Abstract
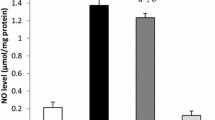
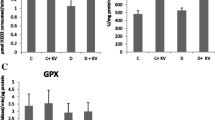
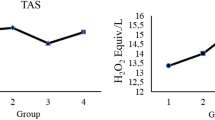
The involvement of oxidative stress in the pathogenesis of diabetes mellitus has been confirmed by numerous studies. In this study, the expression of two antioxidant enzymes, superoxide dismutase (SOD), and catalase which are involved in the detoxification of reactive oxygen species was studied in the streptozotocin-induced diabetic rat liver tissues. The enzyme assays showed a significant decrease in both enzymes activities compared to control animals. The RT-PCR and Western-blot analysis results demonstrated that this decrease in activity is regulated at the level of gene expression, as both catalase and Cu–Zn SOD mRNA and protein expressions were also suppressed. Supplementing the animals with vitamin C, a powerful antioxidant increased both SOD and catalase activities with no change in both mRNA and protein expressions suggesting a role of post-translational modification. However, even though mRNA expressions of both catalase and Cu–Zn SOD were not changed, the protein levels increased in parallel to activities in the case of another antioxidant, α-lipoic acid. An increase in the rate of translation, without changing the rate of transcription indicates a translational effect of lipoic acid in changing the activities of antioxidant enzymes to prevent the oxidative damage in diabetes.









Similar content being viewed by others
References
Hunt JV, Smith CC, Wolff SP (1990) Autoxidative glycosylation and possible involvement of peroxides and free radicals in LDL modification by glucose. Diabetes 39:1420–1424
Wolff SP, Dean RT (1987) Glucose autoxidation and protein modification. The potential role of autoxidative glycosylation in diabetes. Biochem J 245:243–250
Chung SS, Ho EC, Lam KS et al (2003) Contribution of polyol pathway to diabetes-induced oxidative stress. J Am Soc Nephrol 14:233–236
Brownlee M (2001) Biochemistry and molecular cell biology of diabetic complications. Nature 414:813–820
Chance B, Sies H, Boveris A (1979) Hydroperoxide metabolism in mamalian organs. Physiol Rev 59:527–605
Maritim AC, Sanders RA, Watkins JB (2003) Effects of alpha-lipoic acid on biomarkers of oxidative stress in streptozotocin-induced diabetic rats. J Nutr Biochem 14(5):288–294
Padayatty SJ, Daruwala R, Wang YY et al (2002) Vitamin C: from molecular actions to optimum intake. In: Cadenas E, Packer L (eds) Handbook of antioxidants (oxidative stress and disease) CRC, Marcel Dekker, Inc., New York, Basel, Taylor & Francis Group, LLC, 2nd ed. (chapter 7)
Giles AR (1987) Guidelines for the use of animals in biomedical research. Thromb Haemost 58:1078–1084
Lowry OH, Rosebrough NJ, Farr AL et al (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Aebi H (1984) Catalse in vitro. Academy Press, Methods Enzymol, New York 105:121–126
Marklund SL, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47:469–474
Esterbauer H, Cheeseman KH (1990) Determination of aldehydic lipid peroxidation products: malonaldehyde and 4-hidroxynonenal. Methods Enzymol 186:407–421
Levine RL, Garland D, Oliver CN et al (1990) Determination of carbonyl content in oxidatively modified proteins. Methods Enzymol 186:464–478
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA 76(9):4350–4354
Rasband WS (2007) ImageJ. U. S. National Institutes of Health, Bethesda. http://rsb.info.nih.gov/ij/
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162:156–159
MacDonald RJ, Swift GH, Przybyla A (1987) Isolation of RNA using guanidinium salts. Methods Enzymol 152:219–230
Limaye PV, Raghuram N, Sivakami S (2003) Oxidative stress and gene expression of antioxidant enzymes in the renal cortex of streptozotocin-induced diabetic rats. Mol Cell Biochem 243:147–152
Sen CK, Packer L (1996) Antioxidant and redox regulation of gene transcription. FASEB J 10(7):709–720
Strehlow K, Rotter S, Wassmann S, Adam O et al (2003) Modulation of antioxidant enzyme expression and function by estrogen. Circ Res 93(2):170–177
Mayo JC, Sainz RM, Antoli I et al (2002) Melatonin regulation of antioxidant enzyme gene expression. Cell Mol Life Sci 59(10):1706–1713
Maritim A, Dene BA, Sanders RA et al (2003) Effects of pycnogenol treatment on oxidative stress in streptozotocin induced diabetic rats. J Biochem Mol Toxicol 17(3):193–199
Anwar MM, Meki AR (2003) Oxidative stress in streptozotocin-induced diabetic rats: effects of garlic oil and melatonin. Comp Biochem Physiol A 135:539–547
Dinçer Y, Telci A, Kayalı R et al (2002) Effect of α-lipoic acid on lipid peroxidation and antioxidant enzyme activities in diabetic rats. Clin Exp Pharmacol Physiol 29:281–284
Yildirim O, Buyukbingol Z (2002) Effects of supplementation with a combination of cobalt and ascorbic acid on antioxidant enzymes and lipid peroxidation levels in streptozocin diabetic rat liver. Biol Trace Elem Res 90:143–154
Kakkar R, Kalra J, Mantha SV et al (1995) Lipid peroxidation and activity of antioxidant enzymes in diabetic rats. Mol Cell Biochem 151:113–119
Koya D, Hayashi K, Kitada M et al (2003) Effects of antioxidants in diabetes-induced oxidative stress in the glomeruli of diabetic rats. J Am Soc Nephrol 14(3):250–253
Bhor VM, Raghuram N, Sivakami S (2004) Oxidative damage and altered antioxidant enzyme activities in the small intestine of streptozotocin-induced diabetic rats. Int J Biochem Cell Biol 36:89–97
Kagan VE, Shvedova A, Serbinova E et al (1992) Dihydrolipoic acid—a universal antioxidant both in the membrane and in the aqueous phase. Reduction of peroxyl, ascorbyl and chromanoxyl radicals. Biochem Pharmacol 44(8):1637–1649
Obrosova IG, Fathallah L, Liu E, Nourooz-Zadeh J (2003) Early oxidative stress in the diabetic kidney: effect of DL-alpha-lipoic acid. Free Radic Biol Med 34(2):186–195
Moini H, Packer L, Saris NE (2002) Antioxidant and prooxidant activities of alpha-lipoic acid and dihydrolipoic acid. Toxicol Appl Pharmacol 182(1):84–90
Packer L, Kraemer K, Rimbach G (2001) Molecular aspects of lipoc acid in the prevention of diabetic complications. Nutrition 17:888–895
Halliwell B, Gutteridge JMC (1999) Free radicals in biology and medicine, 3rd ed. Clarendon Press, Oxford
Lutsenko EA, Carcamo JM, Golde DW (2002) Vitamin C prevents DNA mutation induced by oxidative stress. J Biol Chem 277(19):16895–16899
Young IS, Torney JJ, Trimble ER (1992) The effect of ascorbate supplementation on oxidative stress in the streptozotocin diabetic rat. Free Radic Biol Med 13(1):41–46
Garg MC, Bansal DD (2000) Protective antioxidant effect of vitamins C and E in streptozotocin induced diabetic rats. Indian J Exp Biol 38(2):101–104
Cao C, Leng Y, Kufe D (2003) Catalase activity is regulated by c-Abl and Arg in the oxidative stress response. J Biol Chem 278(32):29667–29675
Cao C, Leng Y, Liu X et al (2003) Catalase is regulated by ubiquitination and proteosomal degradation. Role of the c-Abl and Arg tyrosine kinases. Biochemistry 42(35):10348–10353
Acknowledgments
The financial support provided by grants from Middle East Technical University (BAP-08-11-DPT2002K120510-TB3) and TUBITAK (106T246-TBAG-HD/175) is gratefully acknowledged. We would also like to thank to our lab mates Deniz İrtem and Erdem Sendinc for their helps.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sadi, G., Yılmaz, Ö. & Güray, T. Effect of vitamin C and lipoic acid on streptozotocin-induced diabetes gene expression: mRNA and protein expressions of Cu–Zn SOD and catalase. Mol Cell Biochem 309, 109–116 (2008). https://doi.org/10.1007/s11010-007-9648-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-007-9648-6