ABSTRACT
Purpose
To determine the lipophilicity trend line from the relationship between the blood-retinal barrier (BRB) permeability and the lipophilicity of permeants and compare it with that of the blood-brain barrier (BBB).
Methods
The retinal (RUI) and brain uptake index (BUI) of 26 radiolabeled compounds across the rat BRB and BBB, respectively, were measured using the carotid artery injection method.
Results
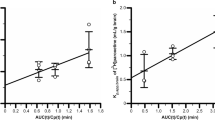
RUI was determined using 13 compounds expected to be transported from blood to the retina by passive diffusion and with a log n-octanol/Ringer distribution coefficient (DC) ranging from −2.56 to 2.48. The RUI values were correlated with the log of the DC [RUI = 46.2 × exp (0.515 × log DC), r 2 = 0.807]. A similar trend was obtained between BUI and lipophilicity. The RUI value for substrates of the influx transporters and P-glycoprotein (P-gp) was greater and smaller than the lipophilicity trend line, respectively. In contrast, [3H]verapamil, which is a substrate of P-gp, has a greater RUI value than the lipophilicity trend line, but not for BUI, suggesting that the BRB has an influx transport system for verapamil.
Conclusions
The lipophilicity trend line constructed from the RUI and DC values is considered to reflect the transport properties of drugs undergoing passive diffusion across the BRB.



Similar content being viewed by others
REFERENCES
Cunha-Vaz JG. The blood-retinal barriers system. Basic concepts and clinical evaluation. Exp Eye Res. 2004;78:715–21.
Hosoya K, Tomi M. Advances in the cell biology of transport via the inner blood-retinal barrier: establishment of cell lines and transport functions. Biol Pharm Bull. 2005;28:1–8.
Stewart PA, Tuor UI. Blood-eye barriers in the rat: correlation of ultrastructure with function. J Comp Neurol. 1994;340:566–76.
Hosoya K, Tachikawa M. Inner blood-retinal barrier transporters: role of retinal drug delivery. Pharm Res. 2009;26:2055–65.
Takata K, Kasahara T, Kasahara M, Ezaki O, Hirano H. Ultracytochemical localization of the erythrocyte/HepG2-type glucose transporter (GLUT1) in cells of the blood-retinal barrier in the rat. Invest Ophthalmol Vis Sci. 1992;33:377–83.
Ohkura Y, Akanuma S, Tachikawa M, Hosoya K. Blood-to-retina transport of biotin via Na(+)-dependent multivitamin transporter (SMVT) at the inner blood-retinal barrier. Exp Eye Res. 2010;91:387–92.
Tomi M, Mori M, Tachikawa M, Katayama K, Terasaki T, Hosoya K. L-type amino acid transporter 1-mediated L-leucine transport at the inner blood-retinal barrier. Invest Ophthalmol Vis Sci. 2005;46:2522–30.
Tomi M, Kitade N, Hirose S, Yokota N, Akanuma S, Tachikawa M, et al. Cationic amino acid transporter 1-mediated L-arginine transport at the inner blood-retinal barrier. J Neurochem. 2009;111:716–25.
Hosoya K, Makihara A, Tsujikawa Y, Yoneyama D, Mori S, Terasaki T, et al. Roles of inner blood-retinal barrier organic anion transporter 3 in the vitreous/retina-to-blood efflux transport of p-aminohippuric acid, benzylpenicillin, and 6-mercaptopurine. J Pharmacol Exp Ther. 2009;329:87–93.
Tagami M, Kusuhara S, Honda S, Tsukahara Y, Negi A. Expression of ATP-binding cassette transporters at the inner blood-retinal barrier in a neonatal mouse model of oxygen-induced retinopathy. Brain Res. 2009;1283:186–93.
Asashima T, Hori S, Ohtsuki S, Tachikawa M, Watanabe M, Mukai C, et al. ATP-binding cassette transporter G2 mediates the efflux of phototoxins on the luminal membrane of retinal capillary endothelial cells. Pharm Res. 2006;23:1235–42.
Kadam RS, Kompella UB. Influence of lipophilicity on drug partitioning into sclera, choroid-retinal pigment epithelium, retina, trabecular meshwork, and optic nerve. J Pharmacol Exp Ther. 2010;332:1107–20.
Cornford EM. The blood-brain barrier, a dynamic regulation interface. Mol Physiol. 1985;7:219–59.
Pardridge WM, Triguero D, Yang J, Cancilla PA. Comparison of in vitro and in vivo models of drug transcytosis through the blood-brain barrier. J Pharmacol Exp Ther. 1990;253:884–91.
Levin VA. Relationship of octanol/water partition coefficient and molecular weight to rat brain capillary permeability. J Med Chem. 1980;23:682–4.
Alm A, Törnquist P. The uptake index method applied to studies on the blood-retinal barrier. I. A methodological study. Acta Physiol Scand. 1981;113:73–9.
Törnquist P, Alm A. Carrier-mediated transport of amino acids through the blood-retinal and the blood-brain barriers. Graefes Arch Clin Exp Ophthalmol. 1986;224:21–5.
Pardridge WM, Fierer G. Blood-brain barrier transport of butanol and water relative to N-isopropyl-p-iodoamphetamine as the internal reference. J Cereb Blood Flow Metab. 1985;5:275–81.
Pardridge WM, Mietus LJ, Frumar AM, Davidson BJ, Judd HL. Effects of human serum on transport of testosterone and estradiol into rat brain. Am J Physiol. 1980;239:E103–8.
Crone C. The permeability of capillaries in various organs as determined by use of the “indicator diffusion” method. Acta Physiol Scand. 1963;58:292–305.
Puchowicz MA, Xu K, Magness D, Miller C, Lust WD, Kern TS, et al. Comparison of glucose influx and blood flow in retina and brain of diabetic rats. J Cereb Blood Flow Metab. 2004;24:449–57.
Pardridge WM, Mietus LJ. Transport of steroid hormones through the rat blood-brain barrier. Primary role of albumin-bound hormone. J Clin Invest. 1979;64:145–54.
Nagase K, Tomi M, Tachikawa M, Hosoya K. Functional and molecular characterization of adenosine transport at the rat inner blood-retinal barrier. Biochim Biophys Acta. 2006;1758:13–9.
Okamoto M, Akanuma S, Tachikawa M, Hosoya K. Characteristics of glycine transport across the inner blood-retinal barrier. Neurochem Int. 2009;55:789–95.
Tachikawa M, Takeda Y, Tomi M, Hosoya K. Involvement of OCTN2 in the transport of acetyl-L-carnitine across the inner blood-retinal barrier. Invest Ophthalmol Vis Sci. 2010;51:430–6.
Stoll J, Wadhwani KC, Smith QR. Identification of the cationic amino acid transporter (System y+) of the rat blood-brain barrier. J Neurochem. 1993;60:1956–9.
Boado RJ, Li JY, Nagaya M, Zhang C, Pardridge WM. Selective expression of the large neutral amino acid transporter at the blood-brain barrier. Proc Natl Acad Sci USA. 1999;96:12079–84.
Yoneyama D, Shinozaki Y, Lu WL, Tomi M, Tachikawa M, Hosoya K. Involvement of system A in the retina-to-blood transport of l-proline across the inner blood-retinal barrier. Exp Eye Res. 2010;90:507–13.
Bleeker GM, van Haeringen NJ, Maas ER, Glasius E. Selective properties of the vitreous barrier. Exp Eye Res. 1968;7:37–46.
Ennis SR, Betz AL. Sucrose permeability of the blood-retinal and blood-brain barriers. Effects of diabetes, hypertonicity, and iodate. Invest Ophthalmol Vis Sci. 1986;27:1095–102.
Pardridge WM, Boado RJ, Farrell CR. Brain-type glucose transporter (GLUT-1) is selectively localized to the blood-brain barrier. Studies with quantitative western blotting and in situ hybridization. J Biol Chem. 1990;265:18035–40.
Park S, Sinko PJ. The blood-brain barrier sodium-dependent multivitamin transporter: a molecular functional in vitro-in situ correlation. Drug Metab Dispos. 2005;33:1547–54.
Bodis-Wollner I. Visual electrophysiology in Parkinson’s disease: PERG, VEP and visual P300. Clin Electroencephalogr. 1997;28:143–7.
Bhaskar PA, Vanchilingam S, Bhaskar EA, Devaprabhu A, Ganesan RA. Effect of L-dopa on visual evoked potential in patients with Parkinson’s disease. Neurology. 1986;36:1119–21.
Kageyama T, Nakamura M, Matsuo A, Yamasaki Y, Takakura Y, Hashida M, et al. The 4F2hc/LAT1 complex transports L-DOPA across the blood-brain barrier. Brain Res. 2000;879:115–21.
Roth S, Rosenbaum PS, Osinski J, Park SS, Toledano AY, Li B, et al. Ischemia induces significant changes in purine nucleoside concentration in the retina-choroid in rats. Exp Eye Res. 1997;65:771–9.
Isakovic AJ, Abbott NJ, Redzic ZB. Brain to blood efflux transport of adenosine: blood-brain barrier studies in the rat. J Neurochem. 2004;90:272–86.
Forrest D, Reh TA, Rüsch A. Neurodevelopmental control by thyroid hormone receptors. Curr Opin Neurobiol. 2002;12:49–56.
Sugiyama D, Kusuhara H, Taniguchi H, Ishikawa S, Nozaki Y, Aburatani H, et al. Functional characterization of rat brain-specific organic anion transporter (Oatp14) at the blood-brain barrier: high affinity transporter for thyroxine. J Biol Chem. 2003;278:43489–95.
Kassem NA, Deane R, Segal MB, Chen R, Preston JE. Thyroxine (T4) transfer from CSF to choroid plexus and ventricular brain regions in rabbit: contributory role of P-glycoprotein and organic anion transporting polypeptides. Brain Res. 2007;1181:44–50.
Schinkel AH, Wagenaar E, van Deemter L, Mol CA, Borst P. Absence of the mdr1a P-Glycoprotein in mice affects tissue distribution and pharmacokinetics of dexamethasone, digoxin, and cyclosporin A. J Clin Invest. 1995;96:1698–705.
Tsuji A, Terasaki T, Takabatake Y, Tenda Y, Tamai I, Yamashima T, et al. P-glycoprotein as the drug efflux pump in primary cultured bovine brain capillary endothelial cells. Life Sci. 1995;51:1427–37.
Clarke G, O'Mahony SM, Cryan JF, Dinan TG. Verapamil in treatment resistant depression: a role for the P-glycoprotein transporter? Hum Psychopharmacol. 2009;24:217–23.
Bankstahl JP, Kuntner C, Abrahim A, Karch R, Stanek J, Wanek T, et al. Tariquidar-induced P-glycoprotein inhibition at the rat blood-brain barrier studied with (R)-11C-verapamil and PET. J Nucl Med. 2008;49:1328–35.
Han YH, Sweet DH, Hu DN, Pritchard JB. Characterization of a novel cationic drug transporter in human retinal pigment epithelial cells. J Pharmacol Exp Ther. 2001;296:450–7.
Mori S, Takanaga H, Ohtsuki S, Deguchi T, Kang YS, Hosoya K, et al. Rat organic anion transporter 3 (rOAT3) is responsible for brain-to-blood efflux of homovanillic acid at the abluminal membrane of brain capillary endothelial cells. J Cereb Blood Flow Metab. 2003;23:432–40.
Leggas M, Adachi M, Scheffer GL, Sun D, Wielinga P, Du G, et al. Mrp4 confers resistance to topotecan and protects the brain from chemotherapy. Mol Cell Biol. 2004;24:7612–21.
Hori S, Ohtsuki S, Tachikawa M, Kimura N, Kondo T, Watanabe M, et al. Functional expression of rat ABCG2 on the luminal side of brain capillaries and its enhancement by astrocyte-derived soluble factor(s). J Neurochem. 2004;90:526–36.
Smeets PH, van Aubel RA, Wouterse AC, van den Heuvel JJ, Russel FG. Contribution of multidrug resistance protein 2 (MRP2/ABCC2) to the renal excretion of p-aminohippurate (PAH) and identification of MRP4 (ABCC4) as a novel PAH transporter. J Am Soc Nephrol. 2004;15:2828–35.
Kakee A, Terasaki T, Sugiyama Y. Selective brain to blood efflux transport of para-aminohippuric acid across the blood-brain barrier: in vivo evidence by use of the brain efflux index method. J Pharmacol Exp Ther. 1997;283:1018–25.
ACKNOWLEDGEMENTS
The authors thank Dr. M. Tomi, Mr. M. Okamoto, and Miss Y. Ohkura for technical assistance. This study was supported, in part, by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (JSPS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hosoya, Ki., Yamamoto, A., Akanuma, Si. et al. Lipophilicity and Transporter Influence on Blood-Retinal Barrier Permeability: A Comparison with Blood-Brain Barrier Permeability. Pharm Res 27, 2715–2724 (2010). https://doi.org/10.1007/s11095-010-0272-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-010-0272-x