Abstract
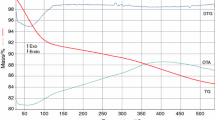
The aim of this paper is to study the gel formation tendency in the TiO2–TeO2–SeO2 system. Telluric acid (H6TeO6), selenous acid (H2SeO3) and titanium butoxide were used as precursors dissolved in ethylene glycol. Transparent monolithic and bright yellow colored gels are situated between 40–100 mol% TiO2, up to 20 mol% SeO2 and 65 mol% TeO2. The phase transformations of the as-prepared gels heat-treated up to 700 °C are investigated by XRD. The chemical states of C, Ti, Se and Te are characterized by means of XPS. Organic and OH groups participate in the amorphous organic–inorganic structure up to 250–300 °C. The structure of the inorganic amorphous phases above 300 °C consists of SeO3, TiO6 and TeOn structural units. The UV–Vis spectra of the binary and three-component gels exhibited a redshift of the cutoff in comparison with those of Ti butoxide gel due to the presence of selenium and tellurium units.
Graphical Abstract
Gel formation region in the ternary TiO2–TeO2–SeO2 system















Similar content being viewed by others
References
El-Malawany R (2002) Tellurite glasses. CRC Press, Physical properties and data
Deb B, Bhattacharya S, Gosh A (2011) EPL 96(3):37005
Bachvarova-Nedelcheva A, Iordanova R, Kostov KL, Ganev V (2012) Opt Mater 34:1781–1787
Bachvarova-Nedelcheva A, Iordanova R, Kostov KL, Ganev V (2014) Opt Mater 36(8):1319–1328
Bingham PA, Connelly AJ, Cassingham NJ, Hyatt NC (2011) J Non-Cryst Solids 357:2726–2734
Tan TTY, Yip CK, Beydoun D, Amal R (2003) Chem Eng J 95:179–186
Zhang P, Sparks DL (1990) Environ Sci Technol 24:1848–1856
Sanuki S, Kojima T, Arai K, Nagaoka S, Majima H (1999) Metall Mater Trans B 30:15–20
Brinker C, Scherer G (1990) Sol-gel science: the physics and chemistry of sol-gel processing. Academic Press Inc, San Diego, CA
Sakka S (2004) Handbook of sol-gel science and technology: processing, characterization and applications, vols I, II, III. Kluwer, Amsterdam
Pierre A, Duboudin F, Tanguy B, Portier J (1994) J Non-Cryst Solids 147&148:569–573
Weng L, Hodgson S, Bao X, Sagoe-Crentsil K (2004) Mater Sci Eng, B 107:89–93
Weng L, Hodgson S (2001) J Mater Sci 36:4955–4959
Hodgson S, Weng L (2002) J Mater Sci 37:3059–3066
Weng L, Hodgson S (2001) Mater Sci Eng, B 87:77–82
Coste S, Lecomte A, Thomas P, Merle-Mejean T, Champarnaud-Mesjard JC (2007) J Sol-Gel Sci Technol 41:79–86
Wei H-Y, Huang W-H, Feng Z-B, Li D-W (2009) Mater Sci Eng, B 164:51–59
Hodgson S, Weng L (2006) J Non-Cryst Solids 297:18–25
Hodgson S, Weng L (2002) J Mater Sci: Mater Electron 17:723–733
Dimitriev Y, Dimitrov V, Arnaudov M (1983) J Mater Sci 18:1353–1358
Dimitriev Y, Dimitrov V, Gatev E, Kashchieva E, Petkov H (1987) J Non-Cryst Solids 95/96:937–944
Dimitriev Y, Bankov A, Ivanova Y, Dimitrov V, Petrakiev A, Tomova M (1980) Build Mater Silic Ind 1:22–26 (in Bulgarian)
Lakov L, Dimitriev Y (1981) Phys Chem Glasses 22(3):69–71
Lakov L, Dimitriev Y (1982) Phys Chem Glass 23:76–78
Bachvarova-Nedelcheva A, Iordanova R, Dimitriev Y, Kashchieva E (2007) J Mater Sci 42(7):3378–3382
Dimitriev Y, Bachvarova-Nedelcheva A, Iordanova R (2008) Mater Res Bull 43:1905–1910
Udovic M, Thomas P, Mirgorodsky A, Durand O, Soulis M, Masson O, Merle-Méjean T, Champarnaud-Mesjard JC (2006) J Solid State Chem 179:3252–3259
Barney ER, Hannon AC, Holland D, Umesaki N, Tatsumisago M, Orman RG, Feller S (2013) J Phys Chem Lett 4:2312–2316
Sabadel J-C, Armand P, Lippens PE, Herreillat DC, Philippot E (1999) J Non-Cryst Solids 244:143–150
Hayakawa T, Koyama H, Nogami M, Thomas Ph (2012) J Univ Chem Techn Metall 47(4):381–386
Zhang S-Y, Chen X, Tian Y, Jin BK, Yang JX (2007) J Crystal Growth 304:42–46
Stengl V, Bakardjieva S, Bludska J (2011) J Mater Sci 46:3523–3536
Nguyen V, Amal R, Beydoun D (2006) J Photochem Photobiol A Chem 179:57–65
Lecomte A, Bamiere F, Coste S, Thomas P, Champarnaud-Mesjard JC (2007) J Eur Ceram Soc 27:1151–1158
Hodgson S, Weng L (2000) J Non-Cryst Solids 276:195–200
Dharma J, Pisal A (2009) Simple method of measuring the band gap energy value of TiO2 in the powder form using a UV/Vis/NIR spectrometer. Application note. PerkinElmer, Shelton, CT
Okáč A (1966) Analytická chemie kvalitativni, Ved. Redactor Čuta F, Academia Nakladatelstvi, Českoslov. Acad. VED, Praha (in Czheck)
Weng L, Hodgson S (2002) Opt Mater 19:313–317
Hodgson S, Weng L (2000) J Sol-Gel Sci Technol 18:145–158
Saylikan F, Asilturk M, Saylikan H, Onal Y, Akarsu M, Aprac E (2005) Turk J Chem 29:697–706
Madarasz J, Braileanu A, Crisan M, Pokol G (2009) J Anal Appl Pyrol 85:549–556
Doeuff S, Henry M, Sanchez C, Livage J (1987) J Non-Cryst Solids 89:206–216
Barboux-Doeuff S, Sanchez C (1994) Mater Res Bull 29:1–13
Zubkova O, Shabadash A (1971) Zh Prikl Spektr 14(5):874–878
Kato K, Tsuge A, Niihara K (1996) J Am Ceram Soc 79(6):1483–1488
Shashikala MN, Elizabeth S, Cary B, Bhat HL (1987) Curr Sci 56:861–863
Ktari L, Dammak M, Mhiri T, Kolsi AW (2002) Phys Chem News 8:01–08
Nakamoto K (1978) IR and Raman spectra of inorganic coordination compounds, 3rd edn. Wiley, London, p 230
Arnaudov M, Dimitrov V, Dimitriev Y (1982) Mater Res Bull 17:1121–1129
Iordanova R, Gegova R, Bachvarova-Nedelcheva A, Dimitriev Y (2015) Phys Chem Glass 56(4):128–138
Falk M, Giguere PA (1958) Can J Chem 36:1680–1685
Gospodinov G, Sukova L, Petrov K (1988) J Inorg Chem 33:1970–1974 (in Russian)
Bachvarova A, Dimitriev Y, Iordanova R (2005) J Non-Cryst Solids 351:998–1002
Uzunova-Bujnova M, Dimitrov D, Radev D, Bojinova A, Todorovsky D (2008) Mater Chem Phys 110:291–298
Beattie IR, Gilson T (1968) Proc R Soc A 307:407–429
Henry M, Leavage J, Sanchez C (1988) Progr Sol State Chem 18:259–341
Velasco MJ, Rubio F, Rubio J, Oteo J (1999) Spectr Lett 32(2):289–304
Yamaguchi O, Tomihisa D, Shimizu K (1988) J Chem Soc, Dalton Trans 564:115–120
Gao X, Wachs IE (1999) Cat Today 51:233–254
Barlier V, Bounor-Legare V, Boiteux G, Davenas J (2008) Appl Surf Sci 254:5408–5412
Nagaveni K, Hegde MS, Ravishankar N, Subbanna GN, Madras G (2004) Langmuir 20:2900–2907
Ngyen Vi VNH, Beydoun D, Amal R (2005) J Photochem Photobiol A Chem 171:113–120
Meng ZD, Zhu L, Ullah K, Ye S, Oh W-Ch (2014) Mater Res Bull 56:45–53
Tripathi K, Husain M et al (2009) Chalcogenide Lett 6(9):517–522
Wang C, Xu B-Q, Wang X, Zhao J (2005) J Solid State Chem 178:3500–3506
Ross-Medgaarden EI, Wachs IE (2007) J Phys Chem C 111:15089–15099
Lu T, Zhang R, Hu Ch, Chen F, Duo Sh, Hu Q (2013) Phys Chem Chem Phys 15:12963–12970
Ohtsu N, Masahashi N, Mizukoshi Y, Wagatsuma K (2009) Langmuir 25(19):11586–11591
Zhong J, Chen F, Zhang J (2010) J Phys Chem C114:933–939
Dai G, Liang S, Liu H, Zhong Z (2013) J Mol Catal A: Chem 368–369:38–42
Ren W, Ai Z, Jia F, Zhang L, Fan X, Zou Z (2007) Appl Catal B Environ 69:138–144
Stankovich S, Dikin D, Piner R, Kohlhaas K, Kleinhammes A, Jia Y, Wu Y, Ngyen ST, Ruoff R (2007) Carbon 45(7):1558–1565
Sachse A, Hulea V, Kostov KL, Belamie E, Alonso B (2015) Catal Sci Technol 5:415–427
Shenasa M, Sainkar S, Lichtman D (1986) J Electr Spectr Rel Phenom 40:329–337
Remy H (1963) Course inorganic chemistry, vol I. Moskva (in Russian)
Häggblad R, Wagner JB, Deniau B, Millet JM, Holmberg J, Grasselli RK, Hansen S, Andersson A (2008) Top Catal 50:52–65
Chang H, Chiang M, Tsai T, Chen T, Whang W, Cheen C (2014) Nanoscale. doi:10.1039/c4nr02765e
Puccetti G, Leblanc RM (1996) J Phys Chem 100:1731–1737
Kallala M, Sanchez C, Cabane B (1993) Phys Rev E 48(5):3692–3704
Sanchez C, Livage J, Henry M, Babonneau F (1988) J Non-Cryst Solids 100:65–76
Khanna M, Wongnawa S (2008) Mater Chem Phys 110:166–175
Zhang Z, Maggard P (2007) J Photochem Photobiol A Chem 186:8–13
Alam MJ, Cameron DC (2002) J Sol-Gel Sci Technol 25:137–145
Svadlak D, Shanelova J, Malek J, Perez-Maqueda L, Criado J, Mitsuhashi T (2004) Thermochim Acta 414:137–143
Hsieh Ch-W, Chiang AST, Lee CC, Yang Sh-J (1992) J Non-Cryst Solids 144:53–62
Li H, Shao G, Chen Z, Song B, Han G (2010) J Am Ceram Soc 93(2):445–449
Shalaby A, Dimitriev Y, Iordanova R, Bachvarova-Nedelcheva A, Iliev T (2011) J Univ Chem Techn Metall 46(2):137–142
Shalaby A, Bachvarova-Nedelcheva A, Iordanova R, Dimitriev Y (2013) J Chem Technol Metall 48(6):585–590
Reznickij LA, Filipova SE (1997) Vestnik Moskovskovo Univ Ser 2 Chem 38(2):132–133 (in Russian)
Dimitriev Y, Dimitrov V, Gatev E, Kashchieva E, Petkov H (1987) J Non-Cryst Solids 95–96:937–944
Hanaor DAH, Sorrell ChC (2011) J Mater Sci 46:855–874
Zhang H, Banfield J (2000) J Phys Chem B 104:3481–3487
Zhang H, Banfield J (1998) J Mater Chem 8(9):2073–2076
Acknowledgments
Authors are grateful to the financial support of The Ministry of Education and Science of Bulgaria, Operational Program “Human Resources Development,” co-financed by the European Social Fund of the European Union, contracts: BGO51PO001-3.3.06-0050.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iordanova, R., Bachvarova-Nedelcheva, A., Gegova, R. et al. Sol–gel synthesis of composite powders in the TiO2–TeO2–SeO2 system. J Sol-Gel Sci Technol 79, 12–28 (2016). https://doi.org/10.1007/s10971-016-4029-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-016-4029-9