Abstract
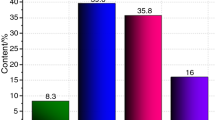
To further understand thermal properties of asphalt binder, four fractions, namely saturates, aromatics, resins and asphaltenes (SARA), were first separated from asphalt binder. Then, physicochemical and pyrolysis properties of SARA fractions were characterized. The results indicate that the contents of aromatics and resins in asphalt binder are higher than that of saturates and asphaltenes. The number-average molecular weights show a successive increase trend from saturates to asphaltenes. Saturates and aromatics contain similar bonds or functional groups, and aromatics contain a smaller number of saturated hydrocarbons than saturates. Also, resins and asphaltenes show several similar transmittance peaks in Fourier transform infrared spectroscopy (FTIR) spectra. Saturates, aromatics and asphaltenes contain more saturated hydrocarbons than that of resins. Further, FTIR spectra of saturates and aromatics are more similar to that of asphalt binder. Some chemical compositions, bonds and functional groups are only shown in FTIR spectra of SARA fractions through which asphalt components can be further understood. Furthermore, differential thermogravimetry curves of asphalt binder and its SARA fractions exhibit obvious unimodal characteristics, indicating that their pyrolysis processes are one-step reaction. The residue yield ratios, the temperatures corresponding to initial decomposition temperature, the main decomposition range and the maximum mass loss rate successively increase from saturates to asphaltenes. It is concluded that the thermal stability of each SARA fraction is successively increased from saturates to asphaltenes.






Similar content being viewed by others
References
Bowers BF, Huang BS, Shu X, Miller BC. Investigation of reclaimed asphalt pavement blending efficiency through GPC and FTIR. Constr Build Mater. 2014;50:517–23.
Gebresellasie K, Lewis JC, Shirokoff J. X-ray spectral line shape analysis of asphalt binders. Energy Fuels. 2013;27:2018–24.
Gong JS, Fu WB, Zhong BJ. A study on the pyrolysis of asphalt. Fuel. 2003;82:49–52.
Tang Q, Zheng YY, Wang TF, Wang JF. Vacuum pyrolysis of plant oil asphalt for transport fuel production catalyzed by alkali metal compounds. Fuel Process Technol. 2014;126:192–8.
Zhang Q, Li QF, Zhang LX, Fang YT, Wang ZQ. Experimental and kinetic investigation of the pyrolysis, combustion, and gasification of deoiled asphalt. J Therm Anal Calorim. 2014;115:1929–38.
Benbouzid M, Hafsi S. Thermal and kinetic analyses of pure and oxidized bitumens. Fuel. 2008;87:1585–90.
Siddiqui MN. Catalytic pyrolysis of Arab heavy residue and effects on the chemistry of asphaltene. J Anal Appl Pyrolysis. 2010;89:278–85.
Yamaguchi K, Sasaki I, Nishizaki I. Effects of film thickness, wavelength and carbon black on photo-degradation of asphalt. J Jpn Pet Inst. 2005;48:150–5.
Wu SP, Mo LT, Cong PL. Flammability and rheological behavior of mixed flame retardant modified asphalt binders. Fuel. 2008;87(1):120–4.
Pei JZ, Wen Y, Li YW. Flame-retarding effects and combustion properties of asphalt binder blended with organo montmorillonite and alumina trihydrate. Constr Build Mater. 2014;72:41–7.
Kok MV, Gul KG. Combustion characteristics and kinetic analysis of Turkish crude oils and their SARA fractions by DSC. J Therm Anal Calorim. 2013;114:269–75.
Cuadri AA, Partal P, Navarro FJ. Bitumen chemical modification by thiourea dioxide. Fuel. 2011;90:2294–300.
Lesueur D. The colloidal structure of bitumen: consequences on the rheology and on the mechanisms of bitumen modification. Adv Colloid Interface Sci. 2009;145:42–82.
Kok MV, Gul KG. Thermal characteristics and kinetics of crude oils and SARA fractions. Thermochim Acta. 2013;569:66–70.
Kok MV, Karacan O, Pamir R. Kinetic analysis of oxidation behavior of crude oil SARA constituents. Energy Fuels. 1998;12:580–8.
Lesueur D, Gerard JF, Claudy P, Letoffe JM, Planche JP, Martin D. A structure related model to describe asphalt linear viscoelasticity. J Rheol. 1996;40:813–36.
Kok MV. Clay concentration and heating rate effect on crude oil combustion by thermogravimetry. Fuel Process Technol. 2012;96:134–9.
Li J, Liu YW, Shi JY. The investigation of thermal decomposition pathways of phenylalanine and tyrosine by TG–FTIR. Thermochim Acta. 2008;467:20–9.
Xu T, Wang HH, Huang XM, Li GF. Inhibitory action of flame retardant on the dynamic evolution of asphalt pyrolysis volatiles. Fuel. 2013;105:757–63.
Braun U, Schartel B, Fichera MA, Jager C. Flame retardancy mechanisms of aluminium phosphinate in combination with melamine polyphosphate and zinc borate in glass–fibre reinforced polyamide 6, 6. Polym Degrad Stab. 2007;92:1528–45.
Baker RR, Coburn S, Liu C, Tetteh J. Pyrolysis of saccharide tobacco ingredients: a TGA–FTIR investigation. J Anal Appl Pyrolysis. 2005;74:171–80.
Jiang ZH, Liu ZJ, Fei BH. The pyrolysis characteristics of moso bamboo. J Anal Appl Pyrolysis. 2012;94:48–52.
Liu Q, Wang S, Zheng Y, Luo Z, Cen K. Mechanism of wood lignin pyrolysis by using TG–FTIR analysis. J Anal Appl Pyrolysis. 2008;82:170–7.
Claudy P, Létoffé JM, King GN, Planche JP. Characterization of asphalts cements by thermomicroscopy and differential scanning calorimetry: correlation to classic physical properties. Fuel Sci Technol Int. 1992;10:735–65.
Xu CM, Gao JS, Zhao SQ, Lin SX. Correlation between feedstock SARA components and FCC product yields. Fuel. 2005;84:669–74.
Sugano M, Iwabuchi Y, Watanabe T. Relations between thermal degradations of SBS copolymer and asphalt substrate in polymer modified asphalt. Clean Technol Environ Policy. 2010;12:653–9.
Zhao S, Kotlyar LS, Sparks BD, Woods JR, Gao J, Chung KH. Solids contents, properties and molecular structures for various asphaltenes from different oilsands. Fuel. 2001;80:1907–14.
Schlepp L, Elie M, Landais P, Romero M. Pyrolysis of asphalt in the presence and absence of water. Fuel Process Technol. 2001;74:107–23.
Radhakrishnan CK, Sujith A, Unnikrishnan G. Thermal behavior of styrene butadiene rubber/poly(ethylene-co-vinyl acetate) blends TG and DSC analysis. J Therm Anal Calorim. 2007;90:191–9.
Yan HW, Cannon WR, Shanefield DJ. Thermal decomposition behaviour of poly (propylene carbonate). Ceram Int. 1998;24:433–9.
Mothe MG, Leite LFM, Mothe CG. Kinetic parameters of different asphalt binders by thermal analysis. J Therm Anal Calorim. 2011;106:679–84.
Sugano M, Iwabuchi Y, Watanabe T. Thermal degradation mechanism of polymer modified asphalt. Chem Eng Trans. 2009;18:1974–9.
Masson JF, Polomark GM, Collins P. Time-dependent microstructure of bitumen and its fractions by modulated differential scanning calorimetry. Energy Fuels. 2002;16:470–6.
Acknowledgements
This study is funded by National College Students’ Innovation and Entrepreneurship Training Program (Grant No. 201310298029) and Ministry of Housing and Urban–Rural Construction of the People’s Republic of China (Grant No. 2013-K5-15) and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD). Also we would like to thank Advanced Analysis & Testing Center of Nanjing Forestry University for the assistance in experiments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, C., Xu, T., Shi, H. et al. Physicochemical and pyrolysis properties of SARA fractions separated from asphalt binder. J Therm Anal Calorim 122, 241–249 (2015). https://doi.org/10.1007/s10973-015-4700-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-015-4700-3