Abstract
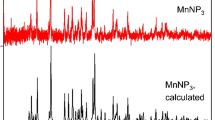
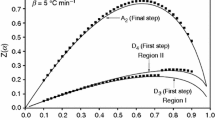
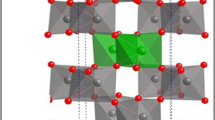
The binary manganese and calcium dihydrogen phosphate monohydrate Mn0.5Ca0.5(H2PO4)2 · H2O was synthesized by a rapid and simple co-precipitation method using phosphoric acid, manganese metal, and calcium carbonate at ambient temperature. Thermal transformation shows complex processes and the final decomposed product was the binary manganese calcium cyclotetraphosphate MnCaP4O12. The activation energies of some decomposed steps were calculated by Kissinger method. Activated complex theory has been applied to each step of the reactions and the thermodynamic functions are calculated. These values for transformation stages showed that they are non-spontaneous processes without the introduction of heat. The differences of physical and chemical properties of the synthesized compound and its decomposed product are compared with M(H2PO4)2 · H2O and M2P4O12 (M = Mn and Ca), which indicate the effects of the presence of Ca ions in substitution of Mn ions and confirm the formation of solid solution.




Similar content being viewed by others
References
Jouini A, Gâcon JC, Ferid M, Trabelsi-Ayadi ML. Luminescence and scintillation properties of praseodymium poly and diphosphates. Opt Mater. 2003;24:175–80.
Kitsugi T, Yamamuro T, Nakamura T, Oka M. Transmission electron microscopy observations at the interface of bone and four types of calcium phosphate ceramics with different calcium/phosphorus molar ratios. Biomaterials. 1995;16:1101–7.
Jian-Jiang B, Dong-Wan K, Kug Sun H. Microwave dielectric properties of Ca2P2O7. J Eur Ceram Soc. 2003;23:2589–92.
Kaplanová M, Trojan M, Brandová D, Navrátil J. On the luminescence on manganese(II) phosphates. J Lumin. 1984;29:199–204.
Wang C-M, Liau H-C, Tsai W-T. Effect of heat treatment on the microstructure and electrochemical behavior of manganese phosphate coating. Mater Chem Phys. 2007;102:207–13.
Antraptseva NM, Shchegrov LN, Ponomareva IG. Thermolysis features of manganese(II) and zinc dihydrogenphosphate solid solution. Russ J Inorg Chem. 2006;51:1493–7.
Trojan M, Brandová D, Paulik F, Arnold M. Mechanism of the thermal dehydration of Co1/2Ca1/2(H2PO4)2·2H2O. J Therm Anal Calorim. 1990;36:929–38.
Trojan M, Brandová D. Mechanism of dehydration of Zn0.5Mg0.5(H2PO4)2·2H2O. Thermochim Acta. 1990;159:1–12.
Brandová D, Trojan M, Paulik F, Paulik J, Arnold M. Study of the thermal dehydration of Mn1/2Ca1/2(H2PO4)2·2H2O. J Therm Anal Calorim. 1990;36:881–9.
Trojan M, Brandová D. Study of thermal dehydration of Mn0.5Mg0.5(H2PO4)2·4H2O. Thermochim Acta. 1990;161:11–21.
Brandová D, Trojan M. Mechanism of dehydration of Zn0.5Mg0.5(H2PO4)2·2H2O. Thermochim Acta. 1990;157:1–9.
Trojan M, Brandová D, Šolc Z. Study of the thermal preparation and stability of tetrametaphosphates of bivalent metals. Thermochim Acta. 1987;110:343–8.
Trojan M. Double tetrametaphosphates Mn2-xCaxP4O12 as special pigments. Dye Pigment. 1990;12:35–47.
Trojan M. Binary cyclotetraphosphates Zn2-xCaxP4O12 as new special pigments. Dye Pigment. 1990;13:1–10.
Trojan M, Šulcová P, MoŠner P. The synthesis of binary zinc(II)−nickel(II)cyclo-tetraphosphates as new special pigments. Dye Pigment. 2000;44:161–4.
Trojan M, Brandová D. A study of the thermal preparation of c-Cd4/3Ca2/3P4O12. Thermochim Acta. 1990;160:349–59.
Trojan M, Šulcová P. Binary Cu(II)−Mn(II) cyclo-tetraphosphates. Dye Pigment. 2000;47:291–4.
Viter VN, Nagornyi PG. Synthesis of continuous substitutional solid solutions M1-xNix(H2PO4)2·2H2O with M = Mg, Mn, Co, or Zn. Russ J Inorg Chem. 2007;52:14–20.
Boonchom B, Maensiri S, Danvirutai C. Soft solution synthesis, non-isothermal decomposition kinetics and characterization of manganese dihydrogen phosphate dihydrate (H2PO4)2·2H2O and its thermal transformation products. Mater Chem Phys. 2008;109:404–10.
Boonchom B, Youngme S, Danvirutai C. A rapid co-precipitation and non-isothermal decomposition kinetics of new binary Mn0.5Co0.5(H2PO4)2·2H2O. Solid State Sci. 2008;10:129–36.
Gabal MA. Kinetics of the thermal decomposition of CuC2O4−ZnC2O4 mixture in air. Thermochim Acta. 2003;402:199–208.
Gao X, Dollimore D. The thermal decomposition of oxalates. Part 26: a kinetic study of the thermal decomposition of manganese (II) oxalate dihydrate. Thermochim Acta. 1993;215:47–63.
Kissinger HE. Reaction kinetics in differential thermal analysis. Anal Chem. 1957;29:1702–6.
Singh BK, Sharma RK, Garg BS. Kinetics and molecular modeling of biologically active glutathione complexes with lead(II) ions. J Therm Anal Calorim. 2006;84:593–600.
Vlaev L, Nedelchev N, Gyurova K, Zagorcheva M. A comparative study of non-isothermal kinetics of decomposition of calcium oxalate monohydrate. J Anal Appl Pyrolysis. 2008;81:253–62.
Vlase T, Jurca G, Doca N. Non-isothermal kinetics by decomposition of some catalyst precursors. Thermochim Acta. 2001;379:65–9.
Vlase T, Vlase G, Doca M, Doca N. Specificity of decomposition of solids in non-isothermal conditions. J Therm Anal Calorim. 2003;72:597–604.
Soumhi EH, Saadoune I, Driss A. A new organic-cation cyclotetraphosphate C10H28N4P4O12·4H2O: crystal structure, thermal analysis, and vibrational spectra. J Solid State Chem. 2001;156:364–9.
Koleva V, Effenberger H. Crystal chemistry of M[PO2(OH)2]2·2H2O compounds (M = Mg, Mn, Fe, Co, Ni, Zn, Cd): structural investigation of the Ni, Zn and Cd salts. J Solid Sate Chem. 2007;180:956–67.
Bagieu-Beucher M, Gondrand M, Perroux M. Etude à haute pression des tétramétaphosphates du type M2P4O12 (M = Ni, Mg, Cu, Co, Fe, Mn, Cd). Données cristallographiques sur tous les composés M2P4O12. J Solid State Chem. 1967;19:353–7.
Hong J, Guo G, Zhang Z. Kinetics and mechanism of non-isothermal dehydration of nickel acetate tetrahydrate in air. J Anal Appl Pyrolysis. 2006;77:111–5.
Ioiţescu A, Vlase G, Vlase T, Doca N. Thermal behaviour of some industrial and food dyes. J Therm Anal Calorim. 2007;88:121–5.
Gabal MA, El-Bellihi AA, Ata-Allah SS. Effect of calcination temperature on Co(II) oxalate dihydrate−iron(II) oxalate dihydrate mixture DTA− TG, XRD, Mössbauer, FT-IR and SEM studies (Part II). Mater Chem Phys. 2003;81:84–92.
Acknowledgements
The authors would like to thank the Chemistry Department, Khon Kaen University for facilities. This work is financially supported by the Thailand Research Fund (TRF) and the Commission on Higher Education (CHE): Research Grant for New Scholar (MRG52_Banjong Boonchom) and King Mongkut’s Institute of Technology Ladkrabang Research (KMITL Research), Ministry of Science and Technology, Thailand.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boonchom, B., Danvirutai, C. Rapid synthesis, kinetics and thermodynamics of binary Mn0.5Ca0.5(H2PO4)2 · H2O. J Therm Anal Calorim 98, 717–723 (2009). https://doi.org/10.1007/s10973-009-0109-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-009-0109-1