not support the view that enhanced species richness improves ecosystem function. Oikos 79:247–258
Wardle DA, Nilsson MC, Zackrisson O, Gallet C (2003) Determinants of litter mixing effects in a Swedish boreal forest. Soil Biol Biochem 35:827–835
Wardle DA, Yeates GW, Barker GM, Bonner KI (2006) The influence of plant litter diversity on decomposer abundance and diversity. Soil Biol Biochem 38:1052–1062
Weedon JT, Cornwell WK, Cornelissen JH, Zanne AE, Wirth C, Coomes DA (2008) Global meta-analysis of wood decomposition rates: a role for trait variation among tree species? Ecol Lett 12:45–56
Acknowledgments
The 13C-CPMAS NMR measurements were performed at the CERMANU-Interdepartmental Research Centre, University of Napoli Federico II. The work has been supported with grants by MIUR (PRIN 2005–050197 and FISR MESCOSAGR).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Juha Mikola..
Rights and permissions
About this article
Cite this article
Bonanomi, G., Capodilupo, M., Incerti, G. et al. Nitrogen transfer in litter mixture enhances decomposition rate, temperature sensitivity, and C quality changes. Plant Soil 381, 307–321 (2014). https://doi.org/10.1007/s11104-014-2119-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-014-2119-4