Abstract
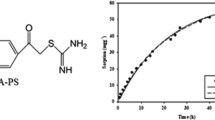
The spherical cross-linked beaded polymers were prepared by condensation of resorcinol and formaldehyde in presence of tri-ethylamine by inverse suspension polymerization technique. The m-cresol, aniline, urea and thiourea were used as co-monomer and polyethylene glycol (PEG 400) was used as porogen. Paraffin oil was used as non-aqueous suspension agent. The polymeric spherical beads were prepared using various types of comonomers exhibiting range of particle size 77.62 to 158.84 μm at 90 °C and 300 rpm for 4 h. The resulting beads were analyzed by elemental analysis, particle size analysis and scanning electron microscope (SEM). The synthesized beads were used for the removal of Cr(VI) from aqueous solutions. A simple and sensitive solid phase extraction procedure was used for the determination of chromium at trace level by spectrophotometric method using 1,5-diphenylcarbazide reagent. The adsorption of Cr(VI) on the resorcinol-formaldehyde beads was monitored by energy-dispersive X-ray spectroscopy (EDX) analysis. The metal adsorption parameters such as contact time, pH, metal ion concentration and adsorbent dose were investigated. For Cr(VI), the maximum adsorption capacity was about 99% at pH 2 for the resorcinol-formaldehyde beads obtained.











Similar content being viewed by others
References
Lahari T, Lakshmi S, Yalangi S, Fadnavis N, Mulani K, Deokar S, Ponrathnam S (2012) Enzyme immobilization on epoxy supports in reverse micellar media: prevention of enzyme denaturation. J Mol Catal B Enzym 74:54–62
Gonte R, Balasubramanian K, Mumbrekar J (2013) Porous and cross-linked cellulose beads for toxic metal ion removal: Hg(II) ions. Journal of Polymers 309:1–9
Okubo M, Mori H (1997) Production of multi-hollow polymer particles by the stepwise acid/alkali method. Colloid Polym Sci 275:634–639
Charles J, Michael D (2002) Hollow latex particles: synthesis and applications. Adv Colloid Interf Sci 99:181
Kim B, Kim J, Suh K (1999) Poly(methyl methacrylate) hollow particles by water-in-oil-in-water emulsion polymerization. Colloid Polym Sci 277:252–256
Yi H, Changyou G, Yanchao S et al (2005) Preparation of porous polylactide microspheres by emulsion-solvent evaporation based on solution induced phase separation. Polym Adv Technol 16:622–627
Bakelite L (1913) The chemical constitution of resinous phenolic condensation products. J Ind Eng Chem 5:506–511
Gould D (1959) Phenolic resins. Reinhold, New York
Eghe A, Ahmed A, Gavin M, Mojtaba M, Mohammad N (2013) Preparation of controlled porosity resorcinol formaldehyde xerogels for adsorption applications. Chem Eng Trans 32:1651–1656
Eduardo V, Philip E, Archie E (1997) An updated review on suspension polymerization. Ind Eng Chem Res 36:939–965
Yuan H, Kalfas G, Ray W (2006) Suspension polymerization. J Macromol Sci Part C Polym Rev 31:215–299
Vivaldo-Lima E, Wood P, Hamielec A, Penlidis A (1997) An updated reviews on suspension polymerization. Ind Eng Chem Res 36:939–965
Dusan B, Jaromir S, Vladimir C (1996) Inverse suspension polymerization of the hydrophilic acrylic monomers in the static continuous phase. J Dispers Sci Technol 18:115–121
Harward D, Hartough P (1947) Modified phenol-formaldehyde resins. US patent 2546946
Haroaki K, Shigeo S (1982) Granular or powdery phenol-aldehyde resins and process for production thereof. US patent 4454298
Angelo P, Laurel M (1952) Reducing-sugar modified aniline-phenol-formaldehyde resins. US patent 2666037
Raymond D, Anthony J (1978) Phenol-aldehyde-amine resin/glycol curative compositions. US patent 4195151
Shrivastava S, Srivastava A, Singh B, Shah D, Verma A, Gutch P (2012) Study on phenolic resin beads: effect of reaction parameters on the properties of polymeric beads. J Appl Polym Sci 123:3741–3747
Singh A, Lal D (2006) Effect of reaction parameters on the particle sizes of crosslinked spherical phenolic beads by suspension polymerization of phenol and formaldehyde. J Appl Polym Sci 100:2323–2330
Zhou H, Huang G, Gao P, Long C (2007) Preparation of porous/hollow particles of phenolic resin. Polym Adv Technol 18:582–585
Dwivedi C, Pathak S, Kumar M, Tripathi S, Bajaj P (2013) Removal of cesium by spherical resorcinol–formaldehyde resin beads: sorption and kinetic studies. J Radioanal Nucl Chem 297:1–8
Wänninen E (1988) The determination of trace metals in natural waters,” ed by T. S. West and H. W. Nürnberg, Blackwell scientific publications, Oxford, London, pp-10
Zouboulis A, Goetz L (1991) Ion flotation as a tool for speciation studies selective separation in the system Cr3+/Cr6+. Toxicol Environ Chem 31–32:539–547
Fernandez Y, Maranon E, Castrillon L, Vazquez I (2005) Removal of cd and Zn from inorganic industrial waste leachate by ion exchange. J Hazard Mater 126:169–175
Kongsricharoern N, Polprasert C (1996) Chromium removal by a bipolar electrochemical precipitation process. Water Sci Technol 34:109–116
Albino Kumar P, Ray M, Chakraborty S (2007) Hexavalent chromium removal from wastewater using aniline formaldehyde condensate coated silica gel. J Hazard Mater 143:24–32
Pearson R (1968) Hard and soft acids, HSAB. Part І. Fundamental principles. J Chem Educ 45:581–587
Zhou L, Liu J, Liu Z (2009) Adsorption of platinum(IV) and palladium(II) from aaqueous solution by thiourea-modified chitosan microspheres. J Hazard Mater 172:439–446
Wang L, Xing R, Liu S, Yu H, Qin Y, Li K, Feng J, Li R, Li P (2010) Recovery of silver (I) using a thiourea-modified chitosan resin. J Hazard Mater 180:577–582
Ertan E, Gulfen M (2009) Separation of gold(III) ions from copper(II) and zinc(II) ions using thiourea-formaldehyde or urea-formaldehyde chelating resins. J Appl Polym Sci 111:2798–2805
Birinci E, Gülfen M, Aydın A (2009) Separation and recovery of palladium(II) from base metal ions by melamine-formaldehyde-thiourea (MFT) chelating resin. Hydrometallurgy 95:15–21
Lam K, Fong C, Yeung K (2007) Separation of precious metals using selective mesoporous adsorbents. Gold Bull 40:192–198
Lam K, Yeung K, McKay G (2006) An investigation of gold adsorption from a binary mixture with selective mesoporous silica adsorbents. J Phys Chem B 110:2187–2194
Tai-Lin L, Hsing-Lung L (2013) Effective and selective recovery of precious metals by Thiourea modified magnetic nanoparticles. Int J Mol Sci 14:9834–9847
Dhore M, Butoliya S, Zade A (2014) Removal of toxic metal ions from water using chelating Terpolymer resin as a function of different concentration time and pH. ISRN Polymer Science Article ID 873520
Talha Gokmen M, Du Prez F (2012) Porous polymer particles- a comprehensive guide to synthesis,characterization, functionalization and applications. Prog Polym Sci 37:365–405
Saralidze K, Koole L, Knetsch M (2010) Polymeric microspheres for medical applications. Materials 3:3537–3564
Mane S, Ponrathnam S, Chavan N (2016) Effect of Porogen concentration on surface area and porous properties of cross linked polymer beads. Can Chem Trans 4:192–200
Vivaldo-Lima E, Wood P, Hamielec A, Penlidis A (1997) An updated review on suspension polymerization. Ind Eng Chem Res 36:939–965
Srivastava S (2009) Co-polymerization of acrylates. Des Monomers Polym 12:1–18
http://en.wikipedia.org/wiki/Polyethylene-glycol. Accesed 26 Aug 2017
Ming G, Kamila G, Stokke B (2015) Swelling dynamics of a DNA-polymer hybrid hydrogel, prepared using polyethylene glycol as a Porogen. Gels 1:219–234
Courtois J, Bystrom E, Irgum K (2006) Novel monolithic materials using poly(ethylene glycol) as porogen for protein separation. Polymer 47:2603–2611
Bajpai K, Shrivastava M (2002) Swelling kinetics of a hydrogel of poly(ethylene glycol) and poly(acrylamide-co-styrene). J Appl Polym Sci 85:1419–1428
Zhihui L, Wentao L, Zhongyuan L, Mingcheng Y, Xujing G, Haitao C, Suqin H, Chengshen Z (2013) Swelling and thermal properties of porous PNIPAM/PEG hydrogels prepared by radiation polymerization. Nucl Sci Tech 24:20201
Akolekar D, Hind A, Bhargava S (1998) Synthesis of Macro-, Meso-, and Microporous Carbons from Natural and Synthetic Sources, and Their Application as Adsorbents for the Removal of Quaternary Ammonium Compounds from Aqueous Solution. J Colloid Interface Sci 199:92–98
Sánchez-Polo M, Rivera-Utrilla J (2002) Adsorbent-adsorbate interactions in the adsorption of cd(II) and Hg(II) on ozonized activated carbons. Environ Sci Technol 36:3850–3854
Mashitah M, Zulfadhly Z, Bhatia S (1999) Binding mechanism of heavy metals biosorption by Pycnoporus sanguineus. J Artif Cells Blood Subst and Immob Biotechnol 27:441–445
Iscen C, Kiran I, Ilhan S (2007) Biosorption of reactive black 5 dye by Penicillium restrictum: the kinetic study. J Hazard Mater 143:335–340
Gao H, Sun Y, Zhou J, Xu R, Daun H (2013) Mussel inspired synthesis of polydopamine-functionalized grapheme hydrogel as reusable adsorbent for water purification. ACS Appl Mater Interfaces 5:425–432
Lagergren S (1898) Zur theorie der sogenannten adsorption geloster stoffe, Kungliga Sevenska Vetenskapsakademiens. Handlingar 24:1–39
Aksu Z (2002) Determination of the equilibrium, kinetic and thermodynamic parameters of the batch biosorption of nickel (II) ions onto Chlorella vulgaris. Process Biochem 38:89–99
Yang R (1999) Gas Separation by Adsorption Processes. – Series on Chemical Engineering, vol 1. Publishers – Imperial College Press, London
Freundlich F, Heller W (1939) The adsorption of cis- and trans-Azobenzen. J Am Chem Soc 21:2228–2230
Mulani K, Daniels S, Rajdeo K, Tambe S, Chavan N (2014) Tannin-aniline-formaldehyde resole resins for arsenic removal from ground water sources. Çanad Chem Transactions 2:450
Freundlich H (1926) Capillary and colloid chemistry. Methuen Co., Ltd., London
Langmuir H (1916) The constitution and fundamental properties of solids and liquids. J Am Chem Soc 38:2221–2295
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mulani, K., Patil, V., Chavan, N. et al. Adsorptive removal of chromium(VI) using spherical resorcinol-formaldehyde beads prepared by inverse suspension polymerization. J Polym Res 26, 41 (2019). https://doi.org/10.1007/s10965-019-1705-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-019-1705-9