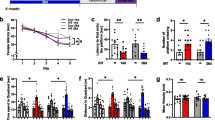
We report here studies addressing the possibility of preventing neurodegenerative changes in the brain using adaptation to periodic hypoxia in rats with experimental Alzheimer’s disease induced by administration of the neurotoxic peptide fragment of β-amyloid (Ab) into the basal magnocellular nucleus. Adaptation to periodic hypoxia was performed in a barochamber (4000 m, 4 h per day, 14 days). The following results were obtained 15 days after administration of Ab. 1. Adaptation to periodic hypoxia significantly blocked Ab-induced memory degradation in rats, as assessed by testing a conditioned passive avoidance reflex. 2. Adaptation to periodic hypoxia significantly restricted increases in oxidative stress, measured spectrophotometrically in the hippocampus in terms of the content of thiobarbituric acid-reactive secondary lipid peroxidation products. 3. Adaptation to periodic hypoxia completely prevented the overproduction of NO in the brains of rats with experimental Alzheimer’s disease, as measured in terms of increases in tissue levels of stable NO metabolites, i.e., nitrites and nitrates. 4. The cerebral cortex of rats given Ab injections after adaptation to periodic hypoxia did not contain neurons with pathomorphological changes or dead neurons (Nissl staining), which were typical in animals with experimental Alzheimer’s disease. Thus, adaptation to periodic hypoxia effectively prevented oxidative and nitrosative stress, protecting against neurodegenerative changes and protecting cognitive functions in experimental Alzheimer’s disease.
Similar content being viewed by others
References
S. O. Bachurin, “Medical chemistry of the targeted search for agents for the treatment and prevention of neurodegenerative diseases using Alzheimer’s disease as an example,” in: Neurodegenerative Diseases and Aging [in Russian], I. A. Zavalishin, N. N. Yakhno, and S. I. Gavrilova (eds.), Moscow (2001), pp. 399–454.
A. F. Vanin, “Dinitrosyl complexes of iron and S-nitrosothiols – two possible forms of stabilization and transport of nitric oxide in biological systems,” Biokhimiya, 63, No. 7, 924–938 (1998).
S. A. Elchaninova, N. A. Korenyak, I. V. Smagina, L. E. Pinegin, and B. Ya. Varshavskii, “Discontinuous hypoxia in the treatment of chronic cerebral ischemia,” Zh. Nevrol. Psikhiat. im. S. S. Korsakova, 102, 29–32 (2002).
A. L. Krushinskii, V. P. Reutov, V. S. Kuzenkov, E. G. Sorokina, V. B. Koshelev, O. E. Fadyukova, L. M. Baider, Z. V. Kuropteva, T. T. Zhumabaeva, L. Kh. Komissarova, T. V. Ryasina, N. S. Kositsyn, and V. G. Pinelis, “Nitric oxide is involved in the mechanisms of short-term adaptation to hypoxia and protective actions during the development of stress lesions in Krushinskii-Molodkina rats,” Izv. Ros. Akad. Nauk. Ser. Biol., 34, No. 3, 329–335 (2007).
V. I. Kulinskii, L. N. Minakina, and T. V. Gavrilina, “Neuroprotective effects of hypoxic preconditioning: the phenomenon and its mechanisms,” Byull. Eksperim. Biol. Med., 133, No. 2, 237–239 (2002).
E. B. Manukhina, F. Viegant,V. I. Torshin, A. V. Goryacheva, I. P. Khomenko, S. V. Kruglov, S. Yu. Mashina, D. A. Pokidyshev, E. V. Popova, M. G. Pshennikova, M. A. Vlasova, O. M. Zelenina, and I. Yu. Malyshev, “Potential non-medication-based approaches to Alzheimer’s disease,” Izv. Ros. Akad. Nauk. Ser. Biol., 31, No. 4, 382–395 (2004).
E. B. Manukhina and I. Yu. Malyshev, “The stress-limiting nitric oxide system,” Ros. Fiziol. Zh. im. I. M. Sechenova, 86, No. 10, 1283–1292 (2000).
E. B. Manukhina, B. V. Smirin, I. Yu. Malyshev, Zh-K. Stokle, B. Muller, A. P. Solodkov, V. I. Shebeko, and A. F. Vanin, “Deposition of nitric oxide in the cardiovascular system,” Izv. Akad. Nauk. Ser. Biol., 29, No. 5, 585–596 (2002).
S. Yu. Mashina,V. V. Aleksandrov, A. V. Goryacheva, M. A. Vlasova, A. F. Vanin, I. Yu. Malyshev, and E. B. Manukhina, “Adaptation to hypoxia prevents impairments of cerebral circulation in neurodegenerative damage: the role of nitric oxide,” Byull. Eksperim. Biol. Med., 142, No. 8, 132–135 (2006).
F. Z. Meerson, Adaptation Medicine: Mechanisms and Protective Effects of Adaptation, Hypoxia Medical Ltd., Moscow (1993).
F. Z. Meerson, V. G. Pinelis, V. B. Kosheleva, L. Yu. Golubeva, T. V. Ryasina, N. Arsen’eva, A. L. Krushinskii, and T. P. Storozhevykh, “Adaptation to periodic hypoxia restricts subdural hemorrhage in epileptiform convulsions in rats,” Byull. Eksperim. Biol. Med., 116, 572–574 (1993).
M. G. Pshennikova, E. V. Popkova, D. A. Pokidyshev, I. P. Khomenko, O. M. Zelenina, S. V. Kruglov, E. B. Manukhina, M. V. Shimkovich, A. V. Goryaeva, and I. Yu. Malyshev, “Effects of adaptation to hypoxia on resistance to neurodegenerative brain damage in rats of different genetic strains,” Vestn. Ros. Akad. Med. Nauk., No. 2, 50–55 (2007).
M. G. Pshennikova, E. V. Popkova, I. P. Khomenko, E. B. Manukhina, A. V. Goryacheva, S. Yu. Mashina, D. A. Pokidyshev, and I. Yu. Malyshev, “Comparison of resistance to neurodegenerative brain damage in august rats and the Wistar population,” Byull. Eksperim. Biol. Med., 139, No. 5, 540–542 (2005).
J. I. Addae, F. F. Youssef, and T. W. Stone, “Neuroprotective role of learning in dementia: a biological explanation,” J. Alzheimer’s Dis., 5, 91–104 (2003).
R. L. Berghmans, “Anti-Alzheimer drugs: ethical aspects of research and practice,” Tijdschr. Gerontol. Geriatr., 31, 100–106 (2000).
M. Bernaudin, A. S. Nedelec, D. Divoux, E. T. MacKenzie, E. Petit, and P. Schumann-Bard, “Normobaric hypoxia induces tolerance to focal permanent cerebral ischemia in association with an increased expression of hypoxia-inducible factor-1 and its target genes, erythropoietin and VEGF, in the adult mouse brain,” J. Cereb. Blood Flow Metab., 22, 393–403 (2002).
D. Boyd-Kimball, R. Sultana, H. F. Poon, B. C. Lunn, F. Casamenti, G. Pepeu, J. B. Klein, and D. A. Butterfield, “Proteomic identification of proteins specifically oxidized by intracerebral injection of amyloid beta-peptide (1–42) into rat brain: implications for Alzheimer’s disease,” Neurosci., 132, 313–324 (2005).
H. Braak, U. Rüb, C. Schultz, and K. Del Tredici, “Vulnerability of cortical neurons to Alzheimer’s and Parkinson’s diseases,” J. Alzheimer’s Dis., 9, No. 3, Supplement, 35–44 (2006).
D. A. Butterfield, “Amyloid beta-peptide (1–42)-induced oxidative stress and neurotoxicity: implications for neurodegeneration in Alzheimer’s disease brain. A review,” Free Radic. Res., 36, 1307–1313 (2002).
V. Calabrese, E. Guagliano, M. Sapienza, C. Mancuso, D. A. Butterfield, and A. M. Stella, “Redox regulation of cellular stress response in neurodegenerative disorders,” Ital. J. Biochem., 55, 263–282 (2006).
V. Calabrese, T. E. Bates, and A. M. Stella, “NO synthase and NOdependent signal pathways in brain aging and neurodegenerative disorders: the role of oxidant/antioxidant balance,” Neurochem. Res., 25, 1315–1341 (2000).
C. A. Davies, D. M. Mann, P. Q. Sumpter, and P. O. Yates, “A quantitative morphometric analysis of the neuronal and synaptic content of the frontal and temporal cortex in patients with Alzheimer’s disease,” J. Neurol. Sci., 78, 151–164 (1987).
S. T. De Kosky and S. W. Scheff, “Synapse loss in frontal cortex biopsies in Alzheimer’s disease: correlation with cognitive severity,” Ann. Neurol., 27, 457–464 (1990).
H. Fai Poon, V. Calabrese, G. Scapagnini, and D. A. Butterfield, “Free radicals: Key to brain aging and heme oxygenase as a cellular response to oxidative stress,” J. Gerontol. Series A: Biol. Sci. Med. Sci., 59, M478–M493 (2004).
M. R. Farlow, M. L. Miller, and V. Pejovic, “Treatment options in Alzheimer’s disease: maximizing benefit, managing expectations,” Dement. Geriatr. Cogn. Disord., 25, 408–422, (2008).
R. A. Floyd, “Antioxidants, oxidative stress, and degenerative neurological disorders,” Proc. Soc. Exp. Biol. Med., 222, 236–245 (1999).
N. V. Gulyaeva, M. V. Stepanichev, M. V. Onufriev, I. V. Sergeev, O. S. Mitrokhina, Yu. V. Moiseeva, and E. N. Tkatchouk, “Interval hypoxia training prevents oxidative stress in striatum and locomotor disturbances in a rat model of parkinsonism,” in: Progress in Alzheimer’s and Parkinson’s Diseases, A. Fisher, I. Hahn, and M. Yoshida (eds.), Plenum Press (1998), pp. 717–723.
M. E. Jung, J. W. Simpkins, A. M. Wilson, H. F. Downey, and R. T. Mallet, “Intermittent hypoxia conditioning prevents behavioral deficit and brain oxidative stress in ethanol withdrawn rats,” J. Appl. Physiol. [Epub ahead of print, May 22, 2008].
U. Keil, A. Bonert, C. A. Marques, I. Scherping, J. Weyerman, J. B. Strosznajder, F. Müller-Spahn, C. Haass, C. Czech, L. Pradier, W. E. Müller, and A. Eckert, “Amyloid beta-induced changes in nitric oxide production and mitochondrial activity lead to apoptosis,” J. Biol. Chem., 279, 50310–50320 (2004).
D. S. Kim, J. Y. Kim, and Y. S. Han, “Alzheimer’s disease drug discovery from herbs: neuroprotectivity from beta-amyloid (1–42) insult,” J. Atern. Complement. Med., 13, 333–340 (2007).
A. M. Lin, C. F. Chen, and L. T. Ho, “Neuroprotective effect of intermittent hypoxia on iron-induced oxidative injury in rat brain,” Exp. Neurol., 176, 328–335 (2002).
N. Mahendra and S. Arkin, “Effects of four years of exercise, language, and social interventions on Alzheimer discourse,” J. Commun. Disord., 36, 395–422 (2003).
T. Malinski, “Nitric oxide and nitroxidative stress in Alzheimer’s disease,” J. Alzheimer’s Dis., 11, 207–218 (2007).
S. I. Marklund, N. G. Westman, E. Lundgren, and G. Roos, “Copperand zinc-containing superoxide dismutase, manganese-containing superoxide dismutase, catalase, and glutathione peroxidase in normal and neoplastic human cell lines and normal human tissues,” Cancer Res., 42, 1955–1961 (1982).
P. I. Moreira, M. S. Santos, C. R. Oliveira, J. C. Shenk, A. Nunomura, M. A. Smith, X. Zhu, and G. Perry, “Alzheimer disease and the role of free radicals in the pathogenesis of the disease,” CNS Neurol. Disord. Targets, 7, 3–10 (2008).
J. A. Neubauer, “Physiological and pathophysiological responses to intermittent hypoxia,” J. Appl. Hypoxia, 90, 1593–1599 (2001).
H. Ohkawa, N. Ohishi, and K. Yagi, “Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction,” Anal. Biochem., 95, 351–358 (1979).
G. M. Pasinetti, Z. Zhao, W. Qin, L. Ho, Y. Shrishailam, D. Macgrogan, W. Ressmann, N. Humala, X. Liu, C. Romero, B. Stetka, L. Chen, H. Ksiezak-Reding, and J. Wang, “Caloric intake and Alzheimer’s disease. Experimental approaches and therapeutic implications,” Interdiscip. Top. Gerontol., 35, 159–175 (2007).
K. N. Prasad, W. C. Cole, and K. C. Prasad, “Risk factors for Alzheimer’s disease: role of multiple antioxidants, non-steroidal anti-inflammatory and cholinergic agents alone or in combination in prevention and treatment,” J. Am. Coll. Nutr., 21, 506–522 (2002).
D. J. Selkoe, “Alzheimer’s disease is a synaptic failure,” Science, 298, 789–791 (2002).
G. L. Semenza, “O2-regulated gene expression: transcriptional control of cardiorespiratory physiology by HIF-1,” J. Appl. Physiol., 96, 1173–1177 (2004).
T. Soucek, R. Cumming, R. Dargusch, P. Maher, and D. Schubert, “The regulation of glucose metabolism by HIF-1 mediates a neuroprotective response to amyloid beta peptide,” Neuron, 39, 43–56 (2003).
P. J. Whitehouse, D. L. Price, A. W. Clark, J. T. Coyle, and M. R. De- Long, “Alzheimer disease: evidence for selective loss of cholinergic neurons in the nucleus basalis,” Ann. Neurol., 10, 122–126 (1981).
M. M. Zaleska, K. Nagy, and R. A. Floyd, “Iron-induced lipid peroxidation and inhibition of dopamine synthesis in striatum synaptosomes,” Neurochemistry, 14, 597–605 (1989).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Rossiiskii Fiziologicheskii Zhurnal imeni I. M. Sechenova, Vol. 95, No. 7, pp. 706–715, July, 2009
Rights and permissions
About this article
Cite this article
Manukhina, E.B., Goryacheva, A.V., Barskov, I.V. et al. Prevention of Neurodegenerative Damage to the Brain in Rats in Experimental Alzheimer’s Disease by Adaptation to Hypoxia. Neurosci Behav Physi 40, 737–743 (2010). https://doi.org/10.1007/s11055-010-9320-6
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-010-9320-6