Abstract
Background and objective
The ideal delivery of radiation to the surgical cavity of brain metastases (BMs) remains the subject of debate. Risks of local failure (LF) and radiation necrosis (RN) have prompted a reappraisal of the timing and/or modality of this critical component of BM management. IORT delivered at the time of resection for BMs requiring surgery offers the potential for improved local control (LC) afforded by the elimination of delay in time to initiation of radiation following surgery, decreased uncertainty in target delineation, and the possibility of dose escalation beyond that seen in stereotactic radiosurgery (SRS). This study provides a retrospective analysis with identification of potential predictors of outcomes.
Methods
Retrospective data was collected on patients treated with IORT immediately following surgical resection of BMs at three institutions according to the approval of individual IRBs. All patients were treated with 50kV portable linear accelerator using spherical applicators ranging from 1.5 to 4.0 cm. Statistical analyses were performed using IBM SPSS with endpoints of LC, DBC, incidence of RN, and overall survival (OS) and p < 0.05 considered significant.
Results
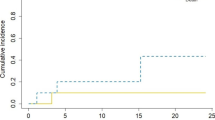
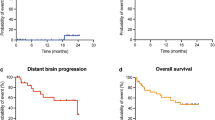
54 patients were treated with IORT with a median age of 64 years. The most common primary diagnosis was non-small cell lung cancer (40%) with the most common location in the frontal lobe (38%). Median follow-up was 7.2 months and 1-year LC, DBC, and OS were 88%, 58%, and 73%, respectively. LMD was identified in 2 patients (3%) and RN present in 4 patients (7%). The only predictor of LC was extent of resection with 1-year LC of 94% for GTR versus 62% for STR (p = 0.049).
Conclusions
IORT is a safe and effective means of delivering adjuvant radiation to the BM resection cavities with high rates of LC and low incidence of RN. Further studies are warranted directly comparing LC outcomes to SRS.



Similar content being viewed by others
References
Atalar B, Choi CY, Harsh GR et al (2013) Cavity volume dynamics after resection of brain metastases and timing of postresection cavity stereotactic radiosurgery. Neurosurgery. 72(suppl. 2):180–5; (discussion 185)
Belletti B, Vaidya JS, D'Andrea S et al (2008) Targeted intraoperative radiotherapy impairs the stimulation of breast cancer cell proliferation and invasion caused by surgical wounding. Clin Cancer Res. 14(suppl. 5):1325–1332
Brown PD, Ballman KV, Cerhan JH, et al (2017) Postoperative stereotactic radiosurgery compared with whole brain radiotherapy for resected metastatic brain disease (NCCTG N107C/CEC.3): A multicentre, randomised, controlled, phase 3 trial. Lancet Oncol;18(suppl. 8):1049–1060
Chang EL, Wefel JS, Hess KR et al (2009) Neurocognition in patients with brain metastases treated with radiosurgery or radiosurgery plus whole-brain irradiation: a randomised controlled trial. Lancet Oncol 10(suppl. 11):1037–1044
Charlton M, Schlichting J, Chioreso C, Ward M, Vikas P (2015) Challenges of rural cancer care in the united states. Oncology (Williston Park) 29(suppl. 9):633–640
Giordano FA, Brehmer S, Murle B et al (2019) Intraoperative radiotherapy in newly diagnosed glioblastoma (INTRAGO): an open-label, dose-escalation phase I/II trial. Neurosurgery 84(suppl. 1):41–49
Goodhead DT, Thacker J, Cox R (1993) Weiss lecture. effects of radiations of different qualities on cells: molecular mechanisms of damage and repair. Int J Radiat Biol ;63(suppl. 5):543–556
Herskind C, Wenz F (2014) Radiobiological aspects of intraoperative tumour-bed irradiation with low-energy X-rays (LEX-IORT). Trans Cancer Res;3(suppl. 1):3-17
Hsieh J, Elson P, Otvos B et al (2015) Tumor progression in patients receiving adjuvant whole-brain radiotherapy vs localized radiotherapy after surgical resection of brain metastases. Neurosurgery 76(suppl. 4):411–420
Iorio-Morin C, Masson-Cote L, Ezahr Y, Blanchard J, Ebacher A, Mathieu D (2014) Early gamma knife stereotactic radiosurgery to the tumor bed of resected brain metastasis for improved local control. J Neurosurg 121(Suppl):69–74
Jarvis LA, Simmons NE, Bellerive M et al (2012) Tumor bed dynamics after surgical resection of brain metastases: implications for postoperative radiosurgery. Int J Radiat Oncol Biol Phys 84(suppl. 4):943–948
Kocher M, Soffietti R, Abacioglu U et al (2011) Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952–26001 study. J Clin Oncol 29(suppl. 2):134–141
Kulcenty K, Piotrowski I, Zaleska K, et al (2019) Wound fluids collected postoperatively from patients with breast cancer induce epithelial to mesenchymal transition but intraoperative radiotherapy impairs this effect by activating the radiation-induced bystander effect. Sci Rep; 9(suppl. 1):7891
Mahajan A, Ahmed S, McAleer MF et al (2017) Post-operative stereotactic radiosurgery versus observation for completely resected brain metastases: a single-centre, randomised, controlled, phase 3 trial. Lancet Oncol. 18(suppl. 8):1040–1048
Marchan EM, Peterson J, Sio TT et al (2018) Postoperative cavity stereotactic radiosurgery for brain metastases. Front Oncol 8:342
Patchell RA, Tibbs PA, Walsh JW et al (1990) A randomized trial of surgery in the treatment of single metastases to the brain. N Engl J Med 322(suppl. 8):494–500
Patel DM, Agarwal N, Tomei KL, Hansberry DR, Goldstein IM (2015) Optimal timing of whole-brain radiation therapy following craniotomy for cerebral malignancies. World Neurosurg 84(suppl. 2):412–419
Piotrowski I, Kulcenty K, Murawa D, Suchorska W (2018) Surgical wound fluids from patients treated with intraoperative radiotherapy induce radiobiological response in breast cancer cells. Med Oncol; 36(suppl. 2):14
Soffietti R, Kocher M, Abacioglu UM et al (2013) A european organisation for research and treatment of cancer phase III trial of adjuvant whole-brain radiotherapy versus observation in patients with one to three brain metastases from solid tumors after surgical resection or radiosurgery: quality-of-life results. J Clin Oncol 31(suppl. 1):65–72
Soliman H, Ruschin M, Angelov L et al (2018) Consensus contouring guidelines for postoperative completely resected cavity stereotactic radiosurgery for brain metastases. Int J Radiat Oncol Biol Phys 100(suppl. 2):436–442
Soltys SG, Adler JR, Lipani JD et al (2008) Stereotactic radiosurgery of the postoperative resection cavity for brain metastases. Int J Radiat Oncol Biol Phys. 70(suppl. 1):187–193
Susko M, Yu Y, Ma L et al (2019) Preoperative dural contact and recurrence risk after surgical cavity stereotactic radiosurgery for brain metastases: new evidence in support of consensus guidelines. Adv Radiat Oncol 4(suppl. 3):458–465
Traylor JI, Habib A, Patel R, et al (2019) Fractionated stereotactic radiotherapy for local control of resected brain metastases. J Neurooncol; 144(2):343-350
Vargo JA, Sparks KM, Singh R, Jacobson GM, Hack JD, Cifarelli CP (2018) Feasibility of dose escalation using intraoperative radiotherapy following resection of large brain metastases compared to post-operative stereotactic radiosurgery. J Neurooncol 140(suppl. 2):413–420
Weil RJ, Mavinkurve GG, Chao ST et al (2015) Intraoperative radiotherapy to treat newly diagnosed solitary brain metastasis: initial experience and long-term outcomes. J Neurosurg 122(suppl. 4):825–832
Yusuf MB, Amsbaugh MJ, Burton E et al (2018) Increasing time to postoperative stereotactic radiation therapy for patients with resected brain metastases: Investigating clinical outcomes and identifying predictors associated with time to initiation. J Neurooncol 136(suppl. 3):545–553
Funding
Funding support was provided to CPC by the NIH/NIGMS via P20GM121322-01A1. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Author information
Authors and Affiliations
Contributions
Each of the authors performed the following tasks in submission of this manuscript: CPC—Study design, Data Collection, Statistical Analysis, Manuscript Drafting, Review/Revision of Manuscript. SB—Data Collection, Review/Revision of Manuscript. JAV—Study design, Data Collection, Statistical Analysis, Manuscript Drafting, Review/Revision of Manuscript. JDH—Data Collection, Review/Revision of Manuscript. KHK—Data Collection, Review/Revision of Manuscript. GSV—Data Collection, Review/Revision of Manuscript. FAG—Data Collection, Review/Revision of Manuscript
Corresponding author
Ethics declarations
Conflict of interest
Dr. Kahl reports personal fees from ELEKTA AB, personal fees from Varian, outside of the submitted work. Dr. Sarria reports grants and personal fees from Carl Zeiss Meditec AG, outside of the submitted work. Dr. Giordano reports grants and personal fees from Carl Zeiss Meditec AG, during the conduct of the study; grants and personal fees from NOXXON Pharma AG, grants and personal fees from ELEKTA AB, personal fees from Bristol-Myers Squibb, personal fees from Roche Pharma AG, personal fees from MSD Sharp and Dohme GmbH, personal fees from AstraZeneca GmbH, other from Implacit GmbH, non-financial support from Oncare GmbH, outside the submitted work; In addition, Dr. Giordano has a patent Radiation Therapy with Immune Response Monitoring (US 62/435405) pending to Carl Zeiss Meditec AG. All other authors have no disclosures relevant to the submitted work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cifarelli, C.P., Brehmer, S., Vargo, J.A. et al. Intraoperative radiotherapy (IORT) for surgically resected brain metastases: outcome analysis of an international cooperative study. J Neurooncol 145, 391–397 (2019). https://doi.org/10.1007/s11060-019-03309-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-019-03309-6