Abstract
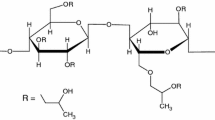
The aim of the present paper was the study of excipient–excipient interactions. It presents the compatibility study between two polymeric compounds used in pharmaceutical technology, namely carboxymethylcellulose and hydroxyethylcellulose, with other commonly used pharmaceutical excipients, such as polyvinylpyrrolidone, sorbitol, magnesium stearate, and talc. The compatibility investigations were carried out under ambient temperature and after decomposition by FTIR spectroscopy studies and later completed by the use of thermal analysis (TG/DTG/HF) data to study the influence of temperature over stability of binary mixtures. Carboxymethylcellulose and hydroxyethylcellulose present thermal-induced interactions with sorbitol and magnesium stearate.



Similar content being viewed by others
References
Karolewicz B. A review of polymers as multifunctional excipients in drug dosage form technology. Saudi Pharm J. 2016;24:525–36.
Elder DP, Kuentz M, Holm R. Pharmaceutical excipients—quality, regulatory and biopharmaceutical considerations. Eur J Pharm Sci. 2016;87:88–99.
Fekete T, Borsa J, Takács E, Wojnárovits L. Synthesis of carboxymethylcellulose/acrylic acid hydrogels with superabsorbent properties by radiation-initiated crosslinking. Radiat Phys Chem. 2016;124:35–139.
Li Z, Wang Y, Pei Y, Xiong W, Xu W, Li B, Li J. Effect of substitution degree on carboxymethylcellulose interaction with lysozyme. Food Hydrocoll. 2017;62:222–9.
Jia W, Cui B, Ye T, Lin L, Zheng H, Yan X, Li Y, Wang L, Liu S, Li B. Phase behavior of ovalbumin and carboxymethylcellulose composite system. Carbohydr Polym. 2014;109:64–70.
De Oliveira RL, Barud HS, De Salvi DTB, Perotti GF, Lima Ribeiro SJ, Regina V, Constantino L. Transparent organic–inorganic nanocomposites membranes based on carboxymethylcellulose and synthetic clay. Ind Crops Prod. 2015;69:415–23.
EL-Haddad MN. Hydroxyethylcellulose used as an eco-friendly inhibitor for 1018c-steel corrosion in 3.5% NaCl solution. Carbohydr Polym. 2014;112:595–602.
Liu X, Chen T, Liu L, Li G. Electrochemical characteristics of heme proteins in hydroxyethylcellulose film. Sens Actuators B. 2006;113:106–11.
Fulias A, Popoiu C, Vlase G, Vlase T, Onetiu D, Savoiu G, Simu G, Patrutescu C, Ilia G, Ledeti I. Thermoanalytical and spectroscopic study on methotrexate—active substance and tablet. Dig J Nanomater Bios. 2014;9:93–8.
Fulias A, Ledeti I, Vlase G, Vlase T. Physico-chemical solid-state characterization of pharmaceutical pyrazolones: an unexpected thermal behaviour. J Pharm Biomed. 2013;81–82:44–9.
Ledeţi I, Ledeţi A, Vlase G, Vlase T, Matusz P, Bercean V, Suta L-M, Piciu D. Thermal stability of synthetic thyroid hormone l-thyroxine and l-thyroxine sodium salt hydrate both pure and in pharmaceutical formulations. J Pharm Biomed Anal. 2016;125:33–40.
Ledeţi I, Vlase G, Ciucanu I, Olariu T, Fuliaş A, Şuta L-M, Belu I. Analysis of solid binary systems containing simvastatin. Rev Chim. 2015;66(2):240–3.
Fuliaş A, Vlase G, Ledeţi I, Şuta L-M. Ketoprofen–cysteine equimolar salt synthesis, thermal analysis, PXRD and FTIR spectroscopy investigation. J Therm Anal Calorim. 2015;121(3):1087–91.
Ledeti A, Vlase G, Ledeti I, Vlase T, Matusz P, Dehelean C, Circioban D, Stelea L, Suta L-M. Thermal stability of desipramine and imipramine. Rev Chim. 2016;67(2):336–8.
Fuliaş A, Vlase G, Vlase T, Şuta L-M, Şoica C, Ledeţi I. Screening and characterization of cocrystal formation between carbamazepine and succinic acid. J Therm Anal Calorim. 2015;121(3):1081–6.
Ledeţi I, Vlase G, Vlase T, Fuliaş A. Kinetic analysis of solid-state degradation of pure pravastatin versus pharmaceutical formulation. J Therm Anal Calorim. 2015;121(3):1103–10.
Fuliaş A, Vlase G, Vlase T, Soica C, Heghes A, Craina M, Ledeti I. Comparative kinetic analysis on thermal degradation of some cephalosporins using TG and DSC data. Chem Cent J. 2013;7(1):70.
De Barros Lima ÍP, Lima NGPB, Barros DMC, Oliveira TS, Mendonça CMS, Barbosa EG, Raffin FN, De Lima E, Moura TFA, Gomes APB, Ferrari M, Aragão CFS. Compatibility study between hydroquinone and the excipients used in semi-solid pharmaceutical forms by thermal and non-thermal techniques. J Therm Anal Calorim. 2015;120(1):719–32.
Teleginski LK, Maciel AB, Mendes C, Silva MAS, Bernardi LS, De Oliveira PR. Fluconazole-excipient compatibility studies as the first step in the development of a formulation candidate for biowaiver. J Therm Anal Calorim. 2015;120(1):771–81.
Karolewicz B, Gajda M, Pluta J, Górniak A. Dissolution study and thermal analysis of fenofibrate-Pluronic F127 solid dispersions. J Therm Anal Calorim. 2015;125(2):751–7.
Ledeti I, Fulias A, Vlase G, Vlase T, Bercean V, Doca N. Thermal behaviour and kinetic study of some triazoles as potential anti-inflammatory agents. J Therm Anal Calorim. 2013;114:1295–305.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vlase, G., Blajovan, R., Albu, P. et al. Study of thermally induced interaction between hydroxyethylcellulose and carboxymethylcellulose with different excipients. J Therm Anal Calorim 130, 397–402 (2017). https://doi.org/10.1007/s10973-017-6309-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-017-6309-1