ABSTRACT
Purpose
To investigate the rectal absorption of vigabatrin in rats, based on the hypothesis that PAT1 (Slc36a1) is involved.
Methods
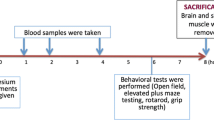
Male Sprague–Dawley rats were dosed rectally with five different gels, varying in buffer capacity, the amount of vigabatrin, and co-administration of proline or tryptophan. Western blotting was used to detect rPAT1 in rat rectal epithelium. X. Laevis oocytes were injected with SLC36A1 cRNA for the expression of hPAT1, prior to two-electrode voltage clamp measurements.
Results
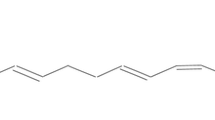
rPAT1 protein was present in rat rectal epithelium. Approximately 7%–9% of a 1 mg/kg vigabatrin dose was absorbed after rectal administration, regardless of the formulation used. Increasing the dose of vigabatrin 10-fold decreased the absolute bioavailability to 4.2%. Co-administration of proline or tryptophan changed the pharmacokinetic profile, indicating a role of PAT1 in the rectal absorption of vigabatrin. Transport of vigabatrin via hPAT1 expressed in X. Laevis oocytes had a Km of 5.2 ± 0.6 mM and was almost completely inhibited by tryptophan.
Conclusions
Although vigabatrin is a PAT1 substrate and the rPAT1 protein is expressed in the rectum epithelium, vigabatrin has low rectal absorption in rats.




Similar content being viewed by others
Abbreviations
- GABA:
-
γ-amino butyric acid
- hPAT1:
-
proton-coupled amino acid transporter human isoform
- mPAT1:
-
proton-coupled amino acid transporter murine isoform
- Pro:
-
L-proline
- rGAPDH:
-
rat glyceraldehyde 3-phosphate dehydrogenase
- rPAT1:
-
proton-coupled amino acid transporter rat isoform
- rSlc36a1 :
-
gene encoding rat solute carrier member 36a1
- SLC36A1 :
-
gene encoding human solute carrier member 36A1
- TEVC:
-
two-electrode voltage clamp
- Vig:
-
vigabatrin
REFERENCES
Willmore LJ, Abelson MB, Ben-Menachem E, Pellock JM, Shields WD. Vigabatrin: 2008 update. Epilepsia. 2009;50:163–73.
Kossoff EH. Infantile spasms. Neurologist. 2010;16:69–75.
Chiron C, Dumas C, Jambaque I, Mumford J, Dulac O. Randomized trial comparing vigabatrin and hydrocortisone in infantile spasms due to tuberous sclerosis. Epilepsy Res. 1997;26:389–95.
Grant SM, Heel RC. Vigabatrin: a review of its pharmacodynamic and pharmacokinetic properties and therapeutic potential in epilepsy and disorders of motor control. Drugs. 1991;41:889–926.
Rey E, Pons G, Olive G. Vigabatrin - clinical pharmacokinetics. Clin Pharmacokinetics. 1992;23:267–78.
Perucca E. Pharmacokinetic variability of new antiepileptic drugs at different ages. Ther Drug Monit. 2005;27:714–7.
Valdizan EM, Armijo JA. Effect of singel and multiple increasing doses of vigabatrin on brain GABA metabolism and correlation with vigabatrin plasma concentration. Biochem Pharmacol. 1992;43:2143–50.
Hoke JF, Yuh L, Antony KK, Okerholm RA, Elberfeld JM, Sussman NN. Pharmacokinetics of vigabatrin following single and multiple oral doses in normal volunteers. J Clin Pharmacol. 1993;33:458–62.
Frisk-Holmberg M, Kerth P, Meyer P. Effect of food on the absorption of vigabatrin. Br J Pharmacol. 1989;27:S23–5.
Deckers CL, Knoester PD, de Haan GJ, Keyser A, Renier WO, Hekster YA. Selection criteria for the clinical use of newer antiepileptic drugs. CNS Drugs. 2003;17:405–21.
Henczi M, Nagy J, Weaver DF. Determination of octanol-water partition coefficients by an HPLC method for anticonvulsant structure-activity studies. J Pharm Pharmacol. 1995;47:345–7.
Crowe A, Teoh YK. Limited P-glycoprotein mediated efflux for anti-epileptic drugs. J Drug Target. 2006;14:291–300.
Abbot EL, Grenade DS, Kennedy DJ, Gatfield KM, Thwaites DT. Vigabatrin transport across the human intestinal epithelial (Caco-2) brush-border membrane is via the H + -coupled amino-acid transporter hPAT1. Br J Pharmacol. 2006;147:298–306.
Grigat S, Fork C, Bach M, Golz S, Geerts A, Schomig E, Grundemann D. The carnitine transporter SLC22A5 is not a general drug transporter, but it efficiently translocates mildronate. Drug Metab Dispos. 2009;37:330–7.
Anderson CM, Grenade DS, Boll M, Foltz M, Wake KA, Munck BG, Daniel H, Ganapathy V, Thwaites DT. H+/amino acid transporter 1 (PAT1) is the imino acid carrier: an intestinal nutrient/drug transporter in human and rat. Gastroenterology. 2004;127:1410–22.
Metzner L, Brandsch M. Transport of pharmacologically active proline derivatives by the human proton-coupled amino acid transporter hPAT1. Eur J Pharn Biopharm. 2004;309:28–35.
Howard A, Goodlad RA, Walters JR, Ford D, Hirst BH. Increased expression of specific intestinal amino acid and peptide transporter mRNA in rats fed by TPN is reversed by GLP-2. J Nutr. 2004;134:2957–64.
Broberg ML, Holm R, Tønsberg H, Frølund S, Ewon KB, Nielsen AL, Brodin B, Jensen A, Kall MA, Christensen KV, Nielsen CU. Function and expression of the proton-coupled amino acid transporter Slc36a1 along the rat gastrointestinal tract: Implications for intestinal absorption of gaboxadol. Submitted Br J Pharmacol (2011).
Frolund S, Holm R, Brodin B, Nielsen CU. The proton-coupled amino acid transporter, SLC36A1 (hPAT1), transports Gly-Gly, Gly-Sar and other Gly-Gly mimetics. Br J Pharmacol. 2010;161:589–600.
Fousteri M, Vermeulen W, van Zeeland AA, Mullenders LH. Cockayne syndrome A and B proteins differentially regulate recruitment of chromatin remodeling and repair factors to stalled RNA polymerase II in vivo. Mol Cell. 2006;23:471–82.
Larsen M, Holm R, Jensen KG, Brodin B, Nielsen CU. Intestinal gaboxadol absorption via PAT1(SLC36A1): modified absorption in vivo following co-administration of L-tryptophan. Br J Pharmacol. 2009;157:1380–9.
Frolund S, Cutillas O, Larsen M, Brodin B, Nielsen CU. delta-aminolevulinic acid is a substrate for SLC36A1 (hPAT1). Br J Pharmacol. 2010;159:1339–53.
Metzner L, Dorn M, Markwardt F, Brandsch M. The orally active antihyperglycemic drug beta-guanidinopropionic acid is transported by the human proton-coupled amino acid transporter hPAT1. Mol Pharm. 2009;6:1006–11.
Chen Z, Fei YJ, Anderson CM, Wake KA, Miyauchi S, Huang W, Thwaites DT, Ganapathy V. Structure, function and immunolocalization of a proton-coupled amino acid transporter (hPAT1) in the human intestinal cell line Caco-2. J Physiol. 2003;546:349–61.
Larsen M, Holm R, Jensen KG, Sveigaard C, Brodin B, Nielsen CU. 5-Hydroxy-L-tryptophan alters gaboxadol pharmacokinetics in rats: Involvement of PAT1 and rOat1 in gaboxadol absorption and elimination. Eur J Pharm Sci. 2010;39:68–75.
Schneider MR, Dahlhoff M, Horst D, Hirschi B, Trulzsch K, Muller-Hocker J, Vogelmann R, Allgauer M, Gerhard M, Steininger S, Wolf E, Kolligs FT. A key role for E-cadherin in intestinal homeostasis and paneth cell maturation. PLoS ONE. 5 (2010).
Van Hoorde L, Braet K, Mareel M. The N-cadherin/catenin complex in colon fibroblasts and myofibroblasts. Cell Adhesion and Communication. 7:139- + (1999).
Jones M, Sabatini PJB, Lee FSH, Bendeck MP, Langille BL. N-cadherin upregulation and function in response of smooth muscle cells to arterial injury. Arterioscler Thromb Vasc Biol. 2002;22:1972–7.
Sagne C, Agulhon C, Ravassard P, Darmon M, Hamon M, El MS, Gasnier B, Giros B. Identification and characterization of a lysosomal transporter for small neutral amino acids. Proc Natl Acad Sci U S A. 2001;98:7206–11.
Boll M, Foltz M, Rubio-Aliaga I, Kottra G, Daniel H. Functional characterization of two novel mammalian electrogenic proton-dependent amino acid cotransporters. J Biol Chem. 2002;277:22966–73.
Wreden CC, Johnson J, Tran C, Seal RP, Copenhagen DR, Reimer RJ, Edwards RH. The H + -coupled electrogenic lysosomal amino acid transporter LYAAT1 localizes to the axon and plasma membrane of hippocampal neurons. J Neurosci. 2003;23:1265–75.
ACKNOWLEDGMENTS & DISCLOSURES
Kasper Gundel Jensen is acknowledged for skilful technical support with the development of the bioanalytical method and the analysis of the plasma samples. Once again, we are grateful to the staff in the animal facilities for their help and support of our research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Holm, R., Kall, M.A., Frølund, S. et al. Rectal Absorption of Vigabatrin, a Substrate of the Proton Coupled Amino Acid Transporter (PAT1, Slc36a1), in Rats. Pharm Res 29, 1134–1142 (2012). https://doi.org/10.1007/s11095-012-0673-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-012-0673-0