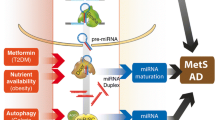
Abstract
Alzheimer’s disease (AD), a well known aging-induced neurodegenerative disease is related to amyloid proteinopathy. This proteinopathy occurs due to abnormalities in protein folding, structure and thereby its function in cells. The root cause of such kind of proteinopathy and its related neurodegeneration is a disorder in metabolism, rather metabolomics of the major as well as minor nutrients. Metabolomics is the most relevant “omics” platform that offers a great potential for the diagnosis and prognosis of neurodegenerative diseases as an individual’s metabolome. In recent years, the research on such kinds of neurodegenerative diseases, especially aging-related disorders is broadened its scope towards metabolic function. Different neurotransmitter metabolisms are also involved with AD and its associated neurodegeneration. The genetic and epigenetic backgrounds are also noteworthy. In this review, the physiological changes of AD in relation to its corresponding biochemical, genetic and epigenetic involvements including its (AD) therapeutic aspects are discussed.








Similar content being viewed by others
Data availablity
Data sharing is not applicable to this review article as no new data of our own were created.
References
Aisen P, Touchon J, Amariglio R, Andrieu S, Bateman R, Breitner J, Donohue M, Dunn B, Doody R, Fox N, Gauthier S, Grundman M, Hendrix S, Ho C, Isaac M, Raman R, Rosenberg P, Schindler R, Schneider L, Sperling R, Tariot P, Welsh-Bohmer K, Weiner M, Vellas B (2017) EU/US/CTAD task force: lessons learned from recent and current alzheimer’s prevention trials. J Prev Alzheimers Dis 4:116–124
Alkalay A, Rabinovici GD, Zimmerman G, Agarwal N, Kaufer D, Miller BL, Jagust WJ, Soreq H (2013) Plasma acetylcholinesterase activity correlates with intracerebral β-amyloid load. Curr Alzheimer Res 10:48–56
Alzheimer’s Association (2020) https://www.alz.org/alzheimers-dementia/research_progress/treatment-horizon. Accessed 20 Oct 2020
Alzheimer’s society (2020) https://www.alzheimers.org.uk/about-us/policy-and-influencing/what-we-think/stem-cell-research
Andersen OM, Reiche J, Schmidt V, Gotthardt M, Spoelgen R, Behlke J, von Arnim CAF, BreiderhoffT JP, Wu X, Bales KR, Cappai R, Masters CL, Gliemann J, Mufson EJ, Hyman BT, Paul SM, Nykjaer AA, Willnow TE (2005) Neuronal sorting protein-related receptor sorLA/LR11 regulates processing of the amyloid precursor protein. Proc Nat Acad Sci (USA) 102:13461–13466
Antúnez C, Boada M, González-Pérez A, Gayán J, Ramírez-Lorca R et al (2011) The membrane-spanning 4-domains, subfamily A (MS4A) gene cluster contains a common variant associated with Alzheimer's disease. Genome Med 3:33
Arendt T (2009) Synaptic degeneration in Alzheimer’s disease. Acta Neuropathol 118:167–179
Arnold SE, Arvanitakis Z, Macauley-Rambach SL, Koenig AM, Wang HY, Ahima RS, Craft S, Gandy S, Buettner C, Stoeckel LE, Holtzman DM, Nathan DM (2018) Brain insulin resistance in type 2 diabetes and Alzheimer disease: Concepts and conundrums. Nat Rev Neurol 14:168–181
Ballatore C, Brunden KR, Huryn DM, Trojanowski JQ, Lee VMY, Smith 3rd AB (2012) Microtubule stabilizing agents as potential treatment for Alzheimer’s disease and related neurodegenerative tauopathies. J Med Chem 55: 8979–8996
Banerjee S (2019) Aging-induced neurodegeneration in relation to brain regional Abeta deposition, locomotor and cognitive function: role of carnosine. J Neurochem 150:125
Banerjee S, Poddar MK (2015) Carnosine: effect on aging-induced increase in brain regional monoamine oxidase-A activity. Neurosci Res 92:62–70
Banerjee S, Poddar MK (2016a) Aging-induced changes in brain regional serotonin receptor binding: effect of carnosine. Neurosci 319:79–91
Banerjee S, Poddar MK (2016b) Can carnosine prevent the aging-induced changes of blood platelet and brain regional monoamine oxidase-A mRNA in relation to its activity? Intl J Pept Res Therap 22:471–480
Banerjee S, Poddar MK (2019) Carnosine in aging-induced neurodegeneration: a promising approach towards better tomorrow for geriatrics. Ann Pharmacol Pharm 4:1163
Banerjee S, Ghosh TK, Poddar MK (2015) Carnosine reverses the aging-induced down regulation of brain regional serotonergic system. Mech Ageing Dev 152:5–14
Banerjee S, Mukherjee B, Poddar MK (2019) Carnosine restores aging-induced elevation of corticosterone status and brain regional amyloid-beta in relation to down regulation of locomotor activity. J Syst Integr Neurosci 5:1–11
Barry LJ, Casimir AF (2000) A general hypothesis. In: Serotonin and behaviour. Neuropsychopharmacol. The fifth ed.
Bart MM, Luckenbach T, Bergner O, Ullrich O, Koch-Brandt C (2001) Multiple receptors mediate apoJ-dependent clearance of cellular debris into nonprofessional phagocytes. Experimental Cell Res 271:130–141
Bateman RJ, Xiong C, Benzinger TL, Fagan AM, Goate A et al (2012) Clinical and biomarker changes in dominantly inherited Alzheimer’s disease. N Engl J Med 367:795–804
Bedard K, Krause KH (2007) The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol Rev 87:245–313
Bell RD, Sagare AP, Friedman AE, Bedi GS, Holtzman DM, Deane R, Zlokovic BV (2007) Transport pathways for clearance of human Alzheimer's amyloid β-peptide and apolipoproteins E and J in the mouse central nervous system. J Cerebral Blood Flow Metabol 27:909–918
Berchtold NC, Coleman PD, Cribbs DH, Rogers J, Gillen DL, Cotman CW (2013) Synaptic genes are extensively downregulated across multiple brain regions in normal human aging and Alzheimer’s disease. Neurobiol Aging 34:1653–1661
Berendsen AAM, Kang JH, van de Rest O, EJM F, de Groot LCPGM, Grodstein F (2017) The dietary approaches to stop hypertension diet, cognitive function, and cognitive decline in American older women. J Am Med Directors Assoc 18:427–432
Berg AH, Scherer PE (2005) Adipose tissue, inflammation, and cardiovascular disease. Circulation Res 96:939–949
Bickel S, Lipp HP, Umbricht D (2008) Early auditory sensory processing deficits in mouse mutants with reduced NMDA receptor function. Neuropsychopharmacol 33:1680–1689
Bieschke J (2013) Natural compounds may open new routes to treatment of amyloid diseases. Neurotherapeutics 10:429–439
Blurton-Jones M, Kitazawa M, Martinez-Coria H, Castello NA, Müller F, Loring JF, Yamasaki TR, Poon WW, Green KN, LaFerla FM (2009) Neural stem cells improve cognition via BDNF in a transgenic model of Alzheimer disease. Proceedings of the National Academy of Sciences (USA) 106:13594–13599. https://doi.org/10.1073/pnas.0901402106
Bohnen NI, Kaufer DI, Hendrickson R, Ivanco LS, Lopresti B, Davis JG, Constantine G, Mathis CA, Moore RY, ST DK (2005) Cognitive correlates of alterations in acetylcholinesterase in Alzheimer’s disease. Neurosci Lett 380:127–132
Boldrini M, Fulmore CA, Tartt AN, Simeon LR, Pavlova I, Poposka V, Rosoklija GB, Stankov A, Arango V, Dwork AJ, Hen R, Mann JJ (2018) Human hippocampal neurogenesis persists throughout aging. Cell Stem Cell 22:589–599
Bossers K, Wirz KT, Meerhoff GF, Essing AH, Van Dongen JW, Houba P, Kruse CG, Verhaagen J, Swaab DF (2010) Concerted changes in transcripts in the prefrontal cortex precede neuropathology in Alzheimer’s disease. Brain 133:3699–3723
Bottiglieri T, Godfrey P, Flynn T, Carney MW, Toone BK, Reynolds EH (1990) Cerebrospinal fluid S-adenosylmethionine in depression and dementia: effects of treatment with parenteral and oral S-adenosylmethionine. J Neurol Neurosurg Psychiatry 53:1096–1098
Bourre JM, Pascal G, Durand G, Masson M, Dumont O, Piciotti M (1984) Alterations in the fatty acid composition of ratbrain cells (neurons, astrocytes, and oligodendrocytes) and of subcellular fractions (myelin and synaptosomes) induced by a diet devoid of n3 fatty acids. J Neurochem 43:342–348
Braak H, Braak E (1991) Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol 82:239–259
Braak H, Thal DR, Ghebremedhin E, Del Tredici K (2011) Stages of the pathologic process in Alzheimer disease: age categories from 1 to 100 years. J Neuropathol Exp Neurol 70:960–969
Braskie MN, Thompson PM (2013) Understanding cognitive deficits in Alzheimer’s disease based on neuroimaging findings. Trends Cogn Sci 17:510–516
Bubu OM, Brannick M, Mortimer J, Umasabor-Bubu O, Sebastião YV, Wen Y, Schwartz S, Borenstein AR, Wu Y, Morgan D, Anderson WM (2017) Sleep, cognitive impairment, and Alzheimer's disease: A systematic review and meta-analysis. Sleep 40:1–18
Butterfield DA, Reed T, Newman SF, Sultana R (2007) Roles of amyloid β-peptide-associated oxidative stress and brain protein modifications in the pathogenesis of Alzheimer’s disease and mild cognitive impairment. Free Radical Bio Med 43:658–677
Buttini M, Orth M, Bellosta S, Akeefe H, Pitas RE, Wyss-Coray T, Mucke L, Mahley RW (1999) Expression of human apolipoprotein E3 or E4 in the brains of Apoe−/− mice: isoform-specific effects on neurodegeneration. J Neurosci 19:4867–4880
Buzsáki G, Wang XJ (2012) Mechanisms of gamma oscillations. Ann. Rev. Neurosci 35:203–225
Cacabelos R (2014) Epigenomic networking in drug development: from pathogenic mechanisms to pharmacogenomics. Drug Dev Res 75:348–365
Cacabelos R, Torrellas C (2014) Epigenetic drug discovery for Alzheimer’s disease. Exp Opin Drug Discov 9:1059–1086
Cacabelos R, Cacabelos P, Torrellas C, Tellado I, Carril JC (2014) Pharmacogenomics of Alzheimer’s disease: novel therapeutic strategies for drug development. Methods Mol Biol 1175:323–556
Caccamo A, Maldonado MA, Bokov AF, Majumder S, Oddo S (2010) CBP gene transfer increases BDNF levels and ameliorates learning and memory deficits in a mouse model of Alzheimer’s disease. Proc Natl Acad Sci (USA) 107:22687–22692
Canter RG, Penney J, Tsai LH (2016) The road to restoring neural circuits for the treatment of Alzheimer’s disease. Nature 539(187):196
Cappai R (2014) Making sense of the amyloid precursor protein: its tail tells an interesting tale. J Neurochem 130:325–327
Carnevale D, Perrotta M, Lembo G, Trimarco B (2016) Pathophysiological links among hypertension and Alzheimer’s disease. High Blood Press Cardiovasc Prev 23:3–7
Chan CB, Ye K (2013) Serine-arginine protein kinases: new players in neurodegenerative diseases? Rev Neurosci 24:401–413
Chang TY, Chang CC, Ohgami N, Yamauchi Y (2006) Cholesterol sensing, trafficking, and esterification. Ann Rev Cell Dev Biol 22:129–157
Chapuis J, Hansmannel F, Gistelinck M, Mounier A, Van Cauwenberghe C et al (2013) Increased expression of BIN1 mediates Alzheimer genetic risk by modulating tau pathology. Mol Psychiatry 18:1225–1234
Ciarlo E, Massone S, Penna I, Nizzari M, Gigoni A, Dieci G, Russo C, Florio T, Cancedda R, Pagan A (2013) An intronic ncRNA-dependent regulation of SORL1 expression affecting Abeta formation is upregulated in post-mortem Alzheimer’s disease brain samples. Dis Model Mech 6:424–433
Citron M, Westaway D, Xia W, Carlson G, Diehl T et al (1997) Mutant Presenilins of Alzheimer's Disease Increase Production of 42-residue Amyloid Beta-Protein in Both transfected cells and transgenic mice. Nat Med 3:67–72
Coppede F, Tannorella P, Pezzini I, Migheli F, Ricci G, Lenco EC, Piaceri I, Polini A, Nacmias B, Monzani F, Sorbi S, Siciliano G, Migliore L (2012) Folate, homocysteine, vitamin B12, and polymorphisms of genes participating in one-carbon metabolism in late-onset Alzheimer’s disease patients and healthy controls. Antioxid Redox Signal 17:195–204
Corder EH, Saunders AM, Strittmatter WJ, Schmechel DE, Gaskell PC, Small GW, Roses AD, Haines JL, Pericak-Vance MA (1993) Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’sdisease in late onset families. Science 261:921–923
Corder EH, Saunders AM, Risch NJ, Strittmatter WJ, Schmechel DE et al (1994) Protective effect of apolipoprotein E type 2 allele for late onset Alzheimer disease. Nat Genetics 7:180–184
Counts SE, Alldred MJ, Che S, Ginsberg SD, Mufson EJ (2014) Synaptic gene dysregulation within hippocampal CA1 pyramidal neurons in mild cognitive impairment. Neuropharmacol 79:172–179
Craft S, Asthana S, Newcomer JW, Wilkinson CW, Matos IT, Baker LD, Cherrier M, Lofgreen C, Latendresse S, Petrova A, Plymate S, Raskind M, Grimwood K, Veith RC (1999) Enhancement of Memory in Alzheimer Disease With Insulin and Somatostatin, but Not Glucose. Arch Gen Psychiatry 56:1135–1140
Cremonini AL, Caffa I, Cea M, Nencioni A, Odetti P, Monacelli F (2019) Nutrients in the prevention of alzheimer’s disease oxidative medicine and cellular longevity 2019: 9874159
Cuenco TK, Lunetta KL, Baldwin CT, McKee AC, Guo J, Cupples LA, Green RC, St. George-Hyslop PH, Chui H, de Carli C, Farrer LA (2008) Association of distinct variants in SORL1 with cerebrovascular and neurodegenerative changes related to Alzheimer disease. Arch Neurol 65:1640–1648
Davignon J, Gregg RE, Sing CF (1988) Apolipoprotein E polymorphism and atherosclerosis. Arteriosclerosis 8:1–21
de Cavanagh EMV, Inserra F, Ferder LN (2011) Angiotensin II blockade: a strategy to slow ageing by protecting mitochondria? Cardiovasc Res 89:31–40
De Strooper B, Saftig P, Craessaerts K, Vanderstichele H, Guhde G, Annaert W, Figura KV, Leuven FV (1998) Deficiency of presenilin-1 inhibits the normal cleavage of amyloid precursor protein. Nature 391:387–390
Desikan RS, Schork AJ, Wang Y, Thompson WK, Dehghan A et al. 2015. Polygenic overlap between C-reactive protein, plasma lipids and Alzheimer's disease. Circulation 131:22061–22069
Devanand DP, Michaels-Marston KS, Liu X, Pelton GH, Padilla M, Marder K, Bell K, Stern Y, Mayeux R (2000) Olfactory deficits in patients with mild cognitive impairment predict Alzheimer's disease at followup. Am J Psychiatry 157:1399–1405
Devi L, Prabhu BM, Galati DF, Avadhani NG (2006) Anandatheerthavarada HK.Accumulation of amyloid precursor protein in the mitochondrial import channels of human Alzheimer’s disease brain is associated with mitochondrial dysfunction. J Neurosci 26:9057–9068
Di Francesco A, Arosio B, Falconi A, Micioni Di Bonaventura MV, Karimi M, Mari D, Casati M, Maccarrone M, D'Addario C (2015) Global changes in DNA methylation in Alzheimer’s disease peripheral blood mononuclear cells. Brain Behav Immun 45:139–144
Dickerson BC, Bakkour A, Salat DH, Feczko E, Pacheco J, Greve DN, Grodstein F, Wright CI, Blacker D, Rosas HD, Sperling RA, Atri A, Growdon JH, Hyman BT, Morris JC, Fischl B, Buckner RL (2009) The cortical signature of Alzheimer’s disease: regionally specific cortical thinning relates to symptom severity in very mild to mild AD dementia and is detectable in asymptomatic amyloidpositive. Cereb Cortex 19:497–510
Dietschy JM, Turley SD (2001) Cholesterol metabolism in the brain. Curr Opi Lipidol:12, 105–112
Ding F, Yao J, Rettberg JR, Chen S, Brinton RD (2013) Early decline in glucose transport and metabolism precedes shift to ketogenic system in female aging and Alzheimer’s mouse brain: Implication for bioenergetic intervention. PLoS One 8:e79977
Do J, Kim JI, Bakes J, Lee K, Kaang BK (2012) Functional roles of neurotransmitters and neuromodulators in the dorsal striatum. Learn Mem 20:21–28
Dong Y, Brewer GJ (2019) Global metabolic shifts in age and Alzheimer’s disease mouse brain pivot at NAD+/NADH redox sites. J Alz Dis 71:119–140
Dong H, Yuedea CM, Yooa HS, Martina MV, Deal C, Mace AG, Csernansky JG (2008) Corticosterone and related receptor expression are associated with increased β-amyloid plaques in isolated Tg2576 mice. Neurosci 155: 154–163
Doody RS, Thomas RG, Farlow M, Iwatsubo T, Vellas B, Joffe S, Kieburtz K, Raman R, Sun X, Aisen PS, Siemers E, Liu-Seifert H, Mohs R (2014) Phase 3 trials of solanezumab for mild-to-moderate Alzheimer’s disease. N Engl J Med 370:311–321
Dragicevic N, Smith A, Lin X, Yuan F, Copes N, Delic V, Tan J, Cao C, Shytle RD, Bradshaw PC (2011) Green tea epigallocatechin-3-gallate (EGCG) and other flavonoids reduce Alzheimer’s amyloid-induced mitochondrial dysfunction. J Alz Dis 26:507–521
Dregan A, Gulliford MC (2013) Leisure-time physical activity over the life course and cognitive functioning in late mid-adult years: a cohort-based investigation. Psychol Med 43:2447–2458
Durga J, van Boxtel MP, Schouten EG, Kok FJ, Jolles J, Katan MB, Verhoef P (2007) Effect of 3-year folic acid supplementation on cognitive function in older adults in the FACIT trial: a randomised, double blind, controlled trial. Lancet 369:208–216
Erum JV, Dam DV, PPD D (2019) Alzheimer’s disease: Neurotransmitters of the sleep-wake cycle. Neurosci Biobehav Rev 105:72–80
Esler WP, Wolfe MS (2001) A portrait of Alzheimer secretases–new features and familiar faces. Science 293:1449–1454
Fan X, Wheatley EG, Villeda SA (2017) Mechanisms of hippocampal aging and the potential for rejuvenation. Ann Rev Neurosci 40:251–272
Fein JA, Sokolow S, Miller CA, Vinters HV, Yang F, Cole GM, Gylys KH (2008) Co-localization of amyloid beta and tau pathology in Alzheimer’s disease synaptosomes. Am J Pathol 172:1683–1692
Ferreira-Vieira TH, Guimaraes IM, Silva FR, Ribeiro FM (2016) Alzheimer's disease: targeting the cholinergic system. Curr Neuropharmacol 14:101–115
Foley P (2010) Lipids in Alzheimer’s disease: A century-old story. Biochim Biophys Acta 1801:750–753
Frölich L, Blum-Degen D, Bernstein HG, Engelsberger S, Humrich J, Laufer S, Muschner D, Thalheimer A, Türk A, Hoyer S, Zöchling R, Boissl KW, Jellinger K, Riederer P (1998) Brain insulin and insulin receptors in Aging and sporadic Alzhimer’s disease. J Neural Transm 105:423–438
Fuso A, Seminara L, Cavallaro RA, D’Anselmi F, Scarpa S (2005) S-adenosylmethionine/ homocysteine cycle alterations modify DNA methylation status with consequent dysregulation of PS1 and BACE and betaamyloid production. Mol Cell Neurosci 28:195–204
Fuso A, Nicolia V, Cavallaro RA, Ricceri L, D’Anselmi F, Coluccia P, Calamandrei G, Scarpa S (2008) B-vitamin deprivation induces hyperhomocysteinemia and brain S-adenosylhomocysteine, depletes brain S-adenosylmethionine, and enhances PS1 and BACE expression and amyloid-beta deposition in mice. Mol Cell Neurosci 37:731–746
Fuso A, Cavallaro RA, Nicolia V, Scarpa S (2012) SEN1 promoter demethylation in hyperhomocysteinemic TgCRND8 mice is the culprit, not the consequence. Curr Alz Res 9:527–535
Gal-Ben-Ari S, Rosenblum K (2011) Molecular mechanisms underlying memory consolidation of taste information in the cortex. Front Behav Neurosci 5:87
Gandy JC, Melendez-Ferro M, Bijur GN, Van Leuven F, Roche JK, Lechat B, Devijver H, Demedts D, Perez-Costas E, Roberts RC (2013) Glycogen synthase kinase-3β (GSK3β) expression in a mouse model of Alzheimer’s disease: a light and electron microscopy study. Synapse 67:313–327
Gao C, Hölscher C, Liu Y, Li L (2011) GSK3: a key target for the development of novel treatments for type 2 diabetes mellitus and Alzheimer disease. Rev Neurosci 23:1–11
Garcia-Alloza M, Gil-Bea FJ, Diez-Ariza M, CPLH C, Francis PT, Lasheras B, Ramirez MJ (2005) Cholinergic-serotonergic imbalance contributes to cognitive and behavioral symptoms in Alzheimer’s disease. Neurophysiologica 43:442–449
Ghosh D, KR LV, Barnett AJ, Brewer GJ (2012) A reversible early oxidized redox state that precedes macromolecular ROS damage in aging nontransgenic and 3xTg-AD mouse neurons. J Neurosci 32:5821–5832
Giaccone G, Tagliavini F, Linoli G, Bouras C, Frigerio L, Frangione B, Bugiani O (1989) Down patients; extracellular preamyloid deposits precede neuritic degeneration and senile plaques. Neurosci Lett 97:232–238
Giau VV, Bagyinszky E, Yang Y et al (2019) Genetic analyses of early-onset Alzheimer’s disease using next generation sequencing. Sci Rep 9:8368
Gibson GE, Karuppagounder SS, Shi Q (2008) Oxidant-induced changes in mitochondria and calcium dynamics in the pathophysiology of Alzheimer’s disease. Ann N Y Acad Sci 1147:221–232
Ginsberg SD, Mufson EJ, Counts SE, Wuu J, Alldred MJ, Nixon RA, Che S (2010) Regional selectivity of rab5 and rab7 protein upregulation in mild cognitive impairment and Alzheimer’s disease. J Alz Dis 22:631–639
Goate A, Chartier-Harlin MC, Mullan M, Brown J, Crawford F, Fidani L, Giuffra L, Haynes A, Irving N, James L (1991) Segregation of a missense mutation in the amyloid precursor protein gene with familial Alzheimer’s disease. Nature 349:704–706
Goldgaber D, Lerman MI, OW MB, Saffiotti U, Gajdusek DC (1987) Characterization and chromosomal localization of a cDNA encoding brain amyloid of Alzheimer’s disease. Science 235:877–880
Gotz J, Ittner LM, Kins S (2006) Do axonal defects in tau and amyloid precursor protein transgenic animals model axonopathy in Alzheimer’s disease? J Neurochem 98:993–1006
Grant B (1999) Dietary links to Alzheimer's disease: 1999 Update. J Alz Dis 1:197–201
Grant C (2008) Metabolic reconfiguration is a regulated response to oxidative stress. J Biol 7:1
Granzotto A, Zatta P (2011) Resveratrol acts not through anti-aggregative pathways but mainly via its scavenging properties against Abeta and Abeta-metal complexes toxicity. PLoS One 6:e21565
Grossberg GT, Manes F, Allegri RF, Gutiérrez-Robledo LM, Gloger S, Xie L, Jia XD, Pejović V, Miller ML, Perhach JL, Graham SM (2013) The safety, tolerability, and efficacy of once-daily memantine (28 mg): a multinational, randomized, double-blind, placebo-controlled trial in patients with moderate-to-severe Alzheimer’s disease taking cholinesterase inhibitors. CNS Drugs 27:469–478
Guerreiro RJ, Lohmann E, Kinsella E, Bras JM, Luu N, Gurunlian N, Dursun B, Bilgic B, Santana I, Hanagasi H, Gurvit H, Gibbs JR, Oliveira C et al (2012) Exome sequencing reveals an unexpected genetic cause of disease: NOTCH3 mutation in a Turkish family with Alzheimer’s disease. Neurobiol Aging 33:1008 e17–1008 e23
Guerreiro R, Wojtas A, Bras J, Carrasquillo M, Rogaeva E, Majounie E, Cruchaga C, Sassi C, Kauwe JS, Younkin S, Hazrati L, Collinge J, Pocock J, Lashley T, Williams J, Lambert JC, Amouyel P, Goate A, Rademakers R, Morgan K, Powell J, St George-Hyslop P, Singleton A, Hardy J, Alzheimer Genetic Analysis Group (2013) TREM2 variants in Alzheimer’s disease. N Engl J Med 368:117–127
Haan MN, Miller JW, Aiello AE, Whitmer RA, Jagust WJ, Mungas DM, Allen LH, Green R (2007) Homocysteine, B vitamins, and the incidence of dementia and cognitive impairment: results from the Sacramento Area Latino Study on Aging. Am J Clin Nutr 85:511–517
Hannun YA, Obeid LM (2008) Principles of bioactive lipid signalling: Lessons from sphingolipids. Nat Rev Mol Cell Biol 9:139–150
Harel A, Wu F, Mattson MP, Morris CM, Yao PJ (2008) Evidence for CALM in directing VAMP2 trafficking. Traffic 9:417–429
Harold D, Abraham R, Hollingworth P, Sims R, Gerrish A, Hamshere ML, Pahwa JS, Moskvina V, Dowzell K, Williams A, Jones N, Thomas C, Stretton A, Morgan AR, Lovestone S, Powell J, Proitsi P, Lupton MK, Brayne C, Rubinsztein DC, Gill M, Lawlor B, Lynch A, Morgan K, Brown KS, Passmore PA, Craig D, McGuinness B, Todd S, Holmes C, Mann D, Smith AD, Love S, Kehoe PG, Hardy J, Mead S, Fox N, Rossor M, Collinge J, Maier W, Jessen F, Schürmann B, Heun R, van den Bussche H, Heuser I, Kornhuber J, Wiltfang J, Dichgans M, Frölich L, Hampel H, Hüll M, Rujescu D, Goate AM, Kauwe JS, Cruchaga C, Nowotny P, Morris JC, Mayo K, Sleegers K, Bettens K, Engelborghs S, De Deyn PP, Van Broeckhoven C, Livingston G, Bass NJ, Gurling H, McQuillin A, Gwilliam R, Deloukas P, Al-Chalabi A, Shaw CE, Tsolaki M, Singleton AB, Guerreiro R, Mühleisen TW, Nöthen MM, Moebus S, Jöckel KH, Klopp N, Wichmann HE, Carrasquillo MM, Pankratz VS, Younkin SG, Holmans PA, O'Donovan M, Owen MJ, Williams J (2009) Genome-wide association study identifies variants at CLU and PICALM associated with Alzheimer's disease. Nat Genetics 41:1088–1093
Harris JA, Devidze N, Verret L, Ho K, Halabisky B, ThwinMT KD, Hamto P, Lo I, Yu G-Q, Palop JJ, Masliah E, Mucke L (2010) Transsynaptic progression of amyloid-β-induced neuronal dysfunction within the entorhinal-hippocampal network. Neuron 68:428–441
Hartmann T, Kuchenbecker J, Grimm MO (2007) Alzheimer's disease: The lipid connection. J Neurochem 103:159–170
Hattori C, Asai M, Onishi H, Sasagawa N, Hashimoto Y, Saido TC, Maruyama K, Mizutani S, Ishiura S (2006) BACE1 interacts with lipid raft proteins. J Neurosci Res 84:912–917
Heckmann JM, Low WC, de Villiers C, Rutherfoord S, Vorster ARao H, Morris CM, Ramesar RS, Kalaria RN (2004) Novel presenilin 1 mutation with profound neurofibrillary pathology in an indigenous Southern African family with early-onset Alzheimer’s disease. Brain 127:133–142
Hellerstein MK (2002) Carbohydrate-induced hypertriglyceride-mia: modifying factors and implications for cardiovascularrisk. Curr Opin Lipidol 13:33–40
Henderson ST (2004) High carbohydrate diets and Alzheimer's disease. Med Hypotheses 62:689–700
Henneman WJ, Sluimer JD, Barnes J, van der Flier WM, Sluimer IC, Fox NC, Scheltens P, Vrenken H, Barkhof F (2009) Hippocampal atrophy rates in Alzheimer disease: added value over whole brain volume measures. Neurol 72:999–1007
Hermona S, Seidman S (2001) Acetylcholinesterase- new roles for an old actor. Nat Rev Neurosci 2:294–302
Herreman A, Hartmann D, Annaert W, Saftig P, Craessaerts K, Serneels L, Umans L, Schrijvers V, Checler F, Vanderstichele H, Baekelandt V, Dressel R, Cupers P, Huylebroeck D, Zwijsen A, Van Leuven F, De Strooper B (1999) Presenilin 2 deficiency causes a mild pulmonary phenotype and no changes in amyloid precursor protein processing but enhances the embryonic lethal phenotype of presenilin 1 deficiency. Proc Natl Acad Sci (USA) 96:11872–11877
Hilpert H, Guba W, Woltering TJ, Wostl W, Pinard E, Mauser H, Mayweg AV, Rogers-Evans M, Humm R, Krummenacher D, Muser T, Schnider C, Jacobsen H, Ozmen L, Bergadano A, Banner DW, Hochstrasser R, Kuglstatter A, David-Pierson P, Fischer H, Polara A, Narquizian R (2013) β-Secretase (BACE1) inhibitors with high in vivo efficacy suitable for clinical evaluation in Alzheimer’s disease. J Med Chem 56:3980–3995
Hollingworth P, Harold D, Sims R, Gerrish A, Lambert JC, Carrasquillo MM, Abraham R, Hamshere ML, Pahwa JS, Moskvina V, Dowzell K, Jones N, Stretton A, Thomas C, Richards A, Ivanov D, Widdowson C, Chapman J, Lovestone S, Powell J, Proitsi P, Lupton MK, Brayne C, Rubinsztein DC, Gill M, Lawlor B, Lynch A, Brown KS, Passmore PA, Craig D, McGuinness B, Todd S, Holmes C, Mann D, Smith AD, Beaumont H, Warden D, Wilcock G, Love S, Kehoe PG, Hooper NM, Vardy ER, Hardy J, Mead S, Fox NC, Rossor M, Collinge J, Maier W, Jessen F, Rüther E, Schürmann B, Heun R, Kölsch H, van den Bussche H, Heuser I, Kornhuber J, Wiltfang J, Dichgans M, Frölich L, Hampel H, Gallacher J, Hüll M, Rujescu D, Giegling I, Goate AM, Kauwe JS, Cruchaga C, Nowotny P, Morris JC, Mayo K, Sleegers K, Bettens K, Engelborghs S, De Deyn PP, Van Broeckhoven C, Livingston G, Bass NJ, Gurling H, McQuillin A, Gwilliam R, Deloukas P, Al-Chalabi A, Shaw CE, Tsolaki M, Singleton AB, Guerreiro R, Mühleisen TW, Nöthen MM, Moebus S, Jöckel KH, Klopp N, Wichmann HE, Pankratz VS, Sando SB, Aasly JO, Barcikowska M, Wszolek ZK, Dickson DW, Graff-Radford NR, Petersen RC, Alzheimer’s Disease Neuroimaging Initiative, van Duijn CM, Breteler MM, Ikram MA, AL DS, Fitzpatrick AL, Lopez O, Launer LJ, Seshadri S, CHARGE consortium, Berr C, Campion D, Epelbaum J, Dartigues JF, Tzourio C, Alpérovitch A, Lathrop M, EADI consortium, Feulner TM, Friedrich P, Riehle C, Krawczak M, Schreiber S, Mayhaus M, Nicolhaus S, Wagenpfeil S, Steinberg S, Stefansson H, Stefansson K, Snaedal J, Björnsson S, Jonsson PV, Chouraki V, Genier-Boley B, Hiltunen M, Soininen H, Combarros O, Zelenika D, Delepine M, Bullido MJ, Pasquier F, Mateo I, Frank-Garcia A, Porcellini E, Hanon O, Coto E, Alvarez V, Bosco P, Siciliano G, Mancuso M, Panza F, Solfrizzi V, Nacmias B, Sorbi S, Bossú P, Piccardi P, Arosio B, Annoni G, Seripa D, Pilotto A, Scarpini E, Galimberti D, Brice A, Hannequin D, Licastro F, Jones L, Holmans PA, Jonsson T, Riemenschneider M, Morgan K, Younkin SG, Owen MJ, O’Donovan M, Amouyel P, Williams J (2011) Common variants at ABCA7, MS4A6A/MS4A4E, EPHA1, CD33, and CD2AP are associated with Alzheimer’s disease. Nat Genet 43:429–435
Hooff GP, Wood WG, Muller WE, Eckert GP (2010) Isoprenoids, small GTPases and Alzheimer's disease. Biochimica Et Biophysica Acta 1801:896–905
Hoppe JB, Coradini K, Frozza RL, Oliveira CM, Meneghetti AB, Bernardi A, Pires ES, RCR B, Salbego CG (2013) Free and nanoencapsulated Curcumin suppress beta-amyloid–induced cognitive impairments in rats: involvement of BDNF and Akt/GSK-3beta signaling pathway. Neurobiol Learn Mem 106:134–144
Howard R, McShane R, Lindesay J, Ritchie C, Baldwin A, Barber R, Burns A, Dening T, Findlay D, Holmes C, Hughes A, Jacoby R, Jones R, Jones R, McKeith I, Macharouthu A, O'Brien J, Passmore P, Sheehan B, Juszczak E, Katona C, Hills R, Knapp M, Ballard C, Brown R, Banerjee S, Onions C, Griffin M, Adams J, Gray R, Johnson T, Bentham P, Phillips P (2012) Donepezil and memantine for moderate-to-severe Alzheimer’s disease. N Engl J Med 366:893–903
Huang Y (2006) Molecular and cellular mechanisms of apolipoprotein E4 neurotoxicity and potential therapeutic strategies. Curr Opin Drug Discov Devel 9:627–641
Iaccarino HF, Singer AC, Martorell AJ, Rudenko A, Gao F, Gillingham TZ, Mathys H, Seo J, Kritskiy O, Abdurrob F, Adaikkan C, Canter RG, Rueda R, Brown EN, Boyden ES, Tsai LH (2016) Gamma frequency entrainment attenuates amyloid load and modifies microglia. Nature 540:230–235
Ittner LM, Gotz J (2011) Amyloid-beta and tau--a toxic pas de deux in Alzheimer’s disease. Nat Rev Neurosci 12:65–72
Iwata N, Tsubuki S, Takaki Y, Watanabe K, Sekiguchi M, Hosoki E, Kawashima-Morishima M, Lee HJ, Hama E, Sekine-Aizawa Y, Saido TC (2000) Identification of the major Abeta1–42-degrading catabolic pathway in brain parenchyma: suppression leads to biochemical and pathological deposition. Nat Med 6:143–150
Iwatsubo T, Odaka A, Suzuki N, Mizusawa H, Nukina N, Ihara Y (1994) Visualization of A beta 42(43) and A beta 40 in senile plaques with end-specific A beta monoclonals: evidence that an initially deposited species is A beta 42(43). Neuron 13:45–53
Jack CR Jr, Shiung MM, Weigand SD, O'Brien PC, Gunter JL, Boeve BF, Knopman DS, Smith GE, Ivnik RJ, Tangalos EG, Petersen RC (2005) Brain atrophy rates predict subsequent clinical conversion in normal elderly and amnestic MCI. Neurol 65:1227–1231
Jack CR Jr, Knopman DS, Jagust WJ, Shaw LM, Aisen PS, Weiner MW, Petersen RC, Trojanowski JQ (2010) Hypothetical model of dynamic biomarkers of the Alzheimer’s pathological cascade. Lancet Neurol 9:119–128
Jacobsen H, Ozmen L, Caruso A, Narquizian R, Hilpert H, Jacobsen B, Terwel D, Tanghe A, Bohrmann B (2014) Combined treatment with a BACE inhibitor and anti-Aβ antibody gantenerumab enhances amyloid reduction in APP London mice. J Neurosci 34:11621–11630
Janson J, Laedtke T, Parisi JE, O’Brien P, Petersen RC, Butler PC (2004) Increased Risk of Type 2 Diabetes in Alzheimer Disease. Diabetes 53:474–481
Johnson AA, Sarthi J, Pirooznia SK, Reube W, Elefant F (2013) Increasing Tip60 HAT levels rescues axonal transport defects and associated behavioral phenotypes in a Drosophila Alzheimer’s disease model. J Neurosci 33:7535–7547
Jonsson T, Atwal JK, Steinberg S, Snaedal J, Jonsson PV, Bjornsson S, Stefansson H, Sulem P, Gudbjartsson D, Maloney J, Hoyte K, Gustafson A, Liu Y, Lu Y, Bhangale T, Graham RR, Huttenlocher J, Bjornsdottir G, Andreassen OA, Jönsson EG, Palotie A, Behrens TW, Magnusson OT, Kong A, Thorsteinsdottir U, Watts RJ, Stefansson K (2012) A mutation in APP protects against Alzheimer's disease and age-related cognitive decline. Nature 488:96–99
Jonsson T, Stefansson H, Steinberg S, Jonsdottir I, Jonsson PV, Snaedal J, Bjornsson S, Huttenlocher J, Levey AI, Lah JJ, Rujescu D, Hampel H, Giegling I, Andreassen OA, Engedal K, Ulstein I, Djurovic S, Ibrahim-Verbaas C, Hofman A, Ikram MA, van Duijn CM, Thorsteinsdottir U, Kong A, Stefansson K (2013) Variant ofassociated with the risk of Alzheimer’s disease. N Engl J Med 368:107–116
Kaddurah-Daouk R, Zhu H, Sharma S, Bogdanov M, Rozen SG, Matson W, Oki NO, Motsinger-Reif AA, Churchill E, Lei Z, Appleby D, Kling MA, Trojanowski JQ, Doraiswamy PM, Arnold SE (2013) Alterations in metabolic pathways and networks in Alzheimer’s disease. Transl Psychiatry 3:e244
Kang J, Lemaire HG, Unterbeck A, Salbaum JM, Masters CL, Grzeschik KH, Multhaup G, Beyreuther K, Müller-Hill B (1987) The precursor of Alzheimer’s disease amyloid A4 protein resembles cell-surface receptor. Nature 325:733–736
Kantrowitz JT, Epstein ML, Beggel O, Rohrig S, Lehrfeld JM, Revheim N, Lehrfeld NP, Reep J, Parker E, Silipo G, Ahissar M, Javitt DC (2016) Neurophysiological mechanisms of cortical plasticity impairments in schizophrenia and modulation by the NMDA receptor agonist D-serine. Brain 139:3281–3295
Keller JN, Lauderback CM, Butterfield DA, Kindy MS, Yu J, Markesbery WR (2000) Amyloid beta-peptide effects on synaptosomes from apolipoprotein E-deficient mice. J Neurochem 74:1579–1586
Kennedy ME, Stamford AW, Chen X, Cox K, Cumming JN, Dockendorf JM, Egan M, Ereshefsky L, Hodgson RA, Hyde LA, Jhee S, Kleijn HJ, Kuvelkar R, Li W, Mattson BA, Mei H, Palcza J, Scott JD, Tanen M, Troyer MD, Tseng JL, Stone JA, Parker EM, Forman MS (2016) The BACE1 inhibitor verubecestat (MK-8931) reduces CNS β-amyloid in animal models and in Alzheimer’s disease patients. Sci Transl Med 8:363ra150
Kim J, Wei Y, Sowers JR (2008) Role of mitochondrial dysfunction in insulin resistance. Circ Res 102:401–414
Kim B, Backus C, Oh S, Hayes JM, Feldman EL (2009) Increased Tau Phosphorylation and Cleavage in Mouse Models of Type 1 and Type 2 Diabetes. Endocrinology 150:5294–5301
Klaips CL, Jayaraj GG, Hartl FU (2018) Pathways of cellular proteostasis in aging and disease. J Cell Biol 217:51–63. https://doi.org/10.1083/jcb.201709072
Köles L, Kató E, Hanuska A, Zádori ZS, Al-Khrasani M, Zelles T, Rubini P, Illes P (2016) Modulation of excitatory neurotransmission by neuronal/glial signalling molecules: interplay between purinergic and glutamatergic systems. Purinergic Signal 12:1–24
Kosik KS (2013) Diseases: Study neuron networks to tackle Alzheimer’s. Nature 503:31–32
Kroner Z (2009) The relationship between Alzheimer’s disease and diabetes: Type 3 diabetes? Altern Med Rev 14:373–379
Kubota T, Takae H, Miyake K (2012) Epigenetic mechanisms and therapeutic perspectives for neurodevelopmental disorders. Pharm (Basel) 5:369–383
Kumar K, Kumar A, Keegan RM, Deshmukh R (2018) Recent advances in the neurobiology and neuropharmacology of Alzheimer’s disease. Biomed Pharmacother 98:297–307
Lahiri DK, Rogers JT, Greig NH, Smbamurti K (2004) Rationale for the development of cholinesterase inhibitors as anti-Alzheimer agents. Curr Pharm Des 10:3111–3119
Lahiri DK, Maloney B, Basha MR, Ge YW, Zawia NH (2007) How and when environmental agents and dietary factors affect the course of Alzheimer’s disease: the “LEARn” model (latent early-life associated regulation) may explain the triggering of AD. Curr Alzheimer Res 4:219e228
Lahmy V, Meunier J, Malmström S, Naert G, Givalois L, Kim SH, Villard V, Vamvakides A, Maurice T (2013) Blockade of Tau hyperphosphorylation and Aβ1–42 generation by the aminotetrahydrofuran derivative ANAVEX2–73, a mixed muscarinic and σ1 receptor agonist, in a nontransgenic mouse model of Alzheimer’s disease. Neuropsychopharmacology 38:1706–1723
Lambert JC, Heath S, Even G, Campion D, Sleegers K, Hiltunen M, Combarros O, Zelenika D, Bullido MJ, Tavernier B, Letenneur L, Bettens K, Berr C, Pasquier F, Fiévet N, Barberger-Gateau P, Engelborghs S, Deyn PD, Mateo I, Franck A, Helisalmi S, Porcellini E, Hanon O, European Alzheimer's Disease Initiative Investigators, MMD P, Lendon C, Dufouil C, Jaillard C, Leveillard T, Alvarez V, Bosco P, MancusoM PF, Nacmias B, Bossù P, Piccardi P, Annoni G, Seripa D, Galimberti D, Hannequin D, Licastro F, Soininen H, Ritchie K, Blanché H, Dartigues JF, Tzourio C, Gut I, Broeckhoven CV, Alpérovitch A, Lathrop M, Amouyel P (2009) Genome-wide association study identifies variants at CLU and CR1 associated with Alzheimer's disease. Nat Genet 41:1094–1099
Lanctot KL, Amatniek J, Ancoli-Israel S, Arnold SE, Ballard C, Cohen-Mansfield J, Ismail Z, Lyketsos C, Miller DS, Musiek E, Osorio RS, Rosenberg PB, Satlin A, Steffens D, TariotP BLJ, Carrillo MC, Hendrix JA, Jurgens H, Boot B (2017) Neuropsychiatric signs and symptoms of Alzheimer’s disease: New treatment paradigms. Alzheimers Dement (N Y) 3:440–449
Landreth G, Jiang Q, Mandrekar S, Heneka M (2008) PPARgamma agonists as therapeutics for the treatment of Alzheimer’s disease. Neurotherapeutics 5:481–489
Lee SF, Shah S, Li H, Yu C, Wilson H, Yu G (2002) Mammalian APH-1 interacts with presenilin and nicastrin and is required for intramembrane proteolysis of amyloid-β precursor protein and Notch. J Biol 277:45013–45019
Lee S, Lemere CA, Frost JL, Shea TB (2012) Dietary supplementation with S-adenosyl methionine delayed amyloid-ß and tau pathology in 3xTg-AD mice. J Alzheimers Dis 28:423–431
Lee JH, Oh IH, Lim HK (2016) Stem cell therapy: a prospective treatment for Alzheimer’s disease. Psychiatry Investig 13:583–589
Li XL, Aou S, Oomura Y, Hori N, Fukunaga K, Hori T (2002) Impairment of long-term potentiation and spatial memory in leptin receptor-deficient rodents. Neuroscience 113:607–615. https://doi.org/10.1016/S0306-4522(02)00162-8
Li H, Wolfe SM, Selkoe JD (2009) Toward structural elucidation of the γ-secretase complex. Structure 17:326–334
Li R, Cui J, Shen Y (2014) Brain sex matters: estrogen in cognition and Alzheimer's disease. Mol CellEndocrinol 389:13–21
Liang WS, Dunckley T, Beach TG, Grover A, Mastroeni D, Ramsey K, Caselli RJ, Kukull WA, McKeel D, Morris JC, Hulette CM, Schmechel D, Reiman EM, Rogers J, Stephan DA (2010) Neuronal gene expression in non-demented individuals with intermediate Alzheimer’s disease neuropathology. NeurobiolAging 31:549–566. https://doi.org/10.1016/j.neurobiolaging.2008.05.013
Lieb W, Beiser AS, Vasan RS, Tan ZS, Au R, Harris TB, Roubenoff R, Auerbach S, De Carli C, Wolf PA, Seshadri S (2009) Association of plasma leptin levels with incident Alzheimer disease and MRI measures of brain aging. JAMA 302:2565–2572. https://doi.org/10.1001/jama.2009.1836
Limke TL, Rao MS (2002) Neuralstem cells in aging and disease. J Cell Mol Med 6:475–496. https://doi.org/10.1111/j.1582-4934.2002.tb00451.x
Lin MT, Beal MF (2006) Alzheimer’s APP mangles mitochondria. Nat Med 12:1241–1243
Liu R, Lei JX, Luo C, Lan X, Chi L, Deng P, Lei S, Ghribi O, Liu QY (2012) Increased EID1 nuclear translocation impairs synaptic plasticity and memory function associated with pathogenesis of Alzheimer’s disease. Neurobiol Dis 45:902–912
Liu X, Jiao B, Shen L (2018) The Epigenetics of Alzheimer's Disease: Factors and Therapeutic Implications. Front Genet 9:579. https://doi.org/10.3389/fgene.2018.00579
Livingston G, Sommerlad A, Orgeta V, Costafreda SG, Huntley J, Ames D, Ballard C, Banerjee S, Burns A, Cohen-Mansfield J, Cooper C, Fox N, Gitlin LN, Howard R, Kales HC, Larson EB, Ritchie K, Rockwood K, Sampson EL, Samus Q, Schneider LS, SelbækG TL, Mukadam N (2017) Dementia prevention, intervention, and care. Lancet 390:2673–2734
Lovell MA, Xiong S, Xie C, Davies P, Markesbery WR (2004) Induction of hyperphosphorylated tau in primary rat cortical neuron cultures mediated by oxidative stress and glycogen synthase kinase-3. J Alzheimer’s Dis 6:659–671
Maldonado H, Ramírez E, Utreras E, Pando ME, Kettlun AM, Chiong M, Kulkarni AB, Collados L, Puente J, Cartier L, Valenzuela MA (2011) Inhibition of cyclin-dependent kinase 5 but not of glycogen synthase kinase 3-β prevents neurite retraction and tau hyperphosphorylation caused by secretable products of human T-cell leukemia virus type I-infected lymphocytes. J Neurosci Res 89:1489–1498
Manczak M, Park BS, Jung Y, Reddy PH (2004) Differential expression of oxidative phosphorylation genes in patients with Alzheimer’s disease: implications for early mitochondrial dysfunction and oxidative damage. Neuromolecular Med 5:147–162
Mandelkow EM, Stamer K, Vogel R, Thies E, Mandelkow E (2013) Clogging of axons by tau, inhibition of axonal traffic and starvation of synapses. Neurobiol 24:1079–1085
Mapstone M, Cheema AK, Fiandaca MS, Zhong X, Mhyre TR, LH MA, Hall WJ, Fisher SG, Peterson DR, Haley JM, Nazar MD, Rich SA, Berlau DJ, Peltz CB, Tan MT, Kawas CH, Federoff HJ (2014) Plasma phospholipids identify antecedent memory impairment in older adults. Nat Med 20:415–418
Marfany A, Sierra C, Camafort M, Domenech M, Coca A (2018) High blood pressure, Alzheimer disease and hypertensive treatment. Panminerva Med 60:8–16
María CL, Verónica BD, Agata FG, Blas F, Thomas W, Eduardo MC, Einar MS, Laura M (2006) Plaque-associated overexpression of insulin-degrading enzyme in the cerebral cortex of aged transgenic Tg2576 mice with Alzheimer pathology. J Neuropathol Exp Neurol 65:976–987
Margles FL (1994) Carnosine in the primary olfactory pathway. Science 184:909–911
Martin B, Ji S, Maudsley S, Mattson MP (2010) Control laboratory rodents are metabolically morbid: Why it matters. PNAS 107:6127–6133. https://doi.org/10.1073/pnas.0912955107
Martin SL, Hardy TM, Tollefsbol TO (2013) Medicinal chemistry of the epigenetic diet and caloric restriction. Curr Med Chem 20:4050–4059
Martinez-Pastor B, Cosentino C, Mostoslavsky R (2013) A Tale of Metabolites: The Cross-Talk between Chromatin and Energy Metabolism. Cancer Discovery 3:497–501
Martins RN, Harper CG, Stokes GB, Masters CL (1986) Increased cerebral glucose-6-phosphate dehydrogenase activity in Alzheimer’s disease may reflect oxidative stress. J Neurochem 46:1042–1045
Masoro EJ (1998) Caloric restriction. Aging (Milano) 10:173–174
Matthews DC, Davies M, Murray J et al (2014) Physical activity, Mediterranean diet and biomarkers-assessed risk of Alzheimer’s: a multi-modality brain imaging study. Advances in Molecular Imaging 4(4):43–57
Mehta D, Jackson R, Paul G, Shi J, Sabbagh M (2017) Why do trials for Alzheimer’s disease drugs keep failing? A discontinued drug perspective for 2010–2015. Expert Opin Investig Drugs 26:735–739. https://doi.org/10.1080/13543784.2017.1323868
Meneses A (2015) Serotonin, neural markers, and memory. Front Pharmacol 6:1–22. https://doi.org/10.3389/fphar.2015.00143
Michelle E, Libby SG (2019) Epigenetic Modifications in Alzheimer’s. Neuropathol Ther Front Neurosci 13:476
Mo J-A, Lim J-H, Sul A-R, Lee M, Youn YC, Kim H-J (2015) Cerebrospinal fluid β-Amyloid1–42 levels in the differential diagnosis of Alzheimer’s disease-systemic review and meta-analysis. PLoS ONE 10:e0116802. https://doi.org/10.1371/journal.pone.0116802
Mobbs CV, Yen K, Hof PR (2007) Mechanisms of dietary restriction in aging and disease. Interdiscip Top Gerontol 35:159–175
Montaron MF, Petry KG, Rodriguez JJ, Marinelli M, Aurousseau C, Rougon G, Moal ML (1999) Adrenalectomy increases neurogenesis but not PSA-NCAM expression in aged dentate gyrus. Eur J Neurosci 11:1479–1485. https://doi.org/10.1046/j.1460-9568.1999.00579.x
Morris MC, Tangney CC, Wang Y, Sacks FM, Bennett DA, Aggarwal NT (2015) MIND diet associated with reduced incidence of Alzheimer’s disease. Alzheimer's & Dementia 11:1007–1014
Morrison LD, Smith DD, Kish SJ (1996) Brain S-adenosylmethionine levels are severely decreased in Alzheimer’s disease. J Neurochem 67:1328–1331
Mouton PR, Chachich ME, Quigley C, Spangler E, Ingram DK (2009) Caloric restriction attenuates amyloid deposition in middle-aged dtg APP/PS1 mice. NeurosciLett 464: 184–187
Mucke L, Selkoe D (2012) Neurotoxicity of amyloid b-protein: synaptic and network dysfunction. Cold Spring HarbPerspect Med 2:a006338
Mullard A (2019) Anti-amyloid failures stack up as Alzheimer antibody flops. Nature Reviews Drug Discovery 18:327. https://doi.org/10.1038/d41573-019-00064-1
Mullins RJ, Diehl TC, Chia CW, Kapogiannis D (2017) Insulin resistance as a link between amyloid-beta and tau pathologies in Alzheimer’s disease. Front Aging Neurosci:9–118
Murer MG, Yan Q, Raisman-Vozari R (2001) Brain-derived neurotrophic factor in the control human brain, and in Alzheimer’s disease and Parkinson’s disease. Prog Neurobiol 63:71–124
Naj AC, Jun G, Beecham GW, Wang LS, Vardarajan BN, Buros J, Gallins PJ, Buxbaum JD, Jarvik GP, Crane PK, Larson EB, Bird TD, Boeve BF, Graff-Radford NR, De Jager PL, Evans D, Schneider JA, Carrasquillo MM, Ertekin-Taner N, Younkin SG, Cruchaga C, Kauwe JS, Nowotny P, Kramer P, Hardy J, Huentelman MJ, Myers AJ, Barmada MM, Demirci FY, Baldwin CT, Green RC, Rogaeva E, St George-Hyslop P, Arnold SE, Barber R, Beach T, Bigio EH, Bowen JD, Boxer A, Burke JR, Cairns NJ, Carlson CS, Carney RM, Carroll SL, Chui HC, Clark DG, Corneveaux J, Cotman CW, Cummings JL, DeCarli C, DeKosky ST, Diaz-Arrastia R, Dick M, Dickson DW, Ellis WG, Faber KM, Fallon KB, Farlow MR, Ferris S, Frosch MP, Galasko DR, Ganguli M, Gearing M, Geschwind DH, Ghetti B, Gilbert JR, Gilman S, Giordani B, Glass JD, Growdon JH, Hamilton RL, Harrell LE, Head E, Honig LS, Hulette CM, Hyman BT, Jicha GA, Jin LW, Johnson N, Karlawish J, Karydas A, Kaye JA, Kim R, Koo EH, Kowall NW, Lah JJ, Levey AI, Lieberman AP, Lopez OL, Mack WJ, Marson DC, Martiniuk F, Mash DC, Masliah E, McCormick WC, McCurry SM, McDavid AN, McKee AC, Mesulam M, Miller BL, Miller CA, Miller JW, Parisi JE, Perl DP, Peskind E, Petersen RC, Poon WW, Quinn JF, Rajbhandary RA, Raskind M, Reisberg B, Ringman JM, Roberson ED, Rosenberg RN, Sano M, Schneider LS, Seeley W, Shelanski ML, Slifer MA, Smith CD, Sonnen JA, Spina S, Stern RA, Tanzi RE, Trojanowski JQ, Troncoso JC, Van Deerlin VM, Vinters HV, Vonsattel JP, Weintraub S, Welsh-Bohmer KA, Williamson J, Woltjer RL, Cantwell LB, Dombroski BA, Beekly D, Lunetta KL, Martin ER, Kamboh MI, Saykin AJ, Reiman EM, Bennett DA, Morris JC, Montine TJ, Goate AM, Blacker D, Tsuang DW, Hakonarson H, Kukull WA, Foroud TM, Haines JL, Mayeux R, Pericak-Vance MA, Farrer LA, Schellenberg GD (2011) Common variants at MS4A4/MS4A6E, CD2AP, CD33 and EPHA1 are associated with late-onset Alzheimer's disease. Nat Genet 43:436–441. doi: https://doi.org/10.1038/ng.801
Narasingappa RB, Javagal MR, Pullabhatla S, Htoo HH, Rao JK, Hernandez J-F, Govitrapong P, Vincent B (2012) Activation of α-secretase by curcumin-amino acid conjugates. BiochemBiophys Res Commun 424:691–696
Nebbioso A, Carafa V, Benedetti R, Altucci L (2012) Trials with epigenetic drugs: an update. MolOncol 6:657–682
Nemoto S, Finkel T (2002) Redox regulation of forkheadproteinsthrough a p66shc-dependent signaling pathway. Science 295:2450–2452
Nemutlu E, Zhang S, Juranic NO, Terzic A, Macura S, Dzeja P (2012) 18O-assisted dynamic metabolomics for individualized diagnostics and treatment of human diseases. Croat Med J 53:529–534. https://doi.org/10.3325/cmj.2012.53.529
Neumann H, Daly MJ (2013) Variant TREM2 as risk factor for Alzheimer’s disease. N Engl J Med 368:182–184
Nicolas G, Charbonnier C, Wallon D, Quenez O, Bellenguez C, Grenier-Boley B, Rousseau S, Richard AC, Rovelet-Lecrux A, Le Guennec K, Bacq D, Garnier JG, Olaso R, et al (2016) SORL1 rare variants: a major risk factor for familial early-onset Alzheimer’s disease. Mol Psychiatry 21:831–836
Nordstedt C, Caporaso GL, Thyberg J, Gandy SE, Greengard P (1993) Identification of the Alzheimer β/A4 amyloid precursor protein in clathrin-coated vesicles purified from PC12 cells. J Biol 268:608–612
Novak P, Schmidt R, Kontsekova E, Zilka N, Kovacech B, Skrabana R, Vince-Kazmerova Z, Katina S, Fialova L, Prcina M, Parrak V, Dal-Bianco P, Brunner M, Staffen W, Rainer M, Ondrus M, Ropele S, Smisek M, Sivak R, Winblad B, Novak M (2017) Safety and immunogenicity of the tau vaccine AADvac1 in patients with Alzheimer’s disease: a randomised, double-blind, placebo-controlled, phase 1 trial. Lancet Neurol 16:123–134
O’Brien RJ, Wong PC (2011) Amyloid precursor protein processing and Alzheimer’s disease. Annu Rev Neurosci 34:185–204
Oh H, Madison C, Baker S, Rabinovici G, Jagust W (2016) Dynamic relationships between age, amyloid-β deposition, and glucose metabolism link to the regional vulnerability to Alzheimer’s disease. Brain 139:2275–2289
Olabarria M, Noristani HN, Verkhratsky A, Rodríguez JJ (2012) Concomitant astroglial atrophy and astrogliosis in a triple transgenic animal model of Alzheimer’s disease. Glia 58:831–838
Oliver SG, Winson MK, Kell DB, Baganz F (1998) Systematic functional analysis of the yeast genome. Trends Biotechnol 16:373–378. https://doi.org/10.1016/S0167-7799(98)01214-1
Osawa S, Funamoto S, Nobuhara M, Wada-Kakuda S, Shimojo M, Yagishita S, Ihara Y (2008) Phosphoinositides suppress gammasecretase in both the detergent-soluble and -insoluble states. J Biol 283:19283–19292. https://doi.org/10.1074/jbc.M705954200
Ott A, Stolk RP, van Harskamp F, Pols HA, Hofman A, Breteler MM (1999) Diabetes mellitus and the risk of dementia: The Rotterdam study. Neurology 53:1937–1942. https://doi.org/10.1212/WNL.53.9.1937
Pant S, Sharma M, Patel K, Caplan S, Carr CM, Grant BD (2009) AMPH-1/Amphiphysin/Bin1 functions with RME-1/Ehd1 in endocytic recycling. Nat Cell Biol 11:1399–1410
Park CR, Seeley RJ, Craft S, Woods SC (2000) Intracerebroventricular insulin enhances memory in a passive-avoidance task. PhyBeh 68:509–514
Patel NV, Gordon MN, Connor KE, Good RA, Engelman RW, Mason J, Morgan DG, Morgan TE, Finch CE (2005) Caloric restriction attenuates Abeta-deposition in Alzheimer transgenic models. Neurobiol Aging 26:995–1000
Pedersen JT, Sigurdsson EM (2015) Tau immunotherapy for Alzheimer’s disease. Trends Mol Med 21:394–402
Peila R, Rodriguez BL, Launer LJ (2002) Type 2 diabetes, APOE gene, and the risk for dementia and related pathologies: The Honolulu-Asia aging study. Diabetes 51:1256–1262
Pericak-Vance MA, Bebout JL, Gaskell PC Jr, Yamaoka LH, Hung WY, Alberts MJ, Walker AP, Bartlett RJ, Haynes CA, Welsh KA, Earl NL, Heyman A, Clark CM, Roses AD (1991) Linkage studies in familial Alzheimer disease: evidence for chromosome 19 linkage. Am J Hum Genet 48:1034–1050
Perry DC et al (2013) Progranulin mutations as risk factors for Alzheimer disease. JAMA neurology 70:774–778
Perry D, Sperling R, Katz R, Berry D, Dilts D, Hanna D, Salloway S, Trojanowski JQ, Bountra C, Krams M, Luthman J, Potkin S, Gribkoff V, Temple R, Wang Y, Carrillo MC, Stephenson D, Snyder H, Liu E, Ware T, McKew J, Fields FO, Bain LJ, Bens C (2015) Building a roadmap for developing combination therapies for Alzheimer’s disease. Expert Rev Neurother 15:327–333
Petit JM, Magistretti PJ (2016) Regulation of neuron-astrocyte metabolic coupling across the sleep-wake cycle. Neuroscience 323:135–156
Pooler AM, Polydoro M, Wegmann S, Nicholls SB, Spires-Jones TL, Hyman BT (2013) Propagation of tau pathology in Alzheimer’s disease: identification of novel therapeutic targets. Alzheimers Res Ther 5(49)
Pottier C, Ravenscroft TA, Brown PH, Finch NA, Baker M, Parsons M, Asmann YW, Ren Y, Christopher E, Levitch D, van Blitterswijk M, Cruchaga C, Campion D, et al (2016) TYROBP genetic variants in early-onset Alzheimer’s disease Neurobiol Aging 48:222 e9–e15
Qian S, Jiang P, Guan XM, Singh G, Trumbauer ME, Hong Y, Howard YC, Van der Ploeg HTV, Zheng H (1998) Mutant human presenilin 1 protects presenilin 1 null mouse against embryonic lethality and elevates Aβ1–42/43 expression. Neuron 20:611–617
Raskin J, Cummings J, Hardy J, Schuh K, Dean RA (2015) Neurobiology of Alzheimer’s disease: Integrated molecular, physiological, anatomical, biomarker, and cognitive dimensions. Curr Alzheimer Res 12:712–722
Reddy PH (2011) Abnormal tau mitochondrialdysfunction, impaired axonal transport of mitochondria, and synaptic deprivation in Alzheimer's disease. Brain Res 1415:136–148
Richetin K, Leclerc C, Toni N, Gallopin T, Pech S, Roybon L, Rampon C (2015) Genetic manipulation of adult-born hippocampal neurons rescues memory in a mouse model of Alzheimer's disease. Brain 138:440–455. https://doi.org/10.1093/brain/awu354
Ridha BH, Barnes J, Bartlett JW, Godbolt A, Pepple T, Rossor MN, Fox NC (2006) Tracking atrophy progression in familial Alzheimer’s disease: a serial MRI study. Lancet Neurol 5:828–834
Rinholm JE, Hamilton NB, Kessaris N, Richardson WD, Bergersen LH, Attwell D (2011) Regulation of oligodendrocyte development and myelination by glucose and lactate. J Neurosci 31:538–548
Rivera EJ, Goldin A, Fulmer N, Tavares R, Wands JR, de la Monte SM (2005) Insulin and insulin-like growth factor expression and function deteriorate with progression of Alzheimer's disease: Link to brain reductions in acetylcholine. J Alzheimers Dis 8:247–268. https://doi.org/10.3233/JAD-2005-8304
Rodríguez-Martín T, Cuchillo-Ibáñez I, Noble W, Nyenya F, Anderton BH, Hanger DP (2013) Tau phosphorylation affects its axonal transport and degradation. Neurobiol Aging 34:2146–2157
Rogaev EI, Sherrington R, Rogaeva EA, Levesque G, Ikeda M, Liang Y, Chi H, Lin K, Holman K, Tsuda T, Mar L, Sorbi S, Nacmias B, Piacentini S, Amaducci L, Chumakov L, Cohen D, Lannfelt L, Fraser PE, Rommens JM, St George-Hyslop PH (1995) Familial Alzheimer's disease in kindreds with missense mutations in a gene on chromosome 1 related to the Alzheimer's disease type 3 gene. Nature 376:775–778
Rogaeva E, Meng Y, Lee JH, Gu Y, Kawarai T, Zou F, Katayama T, Baldwin CT, Cheng R, Hasegawa H, Chen F, Shibata N, Lunetta KL, Pardossi-Piquard R, Bohm C, Wakutani Y, Cupples LA, Cuenco KT, Green RC, Pinessi L, Rainero I, Sorbi S, Bruni A, Duara R, Friedland RP, Inzelberg R, Hampe W, Bujo H, Song YQ, Andersen OM, Willnow TE, Graff-Radford N, Petersen RC, Dickson D, Der SD, Fraser PE, Schmitt-Ulms G, Younkin S, Mayeux R, Farrer LA, St George-Hyslop P (2007) The neuronal sortilin-related receptor SORL1 is genetically associated with Alzheimer disease. Nat Genet 39:168–177. https://doi.org/10.1038/ng1943
Rosenmann H (2013) Immunotherapy for targeting tau pathology in Alzheimer’s disease and tauopathies. Curr Alzheimer Res 10:217–228
Roses AD (1996) Apolipoprotein E alleles as risk factors in Alzheimer's disease. Annu Rev Med 47:387–400
Rudzinski LA, Fletcher RM, Dickson DW, Crook R, Hutton ML, Adamson J, Graff-Radford NR (2008) Early onset familial Alzheimer Disease with spastic paraparesis, dysarthria, and seizures and N135S mutation in PSEN1. Alzheimer Dis AssocDisord 22:299–307
Sanchez JT, Ghelani S, Otto-Meyer S (2015) From development to disease: diverse functions of NMDA-type glutamate receptors in the lower auditory pathway. Neuroscience 285:248–259
Scarmeas N, Stern Y, Tang MX, Mayeux R, Luchsinger JA (2006) Mediterranean diet and risk for Alzheimer’s disease. Annals of Neurology 59:912–921
Scarpa S, Fuso A, D’Anselmi F, Cavallaro RA (2003) Presenilin 1 gene silencing by S-adenosylmethionine: a treatment for Alzheimer disease? FEBS Lett 541:145–148. https://doi.org/10.1016/S0014-5793(03)00277-1
Scheff SW, Price DA, Schmitt FA, Mufson EJ (2006) Hippocampal synaptic loss in early Alzheimer’s disease and mild cognitive impairment. Neurobiol Aging 27:1372–1384
Scheuner D, Eckman C, Jensen M, Song X, Citron M, Suzuki N, Bird TD, Hardy J, Hutton M, Kukull W, Larson E, Levy-Lahad L, Viitanen M, Peskind E, Poorkaj P, Schellenberg G, Tanzi R, Wasco W, Lannfelt L, Selkoe D, Younkin S (1996) Secreted amyloid β-protein similar to that in the senile plaques of Alzheimer's disease is increased in vivo by the presenilin 1 and 2 and APP mutations linked to familial Alzheimer's disease. Nat Med 2:864–870
Schneider L (2020) Insight A resurrection of aducanumab for Alzheimer’s disease. The Lancet Neurology 19:111–112. https://doi.org/10.1016/S1474-4422(19)30480-6
Schnupp JW, King AJ, Smith AL, Thompson ID (1995) NMDA-receptor antagonists disrupt the formation of the auditory space map in the mammalian superior colliculus. J Neurosci 15:1516–1531
Sederberg PB, Schulze-Bonhage A, Madsen JR, Bromfield EB, Litt B, Brandt A, Kahana MJ (2007) Gamma oscillations distinguish true from false memories. PsycholSci 18:927–932
Serot JM, Christman ND, Dubost T, Bene MC, Faure GC (2001) CSF-folate levels are decreased in late onset AD patients. J Neural Transm (Vienna) 108:93–99
Serrano-Pozo A, Frosch MP, Masliah E, Hyman BT (2011) Neuropathological Alterations in Alzheimer Disease. Cold Spring Harb Perspect Med 1:a006189. https://doi.org/10.1101/cshperspect.a006189
Sherrington R, Rogaev EI, Liang Y, Rogaeva EA, Levesque G, Ikeda M, Chi H, Lin C, Li G, Holman K, Tsuda T, Mar L, Foncin J-F, Bruni AC, Montesi MP, Sorbi S, Rainero I, Pinessi L, Nee L, Chumakov I, Pollen D, Brookes A, Sanseau P, Polinsky RJ, Wasco W, Da Silva HAR, Haines JL, Pericak-Vance MA, Tanzi RE, Roses AD, Fraser PE, Rommens JM, George-Hyslop PHS (1995) Cloning of a gene bearing missense mutations in early-onset familial Alzheimer’s disease. Nature 375:754–760
Slattery CF, Beck JA, Harper L, Adamson G, Abdi Z, Uphill J, Campbell T, Druyeh R, Mahoney CJ, Rohrer JD, Kenny J, Lowe J, Leung KK, Barnes J, Clegg SL, Blair M, Nicholas JM, Guerreiro RJ, Rowe JB, Ponto C, Zerr I, Kretzschmar H, Gambetti P, Crutch SJ, Warren JD, Rossor MN, Fox NC, Collinge J, Schott JM, Mead S (2014) R47Hvariant increases risk of typical early-onset Alzheimer’s disease but not of prion or frontotemporal dementia. Alzheimers Dement 10:602.e4–608.e4
Sleegers K et al (2009) Serum biomarker for progranulin-associated frontotemporal lobar degeneration. Annals of neurology 65:603–609
Smith JC, Nielson KA, Woodard JL, Seidenberg M, Durgerian S, Hazlett KE, Figueroa CM, Kandah CC, Kay CD, Matthews MA, Rao SM (2014) Physical activity reduces hippocampal atrophy in elders at genetic risk for Alzheimer’s disease. Front Aging Neurosci 6(61)
Sontag E, Nunbhakdi-Craig V, Sontag JM, Diaz-Arrastia R, Ogris E, Dayal S, Lentz SR, Arning E, Bottiglieri T (2007) Protein phosphatase 2A methyltransferase links homocysteine metabolism with tau and amyloid precursor protein regulation. J Neurosci 27:2751–2759
Sorrells SF, Paredes MF, Cebrian-Silla A, Sandoval K, Qi D, Kelley KW, James D, Mayer S, Chang J, Auguste KI, Chang EF, Gutierrez AJ, Kriegstein AR, Mathern GW, Oldham MC, Huang EJ, Garcia-Verdugo JM, Yang Z, Alvarez-Buylla A (2018) Human hippocampal neurogenesis drops sharply in children to undetectable levels in adults. Nature 555:377–381. https://doi.org/10.1038/nature25975
Spencer JL, Waters EM, Romeo RD, Wood GE, Milner TA, McEwen BS (2008) Uncovering the mechanisms of estrogen effects on hippocampal function. Front Neuroendocrinol 29:219–237
Stebbins GT, Murphy CM (2009) Diffusion tensor imaging in Alzheimer’s disease and mild cognitive impairment. BehavNeurol 21:39–49
Steele ML, Robinson SR (2012) Reactive astrocytes give neurons less support: implications for Alzheimer’s disease. Neurobiol Aging 33:423.e1–423.e13
Steen E, Terry BM, Rivera EJ, Cannon JL, Neely TR, Tavares R, Xu XJ, Wands JR, de la Monte SM (2005) Impaired insulin and insulin-like growth factor expression and signaling mechanisms in Alzheimer's disease–is this type 3 diabetes? J Alzheimers Dis 7:63–80. https://doi.org/10.3233/JAD-2005-7107
Stratman NC, Castle CK, Taylor BM, Epps DE, Melchior GW, Carter DB (2005) Isoform-specific interactions of human apolipoprotein E to an intermediate conformation of human Alzheimer amyloid-beta peptide. Chem Phys Lipids 137:52–61
Strittmatter WJ, Saunders AM, Schmechel D, Pericak-Vance M, Enghild J, Roses AD (1993) Apolipoprotein E: high-avidity binding to beta-amyloid and increased frequency of type 4 allele in late-onset familial Alzheimer disease. Proc Natl Acad Sci USA 90:1977–1981
Sultana R, Poon HF, Cai J, Pierce WM, Merchant M, Klein JB, Markesbery WR, Butterfield DA (2006) Identification of nitrated proteins in Alzheimer’s disease brain using a redox proteomics approach. Neurobiol Dis 22:76–87
Suzuki N, Cheung TT, Cai XD, Odaka A, Otvos L Jr, Eckman C, Golde TE, Younkin SG (1994) An increased percentage of long amyloid beta protein secreted by familial amyloid beta protein precursor (beta APP717) mutants. Science 264:1336–1340
Tan MH, Weldon KI, Albers JJ, Cheung MC, Hauel RJ, Vigne J (1980) Serum HDL-cholesterol, apo-A-I and apo-E levels in patients with abnormal coronary arteries. Clin Invest Med 3:225–232
Tang H, Zhao LZ, Zhao HT, Huang SL, Zhong SM, Qin JK, Chen ZF, Huang ZS, Liang H (2011) Hybrids of oxoisoaporphine tacrine congeners: Novel acetylcholine esterase and acetylcholine esterase-induced β-amyloid aggregation inhibitors. Eur J Med Chem 46:4970–4979
Tanzi RE, Hyman BT (1992) Studies of amyloid beta-protein precursor expression in Alzheimer's disease. Ann NY Acad Sci 640:149–154
Tanzi RE, McClatchey AI, Lamperti ED, Villa-Komaroff L, Gusella JF, Neve RL (1988) Protease inhibitor domain encoded by an amyloid protein precursor mRNA associated with Alzheimer disease. Nature 331:528–530
Tatsuki F, Sunagawa GA, Shi S, Susaki EA, Yukinaga H, Perrin D, Sumiyama K, Ukai-Tadenuma M, Fujishima H, Ohno R-I, Tone D, Ode KL, Matsumoto K, Ueda HR (2016) Involvement of Ca(2+)-dependent hyperpolarization in sleep duration in mammals. Neuron 90:70–85
Tchantchou F, Graves M, Falcone D, Shea TB (2008) S-adenosylmethionine mediates glutathione efficacy by increasing glutathione S-transferase activity: implications for S-adenosyl methionine as a neuroprotective dietary supplement. J Alzheimers Dis 14:323–328
Tebar F, Bohlander SK, Sorkin A (1999) Clathrin assembly lymphoid myeloid leukemia (CALM) protein: localization in endocytic-coated pits, interactions with clathrin, and the impact of over expression on clathrin-mediated traffic. Mol Biol Cell 10:2687–2702
Thorin E (2015) Hypertension and Alzheimer disease: Another brick in the wall of awareness. Hypertension 65:36–38
Tohgi H, Utsugisawa K, Nagane Y, Yoshimura M, Genda Y, Ukitsu M (1999) Reduction with age in methylcytosine in the promoter region −224 approximately −101 of the amyloid precursor protein gene in autopsy human cortex. Brain Res Mol Brain Res 70:288–292
Tomata Y, Larsson SC, Hägg S (2020) Polyunsaturated fatty acids and risk of Alzheimer’s disease: a Mendelian randomization study. Eur J Nutr 59:1763–1766
Tran H, Brunet A, Grenier JM, Datta SR, Fornace AJ Jr, Di Stefano PS, Chiang LW, Greenberg ME (2002) DNA repair pathway stimulated by the fork head transcription factor FOXO3athrough the Gadd45 protein. Science 296:530–534
Trushina E, Dutta T, Persson XM, Mielke MM, Petersen RC (2013) Identification of altered metabolic pathways in plasma and CSF in mild cognitive impairment and Alzheimer’s disease using metabolomics. PLoS One 8:e63644
Tu S, Okamoto S, Lipton SA, Xu H (2014) Oligomeric Aβ-induced synaptic dysfunction in Alzheimer’s disease. Mol Neurodegener 14:48
Tulloch J, Leong L, Thomson Z, Chen S, Lee EG, Keene CD, Millard SP, Yu C-E (2018) Glia-specific APOE epigenetic changes in the Alzheimer’s disease brain. Brain Res 1698:179–186. https://doi.org/10.1016/j.brainres.2018.08.006
Turner PR, O’Connor K, Tate WP, Abraham WC (2003) Roles of amyloid precursor protein and its fragments in regulating neural activity, plasticity and memory. Prog Neurobiol 70:1–32
Ulrich D (2015) Amyloid-β impairs synaptic inhibition via GABA (A) receptor endocytosis. J Neurosci 35:9205–9210
Vakalopoulos C (2017) Alzheimer’s disease: the alternative serotonergic hypothesis of cognitive decline. J Alzheimers Dis 60:859–866
Valls-Pedret C, Sala-Vila A, Serra-Mir M et al (2015) Mediterranean diet and age-related cognitive decline: a randomized clinical trial JAMA. Internal Med 175:1094–1103
Vassar R (2014) BACE1 inhibitor drugs in clinical trials for Alzheimer’s disease. Alzheimers Res Ther 6:89
Wahrle S, Das P, Nyborg AC, McLendon C, Shoji M, Kawarabayashi T, Younkin LH, Younkin SG, Golde TE (2002) Cholesterol-dependent gamma-secretase activity in buoyant cholesterol-rich membrane micro domains. Neurobiol Dis 9:11–23. https://doi.org/10.1006/nbdi.2001.0470
Wang R, Reddy PH (2017) Role of glutamate and NMDA receptors in Alzheimer’s disease. J Alzheimers Dis 57:1041–1048
Wang J, Ho L, Qin W, Rocher AB, Seror I, Humala N, Maniar K, Dolios G, Wang R, Hof PR, Pasinetti GM (2005) Caloric restriction attenuates beta-amyloid neuropathology in a mouse model of Alzheimer’s disease. Faseb J 19:659–661
Wang G, Dinkins M, He Q, Zhu G, Poirier C, Campbell A, Proschel-Mayer M, Bieberich E (2012) Astrocytes secrete exosomes enriched with proapoptotic ceramide and prostate apoptosis response 4 (PAR-4): potential mechanism of apoptosis induction in Alzheimer disease (AD). J Biol Chem 287:21384–21395
Wang J, Yu JT, Tan MS, Jiang T, Tan L (2013) Epigenetic mechanisms in Alzheimer’s disease: implications for pathogenesis and therapy. Ageing Res Rev 12:1024–1041
Wang Z-H, Liu P, Liu X, Yu SP, Wang J-Z, Ye K (2018) Delta-secretase (AEP) mediates tau-splicing imbalance and accelerates cognitive decline in tauopathies. J Exp Med 215:3038–3056
Wei W, Nguyen LN, Kessels HW, Hagiwara H, Sisodia S, Malinow R (2010) Amyloid beta from axons and dendrites reduces local spine number and plasticity. Nat Neurosci 13:190–196
Weisgraber KH (1994) Apolipoprotein E: structure–function rela-tionships. Adv Protein Chem 45:249–302
Whitmer RA, Gunderson EP, Quesenberry CP Jr, Zhou J, Yaffe K (2007) Body mass index in midlife and risk of Alzheimer disease and vascular dementia. Curr Alzheimer Res 4:103–109
Wilkins JM, Trushina E (2018) Application of Metabolomics in Alzheimer’s Disease. Front Neurol 8:1–20
Wilson MR, Easterbrook-Smith SB (2000) Clusterin is a secreted mammalian chaperone. Trends Biochem Sci 25:95–98
Wilson JG, Andriopoulos NA, Fearon DT (1987) CR1 and the cell membrane proteins that bind C3 and C4.A basic and clinical review. Immunol Res 6:92–209
Wu P, Shen Q, Dong S, Xu Z, Tsien JZ, Hu Y (2008) Calorie restriction ameliorates neurodegenerative phenotypes in forebrain-specific presenilin-1 and presenilin-2 double knockout mice. Neurobiol Aging 29:1502–1511
Wyss-Coray T, Rogers J (2012) Inflammation in Alzheimer disease-a brief review of the basic science and clinical literature. Csh Perspect Med 2:a006346
Xia X, Jiang Q, McDermott J, Han J-D (2018) Aging and Alzheimer’s disease: Comparison and associations from molecular to system level. Aging Cell 17:e12802. https://doi.org/10.1111/acel.12802
Yagi T, Kosakai A, Ito D, Okada Y, Akamatsu W, Nihei Y, Nabetani A, Ishikawa F, Arai Y, Hirose N, Okano H, Suzuki N (2012) Establishment of induced pluripotent stem cells from centenarians for neurodegenerative disease research. PLoS One 7:e41572
Yamatsuji T, Matsai T, Okamoto T, Komatsuzaki K, Takeda S, Fukumoto H, Iwatsubo T, Suzuki N, Asami-Odaka A, Ireland S, Kinane TB, Giambarella U, Nishimoto I (1996) G-protein-mediated neuronal DNA fragmentation induced by familial Alzheimer’s disease-associated mutants of APP. Science 272:1349–1352
Yiannopoulou KG, Papageorgiou SG (2013) Current and future treatments for Alzheimer’s disease. TherAdvNeurolDisord 6:19–33. https://doi.org/10.1177/1756285612461679
Yoshikai S, Sasaki H, Doh-ura K, Furuya H, Sakaki Y (1990) Genomic organization of the human amyloid beta-protein precursor gene. Gene 87:257–263
Youdim KA, Martin A, Joseph JA (2000) Essential fatty acids andthe brain: possible health implications. Int J DevNeurosci 18:383–399
Yu J, Xu W, Tan C, Andrieu A, Suckling J, Evangelou E, Pan A, Zhang C, Jia J, Feng L, Kua E, Wang Y, Wang H, Tan M, Li J, Hou X, Wan Y, Tan L, Mok V, Tan L, Dong Q, Touchon J, Gauthier S, Aisen PS, Vellas B (2020) Evidence-based prevention of Alzheimer’s disease: systematic review and meta-analysis of 243 observational prospective studies and 153 randomised controlled trials. J Neurol Neurosurg Psychiatry 91:1201–1209
Zhang CE, Yang X, Li L, Sui X, Tian Q, Wei W, Wang J, Liu G (2014a) Hypoxia-induced tau phosphorylation and memory deficit in rats. Neurodegen Dis 14:107–116
Zhang X, Wu M, Lu F, LuoN HZP, Yang H (2014b) Involvement of alpha7 nAChR signaling cascade in epigallocatechinGallate suppression of beta-amyloid-induced apoptotic cortical neuronal insults. MolNeurobiol 49:66–77
Zhang T, Pang P, Fang Z, Guo Y, Li H, Li X, Tian T, Yang X, Chen W, Shu S, Tang N, Wu J, Zhu H, Pei L, Liu D, Tian Q, Wang J, Wang L, Zhu L-Q, Lu Y (2017a) Expression of BC1 impairs spatial learning and memory in Alzheimer’s disease via APP translation. MolNeurobiol 55:6007–6020. https://doi.org/10.1007/s12035-017-0820-z
Zhang Y, Kim MS, Jia B, Yan J, Zuniga-Hertz JP, Han C, Cai D (2017b) Hypothalamic stem cells control ageing speed partly through exosomalmiRNAs. Nature 548:52–57. https://doi.org/10.1038/nature23282
Zhao YQ, Jordan IK, Lunyak VV (2013) Epigenetics components of aging in the central nervous system. Neurotherapeutics 10:647–663
Acknowledgements
Authors are thankful to the Department of Pharmaceutical Technology, Jadavpur University, Kolkata, India for providing the laboratory space for this theoretical work. The authors are thankful to the University Grants Commission, New Delhi; University of Calcutta, Kolkata; Jadavpur University, Kolkata; Indian Council of Medical Research, New Delhi; Department of Biotechnology, Indian Institute of Science, Bangalore; and Science and Engineering Research Board, Govt. of India, New Delhi, India.
Author information
Authors and Affiliations
Contributions
MKP had the main idea of the article, made substantial contribution in the manuscript corrections and critical revision; SB contributed in the literature search, data analysis, preparation of draft about the physiological aspects, organized the sequence of the whole manuscript, and thoroughly revised the manuscript; AC contributed to the genetics and epigenetics portions in searching the literature, analysis, drafting, corrections and revision; DD made contribution to the biochemical aspects in searching the literature, analysis, and drafting.
Corresponding author
Ethics declarations
Ethical approval
This review article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
All authors declare that there is no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Poddar, M.K., Banerjee, S., Chakraborty, A. et al. Metabolic disorder in Alzheimer’s disease. Metab Brain Dis 36, 781–813 (2021). https://doi.org/10.1007/s11011-021-00673-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00673-z